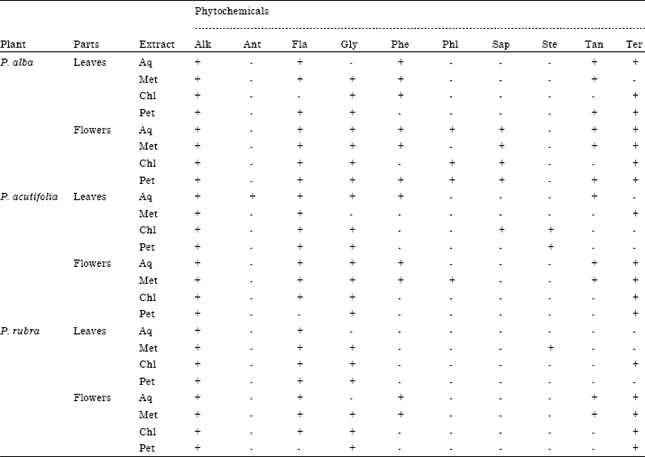
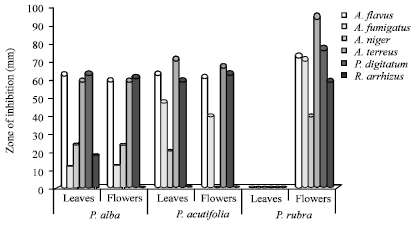
Research Article
Comparative Studies of Plumeria Species for their Phytochemical and Antifungal Properties Against Citrus sinensis Pathogens
R and D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru-560072, Karnataka, India
Saurabh Awasthi
R and D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru-560072, Karnataka, India
K. Dhananjaya
R and D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru-560072, Karnataka, India
H. Mallesha
R and D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru-560072, Karnataka, India
K.R. Ravikumar
R and D Centre, Robust Materials Technology Pvt. Ltd., Bengaluru-560072, Karnataka, India