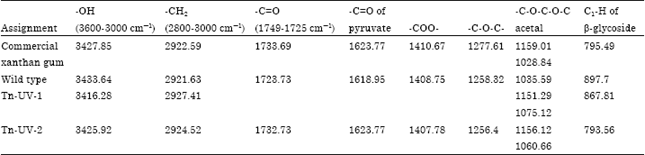
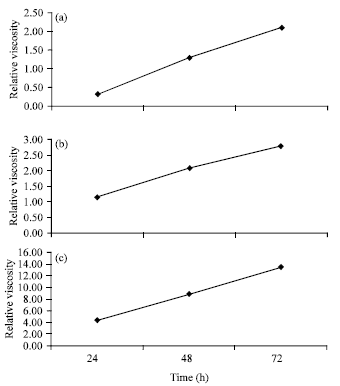
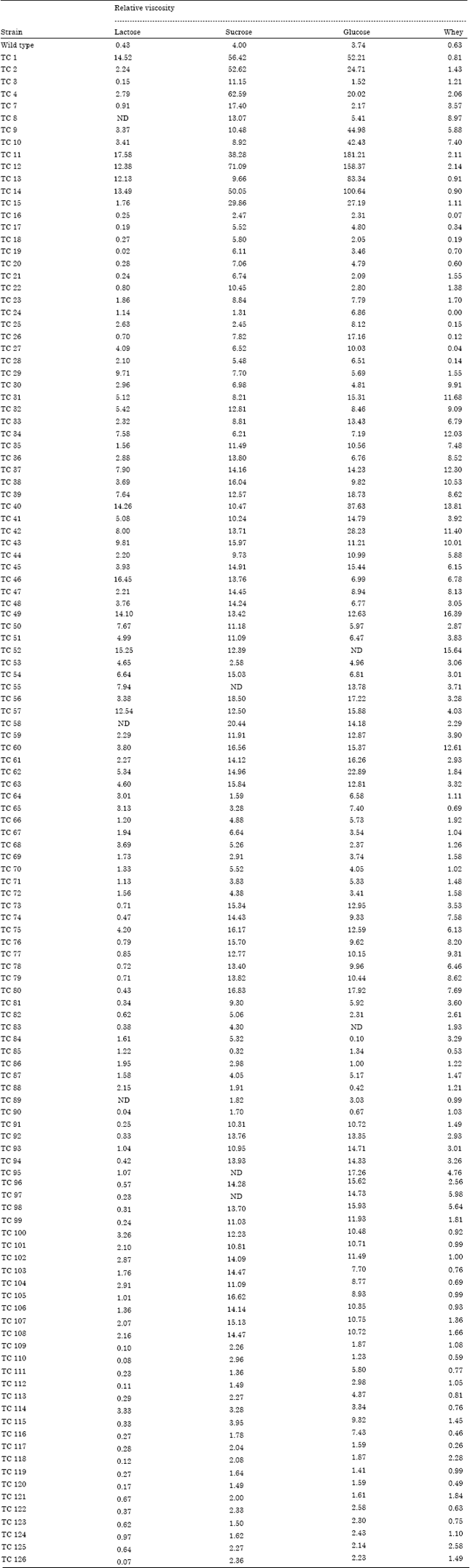
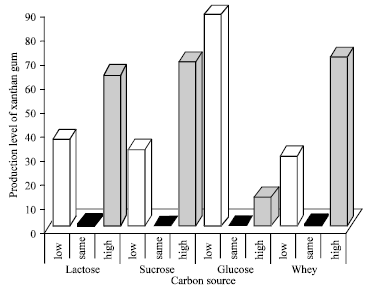
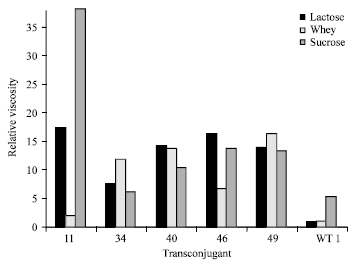
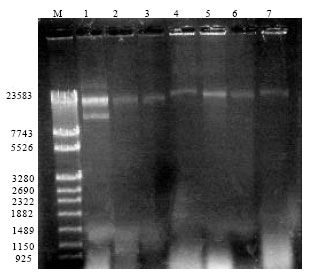
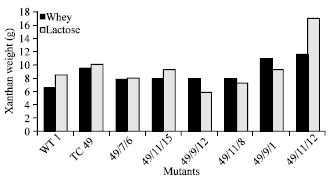
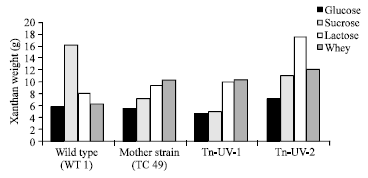
Research Article
Genetically Modified Strains of Xanthomonas campestris Higher Xanthan Producer and Capable to Utilize Whey
Department of Genetics and Cytology, Genetic Engineering and Biotechnology Division, National Research Center, Cairo, Egypt
W.S. Elsayed
Department of Microbiology, Faculty of Sciences, Ain Shams University, Cairo, Egypt
U.M. Badr
Department of Genetics and Cytology, Genetic Engineering and Biotechnology Division, National Research Center, Cairo, Egypt
H.M. Gebreel
Department of Microbiology, Faculty of Sciences, Ain Shams University, Cairo, Egypt
K.M.A. Khalil
Department of Genetics and Cytology, Genetic Engineering and Biotechnology Division, National Research Center, Cairo, Egypt