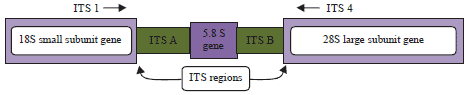
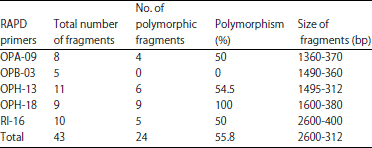
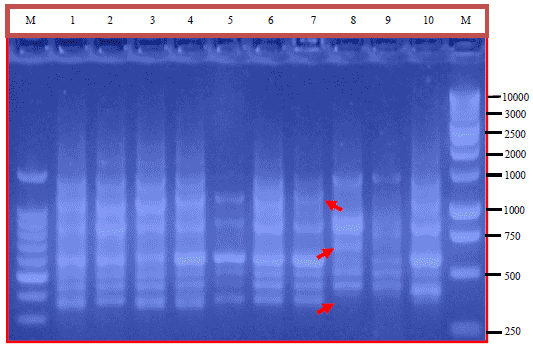
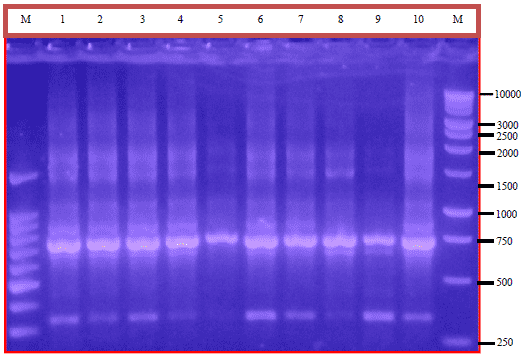
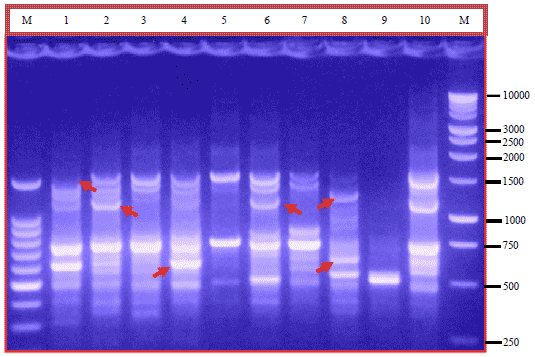
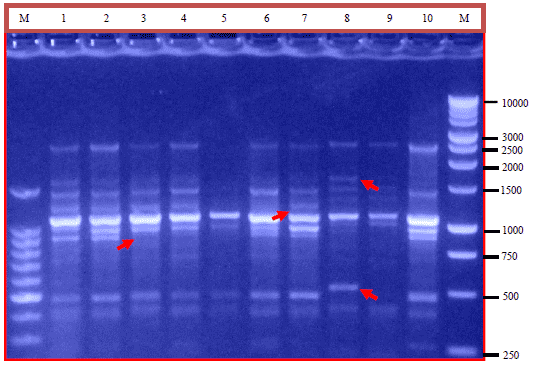
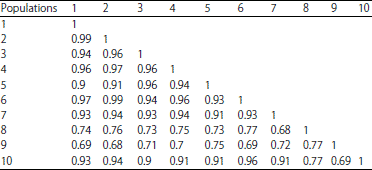
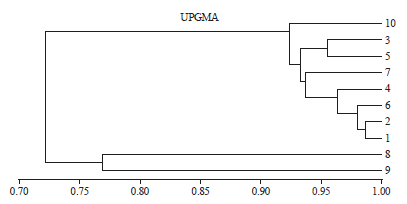
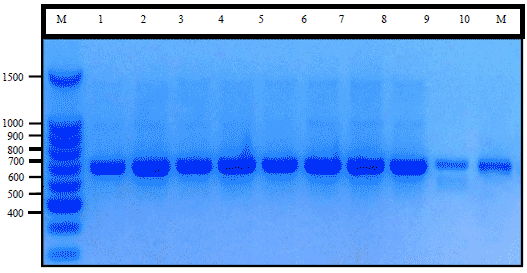
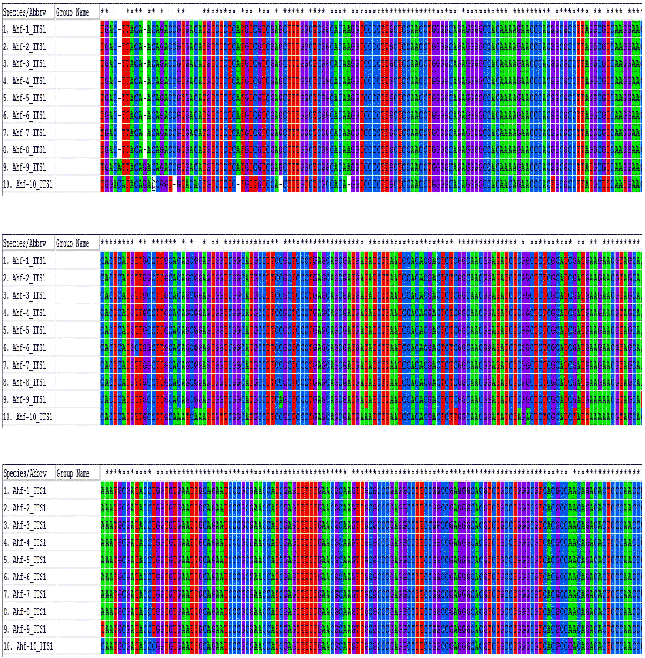
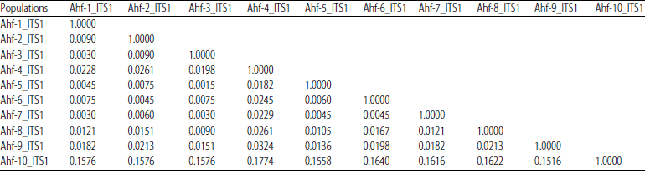
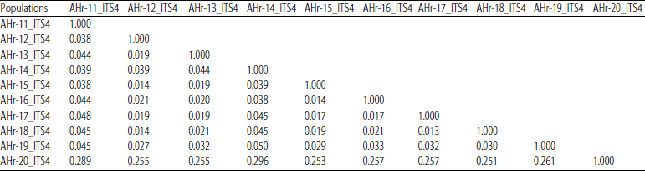
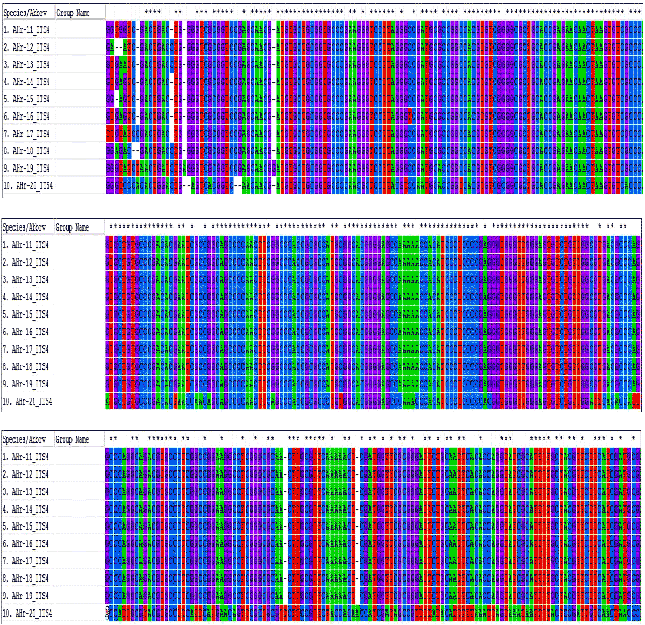
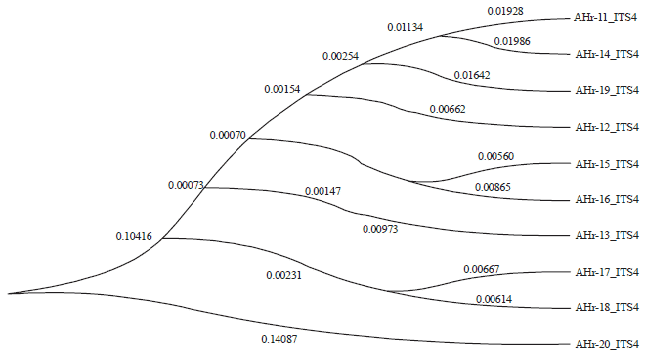
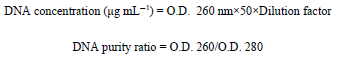Research Article
Molecular Detection of Johnsongrass (Sorghum halepense L.) Evolution in Iraq
Department of Field Crops Sciences, College of Agriculture, University of Anbar, Anbar, Iraq
LiveDNA: 964.24069
Ahmed Talib Saleh
Department of Field Crops Sciences, College of Agriculture, University of Anbar, Anbar, Iraq