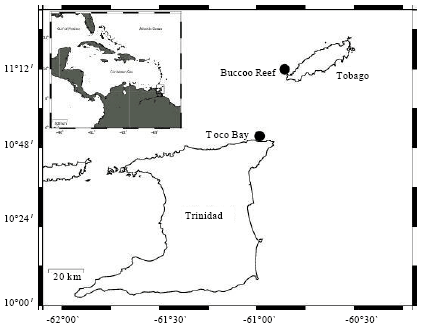
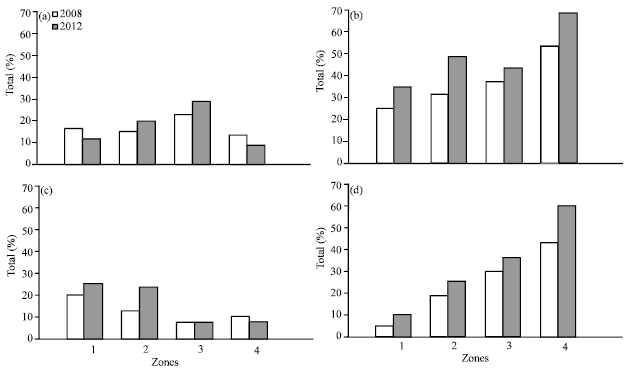
Research Article
Intertidal Distribution Patterns of Zoanthids Compared to their Scleractinian Counterparts in the Southern Caribbean
Department of Curriculum and Teaching, 5040 Haley Center, Auburn University, Auburn, AL. 36849, USA
Dawn A.T. Phillip
Department of Life Sciences, St. Augustine, The University of the West Indies, Trinidad, West Indies