ABSTRACT
Whey, that amazing solution, was considered in the past as a waste byproduct. Recently, scientists had discovered that whey have powerful components; which are whey proteins. Nutritional value of whey proteins had been recognized many years ago. Whey proteins have different fractions, some of them are in large concentration like (β-lactoglobulin, α- lactalbumin, serum albumin, immunoglobulins and glycomacropeptide) and others are in low concentration, such as (lactoferrin, lactoperoxidase, lysozyme and proteose peptone component. Now a days, consumers become more aware of interest, nutritious, healthy foods, has driven much of the research into the healthful effects of whey protein and whey fractions. Whey proteins are magic, potent, promise and active components, which have many various nutritional and therapeutic benefits that encourage to search about them and collect the researches in a review paper to be a guide for all the requires and students. This review is focused on whey protein, its fractions and the therapeutic effect and their application in food processing and pharmaceutical field.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijds.2015.139.159
URL: https://scialert.net/abstract/?doi=ijds.2015.139.159
INTRODUCTION
Milk is a rich source of biologically active compounds which can be used in the production of functional foods. Contemporary consumers have a growing interest in foods which are not only a source of basic nutrition, which also delay and alleviate the consequences of lifestyle diseases (Dziuba and Dziuba, 2014). There is increasing interest in milk and particularly whey, as a potentially rich natural source of bioactive compounds to reduce disease risk and/or to prevent disease development (Huth et al., 2006). Whey is separated during the cheese-making process. It is the liquid remaining after milk has been curdled and strained to remove the caseins. It contains proteins, lactose, vitamins, minerals and traces of fat (Krissansen, 2007). Until 1980, about half of all the produced whey was dumped as waste product into local waterways. However, two events took place to change how whey was addressed: first, more strict environmental laws which forbidding regular dumping of industrial byproducts and second, researchers whom started to investigate all the benefits of whey, which created a new source of revenue (Geiser, 2003).
The discovery of whey as a functional food with nutritional applications elevated whey to a co-product in the manufacturing of cheese (Walzem et al., 2002). Milk contains two primary sources of protein, the casein and whey. Whey proteins account for around 20% of all milk proteins is sold as a nutritional supplement (Krissansen, 2007). On the other hand, Whey proteins do not coagulate under acidic conditions. They are considered to be "Fast proteins" as they reach the jejunum quickly after entering the gastrointestinal tract. After reaching the small intestine, the hydrolysis of whey is slower than that of casein, allowing for greater absorption over the length of the small intestine (Marshall, 2004).
Whey products are evident in salad dressings, infant formulas, emulsifiers, baked goods and medical nutritional formulas (Ha and Zemel, 2003). Whey is also a popular dietary protein supplement purported to provide antimicrobial activity, immune modulation, improved muscle strength and body composition as well as prevents cardiovascular disease and osteoporosis (Marshall, 2004). Whey proteins have been also reported to have utility in many different applications ranging from effects on bone, muscle, blood, brain, pancreas, immune, cancer, infection, metabolism, wound healing and aging (Krissansen, 2007). Emerging research findings largely from in vitro, experimental animals and limited human studies suggest a number of beneficial bioactivities of whey and whey components (Solak and Akin, 2012). Also, whey proteins have increasingly been used as nutritional and functional ingredients in several food formulations, particularly in the form of dry whey, such as Whey Protein Concentrate (WPC) and Whey Protein Isolate (WPI) (Rodrigues et al., 2015).
Whey proteins: Whey is a mixture of proteins, each having unique attributes for nutritional, biological and food ingredient applications. The major proteins present in whey include alpha-lactalbumin (α-La), beta-lactoglobulin (β-Lg), immunoglobulins (IgG), Bovine Serum Albumin (BSA) and glycomacropeptide (GMP). Minor but commercially important proteins are lactoferrin (LF), lactoperoxidase (LP) and lysozyme (Doultani et al., 2004).
Whey proteins contain all the essential amino acids in higher concentrations than vegetable protein sources (Walzem et al., 2002). The amino acids in whey are efficiently absorbed and utilized, relative to free amino acid solutions (Daenzer et al., 2002). They have high concentration of Branched-Chain Amino Acids (BCAAs): Leucine, isoleucine and valine, which are important factors in tissue growth and repair. Leucine is a key amino acid in protein metabolism (Anthony et al., 2001) and has been identified as a key signal in the translation initiation pathway of muscle protein synthesis (Bos et al., 2000). Whey proteins are also rich in the sulfur-containing amino acids cysteine and methionine, which enhance immune function through intracellular conversion to glutathione. Whey is enriched in glutamine, which is a fuel for rapidly dividing cells and has been considered to be "Conditionally-essential" during times of metabolic stress (as experience by endurance athletes) or illness (Krissansen, 2007).
Whey proteins are also a rich source of bioactive peptides generated during their digestion. Bioactive peptides of whey proteins relay their effect by binding to specific receptors in the intestinal lumen before absorption or in target organs after absorption into the bloodstream (Luhovyy et al., 2007). Peptides shorter than four residues can cross intercellular junctions and reach the bloodstream, whereas, larger peptides can be transported via peptide transporter-mediated transport system. The rate of transport is determined by their susceptibility to brush border peptidases (Jakubowicz and Froy, 2013).
Whey protein products: There are four main products of whey proteins which are obtained from various processing techniques. Table 1 provides the composition of whey proteins forms (Shankar and Bansal, 2013), while Table 2 shows the composition of whey proteins products.
Whey Protein Powder (WPP): Whey Protein Powder (WPP) is produced by taking the whey directly from cheese production, then clarified, pasteurized and dried to provide a fine white powder known as whey powder. It has several applications throughout the food industry. It is seen in the food products for confectionery, dairy, beef, snack and bakery products.
| Table 1: | Composition of whey protein forms |
 | |
| (Shankar and Bansal, 2013) | |
| Table 2: | Composition of whey proteins products from Foegeding et al. (2011) |
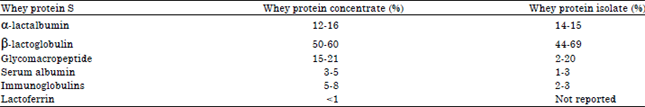 | |
It has many different varieties including acid whey, de-mineralized whey, sweet whey and reduced forms. The reduced and de-mineralized forms are mostly used in the products rather than in the sports supplements (Geiser, 2003; Shankar and Bansal, 2013).
Whey Protein Concentrate (WPC): Whey concentrate typically uses ultra-filtration membrane technology to filter or concentrate whey components based on the membrane pore size and/or molecular weight. The fluid whey is allowed to pass through a semi-permeable membrane (Somaye et al., 2008). This process of whey concentrate will remove the ash, lactose, some minerals and water. In addition to these, when comparing with whey isolates, whey concentrate contains more biologically active proteins and components which make them very attractive supplement for the sports and athlete. Benefits of whey protein concentrate; are great for muscle as well as body building (Shankar and Bansal, 2013).
Whey Protein Isolate (WPI): Whey protein isolate have a protein content of 90% or greater and is produced through a variety of processes like micro-filtration and ion-exchange. Micro-filtration removes additional lactose and fat to increase protein concentration up to 90% or higher. This process maintains the naturally occurring bioactive components in whey. Advanced membrane technology is used for the enrichment of whey concentrate with whey bioactive components such as sphingolipids, lactoferrin (LF), immunoglobulins (Igs), Glycol-Macro-Peptide (GMP) and Transforming Growth Factor-beta (TGF-β) (Le et al., 2005). Benefits of whey protein isolate are:
| • | Lactose free and fat free |
| • | More protein per serving |
| • | Excellent amino acid profile |
| • | Ideal for both fat loss and muscle building (Shankar and Bansal, 2013) |
Hydrolyzed Whey Protein (HWP): The process of hydrolysis breaks down the protein into smaller segments called peptides. This makes the protein easier to digest and reduces the potential for allergic reactions (Geiser, 2003).
The HWP are designed to be applied to nutritional, diabetic and medical foods such as dry mixtures i.e., milk shakes, soups, puddings, omelettes, drinks, nutritional bars, meal replacers and specialized food for sports nutrition, elderly people, diabetic preparations for weight-loss programs and meal replacers for people having disorders of the digestive function of proteolytic enzymes. Protein hydrolyzates is a complex mixture of peptides of different chain lengths and free amino acids that are produced through heating with acid or by addition of proteolytic enzymes amino acids (Manninen, 2009).
Whey protein hydrolyzates are well recognized for the beneficial impacts including improvement in heat stability, enhanced digestibility and absorption, improving the texture of nutritional bars and shelf-life, improvement of foaming and emulsification properties, reduction or elimination of the allergenicity of whey proteins, creation of bioactive peptides (Khan, 2013).
Whey protein fractions: Whey protein fractions are illustrated in Table 3 with some of their properties.
Major whey protein fractions
β-Lactoglobulin (β-Lg): The β-Lg, a member of the lipocalin family, is the most abundant protein in milk and was first discovered in 1934. It has been proposed to participate in the digestion of milk lipids in the neonate by enhancing the activity inhibit this enzyme. It binds to retinol, triglyceride and long-chain fatty acids and enhances their intestinal uptake in pre-ruminant calves. It is the major allergen in cow’s milk, responsible for causing milk allergy. β-Lg has been conjugated with acidic oligosaccharides to reduce its anti-genicity (Krissansen, 2007).
Synthesized in the mammary gland of ruminants (and other species) and designed to be included in milk, this protein has several genetic variants of which β-Lg A is the most common. It is composed mainly of β-sheet motifs and consists of 162 amino acid residues, which lead to a molecular weight of 18300 Da as shows in Table 3. Human milk contains no β-Lg. Besides being a source of essential and branched chain amino acids, a retinol-binding protein has been identified within the β-Lg structure (Marshall, 2004). It binds calcium and zinc and has partial sequence homology to retinol binding proteins. β-Lg has numerous binding sites for minerals, fat-soluble vitamins and lipids and can be used to incorporate desirable lipophilic compounds such as tocopherol and vitamin A into low fat products (Walzem, 1999).
α-Lactalbumin (α-La): The α-La is quantitatively the second most important protein in whey representing 20% (w/w) of the total whey protein inventory and is fully synthesized in the mammary gland. Here, α-La acts as coenzyme for biosynthesis of lactose an important source of energy for the newborn (De Wit, 1989). Seventy percent of protein in human milk is like whey protein and 41% of that protein is α-La. It accounts for 28% of the total protein in human milk. Addition of bovine α-La is strongly advocated to “Humanize” infant formulas and create other products for people with limited or restricted protein intakes (Walzem, 1999). It contains 123 amino acid residues (its sequence is quite homologous to that of lysozyme), which lead to a molecular weight of 14.175 kDa (Jackson et al., 2004).
| Table 3: | Whey protein fractions in bovine whey |
 | |
| Pedersen et al. (2003) and Korhonen (2005), MW: Molecular weight | |
The α-lactalbumin is a protein source that is particularly rich in tryptophan. Evening intake of α-La by human volunteers increased plasma tryptophan availability and improved morning alertness and brain measures of attention. In rats, α-lactalbumin ingestion enhanced serotonin release and induced anxiolytic and rewarding effects, suggesting it has beneficial effects on mood. α-La chelates heavy metals, hence reduces oxidative stress and when orally administered protects against ethanol and stress-induced gastric mucosal injury in rats, suggesting it may have utility as an agent to prevent ulcers. It appears to be effective in inhibiting associations of the pathogens entero-pathogenic E. coli, Salmonella typhimurium and Shigella flexneri with intestinal cells (Krissansen, 2007).
Serum Albumin (SA): The SA is a large protein that makes up approximately 10-15% of total whey protein. It is a source of essential amino acids but there is very little available information regarding its potential therapeutic activity (Gupta et al., 2012). The SA is not synthesized in the mammary gland but appears instead in milk following passive leakage from the blood stream (Fox, 1989). Because of its size and higher levels of structure, BSA can bind to free fatty acids and other lipids as well as flavor compounds (Kinsella et al., 1989), a feature that is severely hampered upon de-naturation. Its heat-induced gelation at pH 6.5 is initiated by an intermolecular thiol-disulphide interchange, similar to what happens with β-Lg (De Wit, 1989).
Immunoglobulins (Igs): The Igs are antibodies or gamma-globulin. There is five classes of antibodies IgA, IgD, IgE, IgG and IgM. All monomeric Igs have the same basic molecular structure, being composed of two identical heavy chains and two identical light chains, with a total molecular mass of approximately 160000 Da. Both the heavy and light chains have constant regions and variable regions. Heavy and light chains are linked together by disulfide bonds, resulting in the classic Y-shape of the immunoglobulin molecule. The number and location of the disulfide bonds is dependent on the class of immunoglobulin. Each immunoglobulin molecule has two antigen binding sites, which comprise the antigen-binding fragment (Fab). The Fab includes the variable amino acid domain. At the other end of the molecule is the constant fragment (Fc), which has a constant amino acid sequence among molecules of the same subclass, which confers the identity of an immunoglobulin as a particular subclass. The Fc region of the molecule is the region that binds to Fc receptors on various cell types (Gapper et al., 2007).
The IgG constitutes approximately 75% of the antibodies in an adult. IgG is transferred from mother to child in utero via cord blood and by breast-feeding and serves as a child’s first line of immune defense referred to as "Passive immunity." IgA is secreted in breast milk and ultimately transferred to the digestive tract in the newborn infant, providing better immunity than a bottle-fed child (Bonang et al., 2000). Colostrum contains significantly greater concentrations of immunoglobulins than mature milk (Table 4). Immunoglobulins reach maximum concentration at the first 24-48 h post-parturition and decline in a time dependent manner following peak concentration (Kholif and El-Loly, 2001; Marshall, 2004). Kholif and El-Loly (2001) noticed significant decrease in milk total Igs and IgM contents (p<0.05) after 24 h postpartum, after 48 h for IgA and after 240 h for IgG contents.
The biological function of milk Igs is to give the offspring an immunological protection against foreign substances such as microbial pathogens, virus and their toxins to help avoid infections in the gastrointestinal tract and respiratory system by stimulating growth of Lactobacillus bifidus and E. coli growth is inhibited (McWilliams, 1986).
| Table 4: | Immunoglobulins concentration in the first milking colostrum and milk |
 | |
| Data compiled from Marnila and Korhonen (2002), Ig: Immunoglobulin, SIgA: Secretory IgA | |
Milk immunoglobulins normally provide passive immunity for the neonate but they are also potentially powerful agents that could be incorporated into diets to remove toxic or undesirable dietary factors (Krissansen, 2007). Furthermore, the immuno-globulins block the action of toxins and viruses.
Glycomacropeptide (GMP): The GMP is also referred to as Casein Macro-Peptide (CMP). The GMP is a protein present in whey at 10-15%, due to the action of chymosin on casein during the cheese making process. It is only present when chymosin is used during processing; therefore, cheeses such as cottage do not produce GMP in the curding process. It is high in branched chain amino acids and lacks the aromatic amino acids including phenylalanine, tryptophan and tyrosine. It is one of the few naturally occurring proteins that lack phenylalanine, making it safe for individuals with phenylketonuria (PKU) (Marshall, 2004).
The GMP has an inhibitory effect on acid gastric secretions and modifies the blood concentration of regulatory digestive peptides. It has been proposed to induce satiety, due to the fact that it induces release of cholecystokinin (Corring et al., 1997) but this notion was not realized in human fed GMP (Gustafson et al., 2001). The GMP inhibits the adhesion of cariogenic bacteria such as Streptococcus spp. mutans, Sanguis and Sobrinus to oral surfaces and it can modify the composition of plaque bacteria to control its acid production and in turn reduce the demineralization of enamel and promote remineralization. The GMP is a source of N-acetyl-necromatic acid and one study showed that dietary intake of GMP can increase the sialic acid content of saliva, with effects on its viscosity and protective function (Gupta et al., 2012).
Minor whey protein fractions
Lactoferrin (LF): Lactoferrin, an iron-binding glycoprotein, is a non-enzymatic antioxidant found in the whey fraction of milk as well as in colostrum (Marshall, 2004). Due to the size and construction, it belongs to the transferrin family, which has a specific ability to bind iron (Legrand et al., 2008). The LF is an iron-chelating, monomeric glycoprotein, characterized by a molecular weight of 80.000 kDa, to which two carbohydrate groups are attached (De Wit, 1989).
Whey lactoferrin is composed of a single polypeptide chain with two binding sites for ferric ions. Before processing, bovine LF is only 15-20% saturated with iron. Iron-depleted lactoferrin, defined as containing less than 5% iron, is referred to as apo-lactoferrin. Human breast milk contains apo-lactoferrin (Gupta et al., 2012).
The LF is a natural defense protein present in most secretions commonly exposed to normal flora including milk, colostrum, tears, nasal secretions, saliva, bile, pancreatic juice, intestinal mucus and genital secretions (Ward et al., 2005). It is secreted by neutrophils and present at high levels at sites of bacterial infection. It is a multifunctional protein that may regulate iron absorption in the intestine, promotes intestinal cell growth, protects against microbial infection, regulates myelopoiesis and regulates systemic immune responses. It is a chameleon in that it can enhance immunity to prevent cancer and yet suppress immunity to block inflammatory disease. The LF when topically applied is a potent regulator of bone cell activity and increases bone formation (Cornish et al., 2004).
Lactoperoxidase (LP) system: The LP is present in a variety of animal secretions, viz. tears, saliva and milk. A member of the family of mammalian per-oxidases, it is one of the most abundant enzymes in plain milk, it represents 1% (w/w) of the total protein pool in whey (Reiter and Perraudin, 1991). The LP, an important enzyme in the whey fractions of milk, is the most abundant enzyme and the majority of it ends up in whey following the curding process. The LP accounts for 0.25-0.5% of total protein found in whey. It has the ability to catalyze certain molecules, including the reduction of hydrogen peroxide. This enzyme system catalyzes peroxidation of thiocyanate and some halides (such as iodine and bromium), which ultimately generates products that inhibit and/or kill a range of bacterial species. During the pasteurization process, it is not inactivated, suggesting its stability as a preservative (Marshall, 2004).
The principal utility of LP is as a protective factor against infectious microbes. Oral administration of LP attenuated pneumonia in influenza virus-infected mice by suppressing the infiltration of inflammatory cells in the lung (Shin et al., 2005).
Lysozyme (LZ): Lysozyme (N-acetylmuramide glycanhydrolase, E.C.3.2.1.17) is a low molecular weight (14.4 kDa) enzymatic protein from the hydrolysis group. It is widely distributed in nature, occurring in many body fluids and tissues of living organisms (Fox and Kelly, 2006). The highest concentration of the enzyme was found in tears and egg white protein, which is currently the basic source of its obtaining on an industrial scale. The relatively large quantities were also noted in human milk. LZ is natural defense mechanism of an organism. Under natural conditions, the antibacterial activity of lysozyme (monomer form) is limited to gram-positive bacteria and only after modification (despite the reduction of hydrolytic activity) its bactericidal activity extends to gram-negative bacteria, including many pathogenic bacteria (Benkerroum, 2008; Lesnierowsk, 2009). It is also one of the mechanisms of non-specific, humoral immune response (Benkerroum, 2008). Antibacterial properties of lysozyme cause a considerable interest in its practical utilization in many sectors of food industries. It is used primarily as an additive to food, showing the preservative properties (Lesnierowski, 2009). The LZ is also used in medical diagnostics, pharmacology and veterinary medicine. The enzyme has found a wide application in the therapies of viral and bacterial infections, treatment of skin as well as eye diseases, periodontitis, leukemia and cancer (Benkerroum, 2008; Zimecki and Artym, 2005).
Also, LZ protects against bacterial infection by breaking down the carbohydrates in bacterial cell walls, killing them. Hence, it is a part of the innate immune response in saliva it protects the oral cavity from pathogens. It also has fungicidal properties, protecting mucosal areas from invasion by pathogenic yeast or fungi. LZ has been shown to inhibit viral replication and infection such HIV. The LZ is thermo-stable, 75% of LZ activity maintains after milk heat treatment 75°C/15 min or 80°C/15 sec (Zagorska and Ciprovica, 2012).
Proteose peptone component 3 (PP3): The proteose peptone fraction of milk has been defined as those proteins that remain in solution after milk has been heated at 95°C/20 min and then acidified to pH 4.7. There are four major components that comprise proteose peptone, with PP3 fragment being the major component at 25% by weight. The PP3 is found only in whey (but not that of humans) and is produced by the fermentation of fat-free bovine milk.
It enhances monoclonal antibody production by human hybridoma cells. Lactophoricin, a synthetic 23 amino acid residue fragment of bovine PP3, inhibits the growth of both gram-positive and negative bacteria (Krissansen, 2007).
Health properties of whey protein fractions: The therapeutic properties of milk proteins have been investigated by many researchers in the past decades. Whey proteins were found to inhibit the growth of tumors more effectively than other food proteins. Food protein peptides can lower blood pressure, inhibit the activity of proline-specific endo-peptidases, stimulate the immune system, act as opioids and opioid antagonists, contract smooth muscles, inhibit blood platelet aggregation, inhibit HIV proteinase and oxidation processes, demonstrate antibacterial and fungicidal activity and surface activity, bind ions, participate in mineral transport, determine sensory properties, improve the nutritional value of foods and control body weight (Dziuba and Dziuba, 2014).
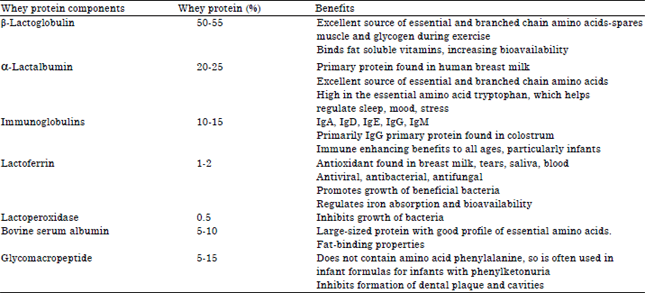
Whey proteins are used in various industries for the supplement of food and beverages. They are also used for medicine purposes and to improve the overall heath (Shankar and Bansal, 2013). Table 5 illustrates some benefits of whey protein fractions as mentioned by Marshall (2004).
Anti-microbial and anti-viral activities of whey proteins: Protein and peptides, in milk, specifically whey, show promise as antibacterial and antiviral modifiers. Whey contains several components which may protect against toxins, bacteria and viruses. These components include Igs, LF and its peptides derivative, lactoferricin, LP, GMP and sphingolipids (Floris et al., 2003). Furthermore, antimicrobial peptides may be generated from whey proteins by proteolysis during gastrointestinal transit (Yalcin, 2006). The LF, α-La and β-Lg have been assayed for inhibitory activity against Human Immunodeficiency Virus type-1 (HIV-1) (Chatterton et al., 2006). In particular, β-Lg may be potential agents for preventing transmission of genital herpes virus infections as well as the spread of HIV (Kokuba et al., 1998).
| Table 5: | Some benefits of whey protein fractions |
 | |
TheLF and lactoferricin inhibit a diverse range of microorganisms including gram-negative bacteria, gram-positive bacteria, yeast, fungi and parasitic protozoa (Takakura et al., 2003). It has been shown to inhibit the growth of some harmful food borne pathogens such as E. coli and Listeria monocytogenes (Floris et al., 2003). The LF also shows significant antiviral activity against HIV, Human Cytomegalovirus (HCMV), herpes viruses, Human Papilloma Virus (HPV), alphavirus and hepatitis C, B and G viruses, among others. In addition, LF is effective against several non-enveloped viruses like rotavirus, entero virus, Palio Virus (PV), adenovirus and Feline Calici Virus (FCV) (Pan et al., 2006). It can enhance the antibacterial activity of lysozme (Ellison and Giehl, 1991). The antimicrobial role of LF is of particular interest to intestinal function and in the prevention of gastro-enteric diseases through control of intestinal micro-flora. While, LF exhibits bactericidal activity against pathogens such as coliforms, it also provides probiotic support for beneficial microorganisms such as bifidobacteria and Lactobacilli ssp, (Yamauchi et al., 2006). Helicobacter pylori is known as the causative agent in the majority of duodenal ulcers. It is believed to be responsible for 50-60% of all gastric carcinomas (Collins et al., 2006).
Whey proteins activate immune cell and/or prevent infection. Whey proteins show promise to help combat rota-viral diarrhea, which is a common infection that results in the death of nearly 500.000 children annually (Wolber et al., 2005). The natural antimicrobial action of LP is being used in a range of oral healthcare products and is finding application in such products directed toward the prevention and treatment of xerostimia (dry mouth). Whey protein concentrate supplementation can possibly decrease the occurrence of associated co-infections (Solak and Akin, 2012).
Anti-oxidant activity: Whey has potent antioxidant activity, likely by contributing cysteine-rich proteins that aid in the synthesis of glutathione (GSH), a potent intracellular antioxidant. The GSH is comprised of glycine, glutamate and cysteine. Cysteine contains a thiol (sulfhydryl) group that serves as an active reducing agent in preventing oxidation and tissue damage (Marshall, 2004). As an antioxidant, glutathione is most effective in its reduced form. The high content of sulfur containing amino acids in whey protein is relevant to its potential to increase the anti-oxidant capacity of the body. The amino acids methionine and cysteine are precursors of glutathione and taurine. Both compounds serve as ‘body’s own’ anti-oxidants. Depletion of GSH results in increased vulnerability of the cell to oxidative stress consequent to the accumulation of intracellular Reactive Oxygen Species (ROS). Maintenance of a high intracellular concentration of GSH is critical for cellular defense against oxidative stress (Schaafsma, 2007).
Collectively, whey proteins may act as a nutritional component to increase endogenous antioxidant enzymes (glutathione peroxidase, catalase and superoxide dismutase) and to reduce oxidative stress markers (MDA, TBARS) jointly with low expression of pro-inflammatory cytokines (IL-1β, IL-6 and TNF-α) in obese, diabetic or stroke patients (Xu et al., 2011; Takayanagi et al., 2011; Sousa et al., 2012).
Immune modulating activity: The immune system is a vast and complex network of cells, organs and molecules that work together to defend the body against foreign microorganisms such as bacteria, parasites and viruses. The immune system has the ability to recognize millions of foreign invaders and anything that triggers an immune response to these invaders is referred to as an antigen (McIntosh et al., 1995).
Whey products and its components are shown to participate in host immunity. Whey contains bioactive components that may offer protection against infections, viruses, enhance immunity and protect against some cancers. In particular, three whey peptides are known to boost the immune system by increasing production of glutathione (α-La, β-Lg and LF). Immune response is the highest in dietary whey proteins. A number of whey proteins (α-La, β-Lg, LF) have been cited for their immunomodulatory effects on the body’s immune system. Immune response of whey proteins (α-La) was higher than casein, soy or whey proteins (Solak and Akin, 2012). The LF is secreted by neutrophils and can stimulate the growth of various cells of the immune defense system including lymphocytes, macrophages/monocytes, humoral immune response and antibody response (Wakabayashi et al., 2003). The LF has shown to play a major role in iron regulation in mammals. Another advantage to the use of iron-saturated LF observed in the human trial was that it did not produce any of the common side effects of iron supplements such as stomach pain and cramps (Uchida et al., 2006).
Anti-carcinogenic activity: Cancer patients undergoing radiation or chemotherapy often have difficulty in meeting their daily nutritional requirements due to nausea and lack of appetite. Whey protein is an excellent protein choice for cancer patients as it is very easy to digest and very gentle to the system (Bounous, 2000). Diets supplemented with LF or with β-Lg enhance protection against the development of putative tumor precursors. The mechanism behind the apparent anticancer activity of dietary whey protein in these studies may be related to their sulfur amino acid contents (cysteine, methionine) (McIntosh et al., 1998).
Whey proteins are more protective against development of intestinal tumors. Dairy proteins, particularly whey offers protection against intestinal induced tumors when compared to other protein sources. Diets containing whey have been shown to reduce intestinal, mammary and colon cancers (Hakkak et al., 2001).
Whey proteins contain other critical components with proven health benefits. Among individual whey proteins, attention has focused on the cancer inhibitory effect of LF and lactoferricin (Tsuda et al., 2002). When human prostate epithelial cells were treated with whey protein isolate, intracellular levels of glutathione dramatically increased (Kent et al., 2003). Whey proteins combat skin cancer. The BSA may also display anticancer activity. Breast cancer in human is inhibited by several commercial BSA preparations during in vitro cell culture (Solak and Akin, 2012). Mader et al. (2005) also demonstrated that the nature of both synthetic and pepsin-generated lactoferricin B is against human leukemia and carcinoma cell lines (Colon, breast and ovary). In an acidic environment, α-lactalbumin forms the HAMLET (human α-lactalbumin made lethal to tumor cells) complex with oleic acid which inhibits the proliferation of various tumors by an apoptosis like mechanism (Svensson et al., 2003).
Anti-inflammation activity: It was observed in overweight and obese subjects that the supplementation of whey proteins (54 g) for 12 weeks did not change the pro inflammatory markers (IL-6, C-reactive protein-CRP and TNF-α). However, in D-galactosamine-induced hepatitis and liver fibrosis in rats, the consumption of whey proteins strongly reduced the plasma levels of pro-inflammatory cytokines (IL-1 beta: 59 and IL-6: 29%) compared to the consumption of the same amount of casein. Collectively, reduction of pro-inflammatory cytokines may be associated with reduction of body weight gain after consumption of whey proteins and it amino acids. Thus, the beneficial effect of whey proteins on hypertension may be mediated by its affect on inflammation as well as the Renin-Angiotensin System (RAS). The ACE-inhibitors used in the management of hypertension have anti-inflammatory properties (Kume et al., 2006; Luhovyy et al., 2007; Sousa et al., 2012).
Cardiovascular disease prevention: Coronary Heart Disease (CHD), the most common and serious form of cardiovascular disease, is the leading cause of death in developed industrialized countries. Many risk factors, both genetic and environmental, contribute to the development of this disease. It is evident that diet is an important modifiable risk factor. Some studies have linked a high fat diet to an increased risk of cardiovascular disease (CVD). Because it is linked to a number of other factors, including increased age, genetics, obesity, sedentary lifestyle and alcohol intake, quality of dietary fat must be taken into consideration (Marshall, 2004). Whey proteins potentially improve cardiovascular health. Milk peptides help lower high blood pressure. Fermented milk with whey protein concentrate lowers triglyceride levels in the blood (Solak and Akin, 2012). Also, whey proteins improve blood pressure and vascular function in overweight and obese individuals (Pal and Ellis, 2010). Some amino acids of the whey proteins, e.g., α-lactalbumin and β-lactoglobulin, are precursors of peptide inhibitors of Angiotensin-Converting Enzyme (ACE) (Luhovyy et al., 2007). The ACE is a key enzyme in the regulation of blood pressure (Pal and Ellis, 2010).
Gastrointestinal support: Whey proteins were reported to be absorbed faster than casein. The lower absorption rate of casein in its native micellar form is because the low pH conditions in the stomach cause casein to clot and delays gastric emptying (Dangin et al., 2001). Therefore, plasma AAs are more rapidly elevated following whey proteins consumption; whereas, changes in plasma AAs are lower and more sustained following micellar casein consumption. Processing of whey proteins or casein fractions via hydrolysis can markedly influence absorption and subsequent plasma AA profiles. Whey proteins have demonstrated a protective effect on the gastric mucosa. This effect is thought to be related to the sulfhydryl component, particularly cysteine and its link with glutamic acid in the production of glutathione. In a study by Rosaneli et al. (2002), rats fed a whey protein concentrate showed a 41% reduction in ulcerative lesions caused by ethanol ingestion while, a 73% reduction rate was observed following repeat doses of whey (Marshall, 2004; McGregor and Poppitt, 2013).
Physical performance: Whey and whey components offer several benefits for individuals with physically active lifestyles. Whey proteins are an excellent source of the essential amino acid, leucine. Leucine is important for athletes as it plays a key role in promoting muscle protein synthesis and muscle growth. Whey proteins are easy to digest protein and are efficiently absorbed into the body. It is often referred to as a "fast" protein for its ability to quickly provide nourishment to muscles (Middleton et al., 2004). Moreover, whey proteins are a rich source of Branched Chain Amino Acids (BCAAs), containing the highest known levels of any natural food source. The BCAAs are important for athletes since unlike the other essential amino acids, they are metabolized directly into muscle tissue and these are the first ones used during periods of exercise and resistance training. Whey proteins also provide the body with BCAAs to replenish decreased levels and start repairing and rebuilding lean muscle tissue. Essential amino acids and whey proteins are equally effective in stimulating muscle protein synthesis in elderly individuals (Paddon-Jones et al., 2006). Whey proteins are rich in the amino acids, arginine and lysine, which may increase the release of growth hormone, a stimulator of muscle growth. Whey proteins can show an increase in lean muscle fiber adaptations (Cribb et al., 2007). Dietary protein and resistance training affect muscle body composition in older persons. Adequate intake of protein combats sarcopenia. Resistance training helps older people gain muscle, hypertrophy muscle and increase whole body fat-free mass (Campbell and Leidy, 2007).
Weight management: Obesity and abnormal lipid levels contribute significantly to the risk of coronary heart disease, a major cardiovascular disease and a serious health problem. Nutritional and dietary therapy, weight loss, exercise and scientifically proven nutritional supplementation might be appropriate to manage dyslipidemia (Houston et al., 2009). High-Protein (HP) diets may reduce body weight gain, fat deposition and improve plasma lipid profile (Lacroix et al., 2004). Furthermore, HP diets have shown to improve hepatic lipid profile in rodent models and in humans ingesting a high-fat diet (Bortolotti et al., 2009). Considerable evidence has recently accumulated to support a beneficial role for dairy products in weight management. Studies also, show that achieving and maintaining a healthy weight can add years to your life and help prevent weight related complications, including diabetes, cancer and heart disease. Diet plays a key role in any weight management program and adding whey proteins often helps make a positive difference (Solak and Akin, 2012). Whey proteins influence on appetite and hunger controlling hormones (Hall et al., 2003). A high-protein diet reduces energy intake and adiposity and that whey proteins are more effective than red meat in reducing body weight gain and increasing insulin sensitivity (Belobrajdic et al., 2004). The meal with α-La preserves lipid oxidation and rapidly delivers amino acids for use during exercise improved the efficiency of exercise training to decrease adiposity (Bouthegourd et al., 2002). Calcium is thought to influence energy metabolism because intracellular calcium regulates adipocyte lipid metabolism and triglyceride storage. Zemel (2002) demonstrated a greater effect of dairy versus nondairy sources of calcium for improving body composition.
Human Immunodeficiency Virus (HIV): Whey protein isolate is rated so highly by the medical profession that it is used to treat HIV patients. Glutathione deficiency is a common problem for individuals infected with HIV. Whey elevates deficient levels of GSH and so provides an extremely important antioxidant involved in the maintenance of functional and structural integrity of muscular tissue undergoing oxidative damage during exercise and aging. The HIV has an antagonistic relationship to GSH, that is, low cellular GSH allows HIV to multiply and high GSH dramatically slows viral replication. Glutathione deficiency is common in individuals infected with HIV. A study by Micke et al. (2002), 30 subjects with HIV were randomized to receive a daily dose of 45 g whey proteins from one of two sources-Protectamin® or Immunocal. The two products have different amino acid profiles and Immunocal is produced at a lower isolation temperature (<72°C). After two weeks of oral supplementation, the Protectamin-supplemented group demonstrated significantly elevated glutathione levels while, the Immunocal group had statistically non-significant elevations.
Hepatitis: Whey proteins supplementations demonstrate variable effects in patients infected with hepatitis B or C. Initially, it was found that bovine lactoferrin prevented Hepatitis C Virus (HCV) infection in vitro in a human hepatocyte line (Ikeda et al., 1998). In an open study on 25 patients diagnosed with either hepatitis B or C, patients were given 12 g whey (Immunocal) twice daily for 12 weeks. Prior to the start of treatment with whey, patients were given 12 g casein protein daily for two weeks. Patients were also given casein following Immunocal for a four-week period. In the 17 patients with HCV, no significant changes were noted. In the group with Hepatitis B Virus (HBV), serum lipid peroxidase levels decreased while IL-2 and NK activity increased. In six of eight HBV patients, serum alanine transferase levels were reduced while, plasma glutathione levels increased in five of the same eight. This trial shows promise for the use of whey protein in the treatment of HBV (Marshall, 2004).
Bone health: Milk has been proposed as a nutritional food that aids in the prevention of osteoporosis due to its bio-available calcium content (Silverwood, 2003). Researchers have begun to examine the different components of milk to determine if a particular isolate is responsible for the bone-protective effects. Initially, in vitro and animal studies determined Milk Basic Protein (MBP), a component of whey, has the ability to stimulate proliferation and differentiation of osteoblastic cells as well as suppress bone resorption. The MBP is prepared from fractionated whey through cation exchange resin. The total protein concentration of MBP is 98%, containing lactoferrin, lactoperoxidase and other minor components. Some whey components (e.g., proteose-peptones) may protect against tooth tissue demineralization and other whey components, because of their immunostimulatory effects, may have favorable effects on dental plaque (Marshall, 2004).
Protein is an important structural component of bone. Some research has shown that protein stimulates the production of insulin-like growth factor-1 (IGF-1), which plays a significant role in bone formation (Dawson-Hughes, 2003). Although, high intakes of protein are known to increase urinary calcium excretion, also more studies have demonstrated that, along with adequate calcium intake, protein and calcium may work synergistically to improve bone mass (Spence and Weaver, 2003). Also, Alexis et al. (2014) mentioned that in the young female, proper calcium and vitamin D intake are keys in the prevention of osteoporosis later in life. Sarcopenia and osteoporosis are dual concerns for the aging female, since both increase the risk of fractures. Sarcopenia prevention can occur through proper nutrition, including sufficient protein intake. Exercise is also important for sarcopenia and osteoporosis prevention. Requirements for proper nutrition vary between patients and a patient specific approach is crucial to overall health in the young and aging female.
Therefore, increased dietary protein can improve bone mineral density in elderly populations already meeting their dietary calcium requirements. With emerging information about protein’s function in bone formation, whey proteins supplementation can play a key role in decreasing bone loss in the elderly. The recommended calcium intake for adults over 51 is 1.2 mg day–1. Whey proteins provide about 500-800 mg of calcium per 100 g protein, depending on the type of whey product considered. Whey proteins can serve as a good source of dietary calcium (Zemel et al., 2000).
Diabetes: Today, most of the people are affected by type 2 diabetes and it is one of the growing health problems over the world. It becomes a concern for teenagers and children. Healthy nutrition practices may play a role in managing and helping the type-2 diabetes. Here, whey proteins are the high biological, a high quality and value protein which is good choice for the people who has diabetics. Whey proteins help to control the blood glucose levels and also provides additional beneficial for weight management which is a concern for type-2 diabetics (Shankar and Bansal, 2013). Ingestion of whey proteins lead to more rapid secretion of insulin than micellar casein (McGregor and Poppitt, 2013).
Aging: Aging is associated with an increased generation of free radicals from everyday biochemical processes and this can lead to oxidative stress. Oxidative stress damages cell membranes and proteins and is responsible for the onset of many of the diseases that are usually associated with aging. An integral role of the immune system is to reduce oxidative stress. Individuals who live a long and healthy life appear to be equipped with optimal cellular defense mechanisms that maintain a robust immune response (Kinney and Allison, 2003; Wu et al., 2004).
Also, aging is accompanied by a progressive decline in skeletal muscle mass, which is known as sarcopenia. Aging leads to loss of neural and muscular function that is associated with progressive and costly disability and dependence. By ages 70-80 years, both men and women experience a 20-40% decrease in muscle strength. Approximately 30% of those 60 years or older have sarcopenia and as our population continues to age, the prevalence is likely to increase. Sarcopenia appears to result from loss of muscle mass rather than loss of strength per unit of muscle. Recent data suggest that the skeletal muscle protein synthetic response to food intake is impaired in older adults. This proposed anabolic resistance is now considered a key factor in the etiology of sarcopenia. The greater anabolic properties of whey than of casein are mainly attributed to the faster digestion and absorption kinetics of whey, which results in a greater increase in postprandial plasma amino acid availability and thereby further stimulates muscle protein synthesis. These findings have led to the development of the "fast" and "slow" protein concept (Doherty, 2003; Tang et al., 2009; Pennings et al., 2011).
Appetite suppression: High protein foods have long been shown to have favorable effects on satiety. Satiety is an important factor in the regulation of food intake and also in the control of obesity (Ropelle et al., 2010). Dietary protein and specific amino acids are involved in the control of gastric and intestinal motility and in pancreatic secretion and are more potent in inducing satiety than carbohydrates or fats (Tome et al., 2009). Amino acids liberated from whey proteins during its in vivo digestion may also stimulate the release of hormones. Insulin secretion mediated by whey ingestion may directly affect food intake regulation by suppressing appetite and as a consequence affect body weight. Indeed, insulin levels stimulated by the ingestion of whey, in addition to modifying the glycemic response, were strongly associated with satiety and decreased food intake. Other hormones are also involved in the regulation of food intake either directly in the hypothalamus, such as ghrelin, or indirectly via the vagal nerve, such as cholecystokinin (CCK) and Peptide YY (PYY) (Jakubowicz and Froy, 2013).
Gut peptides that regulate the digestive process and neuronal signaling in the Central Nervous System (CNS) regulate hunger and satiety. Table 6 lists several peripheral hormones and their roles in the regulation of food intake (Pimentel et al., 2012). Milk serum proteins are more potent stimulants of CCK and GLP-1 than casein. Among the peptides involved with whey protein, glycomacropeptide is an effective secretagogue of CCK. The CCK is a hormone secreted by I cells of the small intestine that has as one of its functions to modulate satiety.
Glycomacropeptide is a powerful stimulator of CCK, which is an appetite suppressing hormone that plays many essential roles relating to gastrointestinal function, including the regulation of food intake.
| Table 6: | Characteristics and functions of hormones related to appetite regulation |
 | |
| CCK: Cholecystokinin, GLP-1: Glucagon-like peptide 1, GIP: Glucose-dependent insulin otropic peptide | |
In addition to being a regulator of food intake, CCK stimulates gall bladder contraction and bowel motility, regulates gastric emptying and stimulates the release of enzymes from the pancreas. Research on the effects of GMP and CCK could lead to the use of certain milk proteins as an appetite suppressant and/or diet aid (Walzem, 1999).
Wound healing: Protein and its amino acids are the building blocks that initiate the growth of new skin during the healing process. Inadequate amounts of protein or diets high in poor quality proteins, such as gelatin, may delay the healing process. Whey proteins are a very high quality protein and are often the preferred choice for high protein products recommended by physician following surgery or burn therapy (Gupta et al., 2012).
Infant nutrition: Whey proteins contain many of the same components found in human breast-milk and for this reason, is a key ingredient in a wide variety of infant formulas, including those for premature infants. Certain types of whey proteins based infant formulas have also been shown to help to reduce crying in colicky infants. While, breast-feeding is preferred, infant formulas containing whey proteins are the next best thing when breast-feeding is not an option. In addition, whey proteins are an excellent protein choice for the expectant mother who needs increased amounts of proteins. Pregnancy can increase the body’s protein needs by up to 33% (Gupta et al., 2012).
CONCLUSION AND FUTURE PERSPECTIVES
| • | Whey proteins improve several risk factors for obesity, hypertension, oxidative stress and diabetes. Also, a relationship between consumption of whey proteins source foods and oxidative stress, hepato-protective effects and increased resting energy expenditure has been suggested |
| • | Large randomized milk protein supplementation trials combined with lifestyle changes are still required however to generate robust evidence supporting the use of milk protein products to improve or manage metabolic health |
| • | Although, the potential of milk proteins and peptides for the formulation of functional foods has been long demonstrated, they are still not mass produced. Therefore, there is a need to develop novel technologies, such as membrane separation, nano-filtration and ultra-filtration |
| • | Macro-encapsulation and nano-encapsulation methods supply new solutions that improve peptide stability in foods and during digestion |
| • | Progress in the commercial exploitation of whey proteins for health and medicine is dependent upon identifying new indications to treat using the existing whey bioactive, finding new commercially viable technologies aimed at fractionating and isolating minor proteins with potent biological activity and modifying the major proteins to either enhance their activities or acquire new functions |
| • | Further studies with application of proteomic, peptidomic and metabolomic method are required to investigate the physiological mechanisms responsible for the biological activity of peptides |
| • | Therapeutic minor milk proteins may have to be commercialized using recombinant technologies to synthesize the human equivalents. Progress will also come from a more thorough investigation of the activities and functions of whey peptides derived from hydrolyzing the major milk proteins |
REFERENCES
- Anthony, J.C., T.G. Anthony, S.R. Kimball and L.S. Jefferson, 2001. Signaling pathways involved in translational control of protein synthesis in skeletal muscle by leucine. J. Nutr., 131: 856S-860S.
Direct Link - Belobrajdic, D.P., G.H. McIntosh and J.A. Owens, 2004. A high-whey-protein diet reduces body weight gain and alters insulin sensitivity relative to red meat in wistar rats. J. Nutr., 134: 1454-1458.
Direct Link - Benkerroum, N., 2008. Antimicrobial activity of lysozyme with special relevance to milk. Afr. J. Biotechnol., 7: 4856-4867.
Direct Link - Bonang, G., H.E. Monintja, Sujudi and D. van der Waaij, 2000. Influence of breastmilk on the development of resistance to intestinal colonization in infants born at the Atma Jaya Hospital, Jakarta. Scand. J. Infect. Dis., 32: 189-196.
CrossRefPubMedDirect Link - Bortolotti, M., R. Kreis, C. Debard, B. Cariou and D. Faeh et al., 2009. High protein intake reduces intrahepatocellular lipid deposition in humans. Am. J. Clin. Nut., 92: 1002-1010.
CrossRefDirect Link - Bounous, G., 2000. Whey protein concentrate (WPC) and glutathione modulation in cancer treatment. Anticancer Res., 20: 4785-4792.
PubMedDirect Link - Bouthegourd, J.C.J., S.M. Roseau, L. Makarios-Lahham, P.M. Leruyet, D.G. Tome and P.C. Even, 2002. A preexercise α-lactalbumin-enriched whey protein meal preserves lipid oxidation and decreases adiposity in rats. Am. J. Physiol.-Endocrinol. Metab., 283: E565-E572.
CrossRefDirect Link - Campbell, W.W. and H.J. Leidy, 2007. Dietary protein and resistance training effects on muscle and body composition in older persons. J. Am. College Nutr., 26: 696S-703S.
CrossRefDirect Link - Chatterton, D.E.W., G. Smithers, P. Roupas and A. Brodkrob, 2006. Bioactivity of β-lactoglobulin and α-lactalbumin-technological implications for processing. Int. Dairy J., 16: 1229-1240.
Direct Link - Cornish, J., K.E. Callon, D. Naot, K.P. Palmano and T. Banovic et al., 2004. Lactoferrin is a potent regulator of bone cell activity and increases bone formation in vivo. Endocrinology, 145: 4366-4374.
CrossRefDirect Link - Cribb, P.J., A.D. Williams, C.G. Stathis, M.F. Carey and A. Hayes, 2007. Effects of whey isolate, creatine and resistance training on muscle hypertrophy. Med. Sci. Sports Exercise, 39: 298-307.
Direct Link - Daenzer, M., K.J. Petzke, B.J. Bequette and C.C. Metges, 2002. Whole-body nitrogen and splanchnic amino acid metabolism differ in rats fed mixed diets containing casein or its corresponding amino acid mixture. J. Nutr., 131: 1965-1972.
PubMedDirect Link - Dangin, M., Y. Boirie, C. Garcia-Rodenas, P. Gachon and J. Fauquant et al., 2001. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am. J. Physiol. Endocrinol. Metab, 280: E340-E348.
PubMedDirect Link - Dawson-Hughes, B., 2003. Interaction of dietary calcium and protein in bone health in humans. J. Nutr., 133: 852S-854S.
Direct Link - Doherty, T.J., 2003. Invited review: Aging and sarcopenia. J. Applied Physiol., 95: 1717-1727.
PubMed - Doultani, S., K.N. Turhan and M.R. Etzel, 2004. Fractionation of proteins from whey using cation exchange chromatography. Process Biochem., 39: 1737-1743.
CrossRefDirect Link - Dziuba, B. and M. Dziuba, 2014. Milk proteins-derived bioactive peptides in dairy products: Molecular, biological and methodological aspects. Acta Sci. Pol. Technol. Aliment, 13: 5-25.
Direct Link - Ellison, R.T. and T.J. Giehl, 1991. Killing of gram-negative bacteria by lactoferrin and lysozyme. J. Clin. Invest., 88: 1080-1091.
CrossRefDirect Link - Floris, R., I. Recio, B. Berkhout and S. Visser, 2003. Antibacterial and antiviral effects of milk proteins and derivatives thereof. Curr. Pharmaceut. Design, 9: 1257-1275.
Direct Link - Fox, P.F. and A.L. Kelly, 2006. Indigenous enzymes in milk: Overview and historical aspects-Part 2. Int. Dairy J., 16: 517-532.
CrossRefDirect Link - Gapper, L.W., D.E.J. Copestake, D.E. Otter and H.E. Indyk, 2007. Analysis of bovine immunoglobulin G in milk, colostrum and dietary supplements: A review. Anal. Bioanal. Chem., 389: 93-109.
CrossRefPubMedDirect Link - Gupta, G., D. Prakash, A.P. Garg and S. Gupta, 2012. Whey proteins: A novel source of bioceuticals. Middle-East J. Sci. Res., 12: 365-375.
Direct Link - Gustafson, D.R., D.J. McMahon, J. Morrey and R. Nan, 2001. Appetite is not influenced by a unique milk peptide: Caseinomacropeptide (CMP). Appetite, 36: 157-163.
CrossRefDirect Link - Ha, E. and M.B. Zemel, 2003. Functional properties of whey, whey components and essential amino acids: Mechanisms underlying health benefits for active people (review). J. Nutr. Biochem., 14: 251-258.
CrossRefDirect Link - Hakkak, R., S. Korourian, M.J.J. Ronis, J.M. Johnston and T.M. Badger, 2001. Dietary whey protein protects against azoxymethane-induced colon tumors in male rats. Cancer Epidemiol. Biomarkers Prevent., 10: 555-558.
PubMedDirect Link - Hall, W.L., D.J. Millward, S.J. Long and L.M. Morgan, 2003. Casein and whey exert different effects on plasma amino acid profiles, gastrointestinal hormone secretion and appetite. Br. J. Nutr., 89: 239-248.
CrossRefDirect Link - Houston, M.C., S. Fazio, F.H. Chilton, D.E. Wise, K.B. Jones, T.A. Barringer and D.A. Bramlet, 2009. Nonpharmacologic treatment of dyslipidemia. Prog. Cardiovasc. Dis., 52: 61-94.
CrossRefPubMedDirect Link - Huth, P.J., D.B. Di Rienzo and G.D. Miller, 2006. Major scientific advances with dairy foods in nutrition and health. J. Dairy Sci., 89: 1207-1221.
CrossRefDirect Link - Ikeda, M., K. Sugiyama, T. Tanaka, K. Tanaka, H. Sekihara, K. Shimotohno and N. Kato, 1998. Lactoferrin markedly inhibits hepatitis C virus infection in cultured human hepatocytes. Biochem. Biophys. Res. Commun., 245: 549-553.
CrossRefDirect Link - Jackson, J.G., D.B. Janszen, B. Lonnerdal, E.L. Lien K.P. Pramuk and C.F. Kuhlman, 2004. A multinational study of α-lactalbumin concentrations in human milk. J. Nutr. Biochem., 15: 517-521.
CrossRefDirect Link - Jakubowicz, D. and O. Froy, 2013. Biochemical and metabolic mechanisms by which dietary whey protein may combat obesity and Type 2 diabetes. J. Nutr. Biochem., 24: 1-5.
CrossRefDirect Link - Kent, K.D., W.J. Harper and J.A. Bomser, 2003. Effect of whey protein isolate on intracellular glutathione and oxidant-induced cell death in human prostate epithelial cells. Toxicol. In Vitro, 17: 27-33.
PubMedDirect Link - Khan, S.H., 2013. Whey protein hydrolysates: Techno-functional perspective. Int. J. Applied Biol. Pharmaceut. Technol., 4: 1-3.
Direct Link - Kinney, J.M. and S.P. Allison, 2003. Clues to ageing from cells, organs and outer space. Curr. Opin. Clin. Nutr. Metab. Care, 6: 3-7.
Direct Link - Kokuba, H., L. Aurelian and A.R. Neurath, 1998. 3-Hydroxyphthaloyl beta-lactoglobulin. IV Antiviral activity in the mouse model of genital herpesvirus infection. Antiviral Chem. Chemother., 9: 353-357.
Direct Link - Krissansen, G.W., 2007. Emerging health properties of whey proteins and their clinical implications. J. Am. Coll. Nutr., 26: 713S-723D.
CrossRefDirect Link - Kume, H., K. Okazaki and H. Sasaki, 2006. Hepatoprotective effects of whey protein on D-galactosamine-induced hepatitis and liver fibrosis in rats. Biosci. Biotechnol. Biochem., 70: 1281-1285.
CrossRefDirect Link - Lacroix, M., C. Gaudichon, A. Martin, C. Morens, V. Mathe, D. Tome and J.F. Huneau, 2004. A long-term high-protein diet markedly reduces adipose tissue without major side effects in Wistar male rats. Am. J. Physiol. Regul. Integr. Comp. Physiol., 287: R934-R942.
CrossRefDirect Link - Le, Y., X. Yu, L. Ruan, O. Wang and D. Qi et al., 2005. The immunopharmacological properties of transforming growth factor beta. Int. Immunopharmacol., 5: 1771-1782.
CrossRefDirect Link - Legrand, D., A. Pierce, E. Elass, M. Carpentier, C. Mariller and J. Mazurier, 2008. Lactoferrin structure and functions. Adv. Exp. Med. Biol., 606: 163-194.
CrossRefPubMedDirect Link - Luhovyy, B.L., T. Akhavan and G.H. Anderson, 2007. Whey proteins in the regulation of food intake and satiety. J. Am. Coll. Nutr., 26: 704S-712S.
Direct Link - Mader, J.S., J. Salsman, D.M. Conrad and D.W. Hoskin, 2005. Bovine lactoferricin selectively induces apoptosis in human leukemia and carcinoma cell lines. Mol. Cancer Ther., 4: 612-624.
CrossRefDirect Link - Manninen, A.H., 2009. Protein hydrolysates in sports nutrition. Nutr. Metab., Vol. 6.
CrossRefDirect Link - Marshall, K., 2004. Therapeutic applications of whey protein. Altern. Med. Rev., 9: 136-156.
PubMedDirect Link - McGregor, R.A. and S.D. Poppitt, 2013. Milk protein for improved metabolic health: A review of the evidence. Nutr. Metab., Vol. 10.
CrossRef - McIntosh, G.H., G.O. Regester, R.K. Le Leu, P.J. Royle and G.W. Smithers, 1995. Dairy proteins protect against dimethylhydrazine-induced intestinal cancers in rats J. Nutr., 125: 809-816.
PubMedDirect Link - McIntosh, G.H., P.J. Royle, R.K. Le Leu, G.O. Regester and M.A. Johnson et al., 1998. Whey proteins as functional food ingredients? Int. Dairy J., 8: 425-434.
CrossRefDirect Link - Micke, P., K.M. Beeh and R. Buhl, 2002. Effects of long-term supplementation with whey proteins on plasma glutathione levels of HIV-infected patients. Eur. J. Nutr., 41: 12-18.
Direct Link - Middleton, N., P. Jelen and G. Bell, 2004. Whole blood and mononuclear cell glutathione response to dietary whey protein supplementation in sedentary and trained male human subjects. Int. J. Food Sci. Nutr., 55: 131-141.
CrossRefDirect Link - Paddon-Jones, D., M. Sheffield-Moore, C.S. Katsanos, X.J. Zhang and R.R. Wolfe, 2006. Differential stimulation of muscle protein synthesis in elderly humans following isocaloric ingestion of amino acids or whey protein. Exp. Gerontol., 41: 215-219.
CrossRefDirect Link - Pal, S. and V. Ellis, 2010. The chronic effects of whey proteins on blood pressure, vascular function, and inflammatory markers in overweight individuals. Obesity, 18: 1354-1359.
CrossRefDirect Link - Pan, Y., A. Lee, J. Wan, M.J. Coventry, W.P. Michalski, B. Shiell and H. Roginski, 2006. Antiviral properties of milk proteins and peptides. Int. Dairy J., 16: 1252-1261.
CrossRefDirect Link - Pedersen, L., J. Mollerup, E. Hansen and A. Jungbauer, 2003. Whey proteins as a model system for chromatographic separation of proteins. J. Chromatogr. B, 790: 161-173.
CrossRefDirect Link - Pennings, B., Y. Boirie, J.M.G. Senden, A.P. Gijsen, H. Kuipers and L.J.C. van Loon, 2011. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr., 93: 997-1005.
Direct Link - Pimentel, G.D., T.O. Micheletti, F. Pace, J.C. Rosa, R.V. Santos and F.S. Lira, 2012. Gut-central nervous system axis is a target for nutritional therapies. Nutr. J., Vol. 11.
Direct Link - Rodrigues, R.M., A.J. Martins, O.L. Ramos, F.X. Malcata, J.A. Teixeira, A.A. Vicente and R.N. Pereira, 2015. Influence of moderate electric fields on gelation of whey protein isolate. Food Hydrocolloids, 43: 329-339.
CrossRefDirect Link - Ropelle, E.R., M.B. Flores, D.E. Cintra, G.Z. Rocha and J.R. Pauli et al., 2010. IL-6 and IL-10 anti-inflammatory activity links exercise to hypothalamic insulin and leptin sensitivity through IKKβ and ER stress inhibition. PLoS Biol., Vol. 8.
CrossRef - Rosaneli, C.F., A.E. Bighetti, M.A. Antonio, J.E. Carvalho and V.C. Sgarbieri, 2002. Efficacy of a Whey protein concentrate on the inhibition of stomach ulcerative Lesions caused by ethanol ingestion. J. Med. Food, 5: 221-228.
PubMedDirect Link - Schaafsma, G., 2007. Health issues of whey proteins: 3. Gut health promotion. Curr. Top. Nutraceutical Res., 5: 29-34.
Direct Link - Shankar, J.R. and G.K. Bansal, 2013. A study on health benefits of whey proteins. Int. J. Adv. Biotechnol. Res., 4: 15-19.
Direct Link - Shin, K., H. Wakabayashi, K. Yamauchi, S. Teraguchi, Y. Tamura, M. Kurokawa and K. Shiraki, 2005. Effects of orally administered bovine lactoferrin and lactoperoxidase on influenza virus infection in mice. J. Med. Microbiol., 54: 717-723.
PubMed - Solak, B.B. and N. Akin, 2012. Health benefits of whey protein: A review. J. Food Sci. Eng., 2: 129-137.
Direct Link - Sousa, G.T.D., F.S. Lira, J.C. Rosa, E.P. de Oliveira, L.M. Oyama, R.V. Santos and G.D. Pimente, 2012. Dietary whey protein lessens several risk factors for metabolic diseases: A review. Lipids Health Dis., Vol. 11.
CrossRef - Spence, L.A. and C.M. Weaver, 2003. New perspectives on dietary protein and bone health: Preface. J. Nutr., 133: 850S-851S.
Direct Link - Takakura, N., H. Wakabayashi, H. Ishibashi, S. Teraguchi, Y. Tamura, H. Yamaguchi and S. Abe, 2003. Oral lactoferrin treatment of experimental oral candidiasis in mice. Antimicrob. Agents Chemother., 47: 2619-2623.
CrossRefDirect Link - Takayanagi, T., H. Sasaki, A. Kawashima, Y. Mizuochi and H. Hirate et al., 2011. A new enteral diet, mhn-02, which contains abundant antioxidants and whey peptide, protects against carbon tetrachloride-induced hepatitis. J. Parenteral Enteral Nutr., 35: 516-522.
CrossRefDirect Link - Tang, J.E., D.R. Moore, G.W. Kujbida, M.A. Tarnopolsky and S.M. Phillips, 2009. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol., 107: 987-992.
CrossRefDirect Link - Tome, D., J. Schwarz, N. Darcel and G. Fromentin, 2009. Protein, amino acids, vagus nerve signaling and the brain. Am. J. Clin. Nutr., 90: 838S-843S.
Direct Link - Tsuda, H., K. Sekine, K.I. Fujita and M. Iigo, 2002. Cancer prevention by bovine lactoferrin and underlying mechanisms a review of experimental and clinical studies. Biochem. Cell Biol., 80: 131-136.
CrossRefDirect Link - Uchida, T., T. Oda, K. Sato and H. Kawakami, 2006. Availability of lactoferrin as a natural solubilizer of iron for food products. Int. Dairy J., 16: 95-101.
CrossRefDirect Link - Wakabayashi, H., M. Takase and M. Tomita, 2003. Lactoferricin derived from milk protein lactoferrin. Curr. Pharm. Des., 9: 1277-1287.
PubMedDirect Link - Walzem, R.L., C.J. Dillard and J.B. German, 2002. Whey components: Millennia of evolution create functionalities for mammalian nutrition: What we know and what we may be overlooking. Crit. Rev. Food Sci. Nutr., 42: 353-375.
CrossRefDirect Link - Ward, P.P., E. Paz and O.M. Conneely, 2005. Multifunctional roles of lactoferrin: A critical overview. Cell. Mol. Life Sci., 62: 2540-2548.
PubMedDirect Link - Wolber, F.M., A.M. Broomfield, L. Fray, M.L. Cross and D. Dey, 2005. Supplemental dietary whey protein concentrate reduces rotavirus-induced disease symptoms in suckling mice. J. Nutr., 135: 1470-1474.
Direct Link - Wu, G., Y.Z. Fang, S. Yang, J.R. Lupton and N.D. Turner, 2004. Glutathione metabolism and its implications for health. J. Nutr., 134: 489-492.
PubMedDirect Link - Xu, R., N. Liu, X. Xu and B. Kong, 2011. Antioxidative effects of whey protein on peroxide-induced cytotoxicity. J. Dairy Sci., 94: 3739-3746.
CrossRefDirect Link - Yalcin, A.S., 2006. Emerging therapeutic potential of whey proteins and peptides. Curr. Pharm. Des., 12: 1637-1643.
CrossRefDirect Link - Yamauchi, R., E. Wada, D. Yamada, M. Yoshikawa and K. Wada, 2006. Effect of β-lactotensin on acute stress and fear memory. Peptides, 27: 3176-3182.
CrossRefDirect Link - Zagorska, J. and I. Ciprovica, 2012. The influence of heat treatment on antimicrobial proteins in milk. World Acad. Sci. Eng. Technol., 64: 1127-1131.
Direct Link - Zemel, M.B., H. Shi, B. Greer, D. DiRienzo and P.C. Zemel, 2000. Regulation of adiposity by dietary calcium. Federat. Am. Soc. Exp. Biol. J., 14: 1132-1138.
Direct Link - Zimecki, M. and J. Artym, 2005. [Therapeutic properties of proteins and peptides from colostrum and milk]. Postepy Hig. Med. Dosw., 59: 309-323, (In Polish).
PubMed - Svensson, M., A.K. Mossberg, J. Pettersson, S. Linse and C. Svanborg, 2003. Lipids as cofactors in protein folding: Stereo-specific lipid-protein interactions are required to form HAMLET (human α‐lactalbumin made lethal to tumor cells). Protein Sci., 12: 2805-2814.
Direct Link








