Review Article
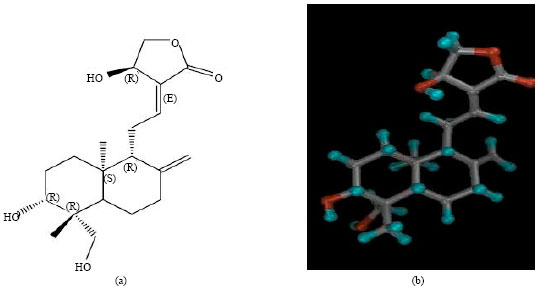
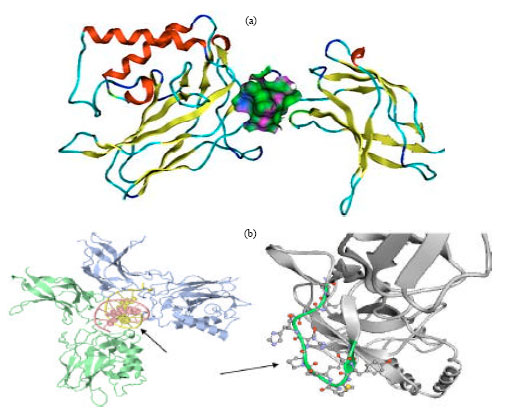
Andrographolide: A Review of its Anti-inflammatory Activity via Inhibition of NF-kappaB Activation from Computational Chemistry Aspects
Faculty of Pharmacy, Universitas Padjadjaran, Jatinangor, West Java, Indonesia
As ari Nawawi
School of Pharmacy, Institut Teknologi Bandung, West Java, Indonesia
Abdul Mutalib
Center for Radioisotope and Radiopharmaca, National Nuclear Agency, Serpong, Indonesia
Slamet Ibrahim
School of Pharmacy, Institut Teknologi Bandung, West Java, Indonesia