Research Article
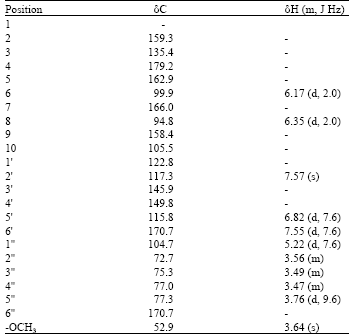
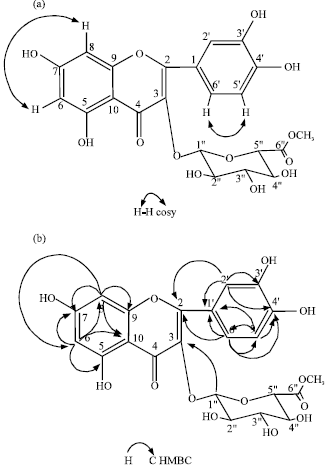
Anti-inflammatory Properties of Quercetin-3-O-β-D-glucuronide-methyl Ester from Polygonum perfoliatum in Mice
Reseach Center for Quality Control of Natural Medicine, Guizhou, Normal University, 550001, Guiyang, China
Xin Zhou
Reseach Center for Quality Control of Natural Medicine, Guizhou, Normal University, 550001, Guiyang, China
Chao Zhao
Reseach Center for Quality Control of Natural Medicine, Guizhou, Normal University, 550001, Guiyang, China
Huaguo Chen
Reseach Center for Quality Control of Natural Medicine, Guizhou, Normal University, 550001, Guiyang, China
Yang Zhao
Reseach Center for Quality Control of Natural Medicine, Guizhou, Normal University, 550001, Guiyang, China