ABSTRACT
Five media supplemented with different concentrations of NAA and 2,4-D growth regulators and two different mature embryo sources were tested in order to obtain the best wheat callus formation. One of these mature embryo sources is whole seed with moved embryo and the other was embryo set free from seeds. The highest callus formation rate was observed in embryo set free cultured on MS supplemented with 4 mg l-1 2,4-D and 1 mg l-1 NAA. There was no callus formation on basal medium MS. In overall, percentage of embryogenic callus formation in free embryo procedure was higher than in endosperm supported embryo procedure. In embryogenic wheat callus formation, 2,4-D seemed an effective growth regulator as all 2,4-D supplemented media showed higher callus formation than alone NAA. On the other hand, NAA (1 mg l-1) in the 2,4-D supplemented MS medium enhanced wheat callus induction.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.17.19
URL: https://scialert.net/abstract/?doi=ajps.2004.17.19
INTRODUCTION
One of the important steps in the application of biotechnology for crop improvement is the successful plant regeneration from cells, organs or tissues. Many investigations on tissue culture and plant regeneration in a variety of plants including wheat have been well documented. Plant regeneration from wheat tissue culture has mainly been obtained from callus derived from immature embryos[1-4]. Callus induction from immature wheat embryos are commonly used for genetic transformation or tissue culture[5,6]. Thus, first of all, for biotechnological research on wheat requires reliable callus induction and then plant regeneration. Especially, for an efficient genetic transformation, establishment of a successful wheat tissue culture system using immature or mature embryos is essential.
Immature embryos in wheat have been reported as a good source of callus induction[7]. However, immature embryo production is time-dependent procedure and difficult to obtain all seasons of the year. Varshney et al.[8] stated that the maturity stage of explants is also important for embryogenic callus formation. Therefore, this problem led the researchers to develop mature embryo derived callus. Although mature embryos from dried seeds are available at all times throughout the year the problem is low callus induction frequency. In order to overcome this problem, Ozgen et al.[9] successfully used endosperm supported callus induction method from mature embryo cultures.
The aim of this study was to obtain an efficient and alternative callus induction from free mature embryos to endosperm supported embryos.
MATERIALS AND METHODS
Winter wheat T. aestivum L. cv. (Sana) was used as source of mature embryos. The seeds were kept in 70% (v/v) ethanol for 5 min for surface sterilisation and then washed for several times with sterile distilled water. The seeds were treated with commercial bleach plus one drop of Twin-80 as a wetting agent for 20 min and again washed with 2-3 times with sterile distilled water. The sterile seeds were imbibed in sterile water for 2 h at 33°C.
After sterilisation procedure and imbibing the seeds, two different explants were used. One is whole seed with moved (not set free) embryo with a scalpel under sterile condition and the other was embryo set free from seeds, which was described as free embryo. The embryos were then transferred onto callus induction media containing different growth regulators.
The media were prepared by using standard MS[10] as a basal medium. MS was supplemented with sucrose (30 g l-1) and Difco-bacto agar (8 g l-1) and adjusted to pH 5.8. Growth regulators were added to media prior to autoclaving. After autoclaving the media, 20 ml of sterile molten medium was poured into 90x15 mm sterile Pyrex Petri dishes. The Petri dishes were then sealed with Nescofilm (Bado Chemical Ind. Ltd., Japan). Five different callus induction media based on the basal MS medium and vitamins were prepared as follows:
| 1. | MS (control) |
| 2. | MS+8 mg l-1 2,4-D |
| 3. | MS+2 mg l-1 NAA |
| 4. | MS+4 mg l-1 2,4-D+1 mg NAA |
| 5. | MS+4 mg l-1 2,4-D+2 mg NAA |
The seeds with moved embryo and free embryos were placed furrow and cutting edge downwards in Petri dishes, respectively. The Petri dishes were kept in total darkness at 25±1°C for three weeks and then number of explants with callus were recorded.
Each treatment replication comprised six Petri dishes and each dish consisted of ten explants. The experimental design was a randomized block design. Least Significant Difference (LSD) test was applied for means separation at 0.05 significance level. The statistical analysis was carried out by the SAS computer package (SAS Institute Inc. 1998, Carry, North Carolina, USA).
RESULTS AND DISCUSSION
Five media supplemented with different concentrations of NAA and 2,4-D growth regulators and two different mature embryo sources were tested in order to obtain the best wheat callus formation. Visible callus formation was obtained within a few days and observation was taken at 12th day of the culture. Variance analysis of the results showed that there were significant differences between treatments (Table 1). The highest callus formation rate was observed in free embryo cultured on MS supplemented with 2,4-D (4 mg l-1) and NAA (1 mg l-1). There was no callus formation on basal medium MS. All embryos on MS produced shoots and roots. In overall, percentage of embryogenic callus formation in free embryo procedure was higher than in endosperm supported embryo procedure. In embryogenic wheat callus formation, 2,4-D seemed an effective growth regulator as all 2,4-D supplemented media showed higher callus formation than alone NAA, which is in line with previous reports[7,9,11]. On the other hand, NAA (1 mg l-1) in the 2,4-D supplemented MS medium enhanced wheat callus induction.
In previous studies on endosperm supported embryo culture, it was stated that 2,4-D solution pre-treatment could be necessary for efficient callus formation[12]. However, in our case, the seeds were not treated with 2,4-D solutions prior to callus culture.
| Table 1: | The effect of medium on induction of callus derived from endosperm supported and free embryos |
 | |
| * | Means with the same letter are not significantly different at 0.05 significance level |
| LSD0.05 = 13.41 | |
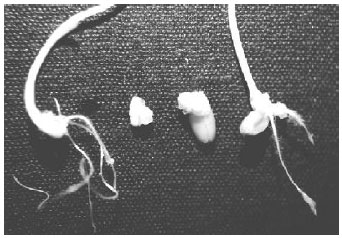 | |
| Fig. 1: | Wheat callus formation. From left to right: 1. Shoot and root growth from free embryo, 2. Callus formation from free embryo, 3. Callus formation from endosperm supported embryo and 4. shoot and root growth from endosperm supported embryo |
The results showed that embryo could be cultured without endosperm supporting and any pre-treatment as also can be seen in Fig. 1. Therefore using free embryo for callus formation is practically easy and less space requires in a culture vessel. In addition, it is easier to identify embrogenic callus that is derived from embryo free from endosperm rather than embryo with endosperm tissue. As a conclusion, the development callus induction protocol for wheat from endosperm free mature embryos is helpful for crop improvement of wheat through the modern plant tissue culture as well as genetic transformation.
REFERENCES
- Sears, R.G. and E.L. Deckard, 1982. Tissue culture variability in wheat: Callus induction and plant regeneration. Crop Sci., 22: 546-550.
CrossRefDirect Link - Wang, A.S., 1987. Callus induction and plant regeneration from maize mature embryos. Plant Cell Rep., 6: 360-362.
CrossRefDirect Link - Ozgen, M., M. Turet, S. Ozcan and C. Sancak, 1996. Callus induction and plant regeneration from immature and mature embryos of winter durum wheat genotypes. Plant Breed., 115: 455-458.
CrossRefDirect Link - Simmonds, J.A. and J.L. Grainger, 1993. The toxicity of antibiotics to protoplast cultures of Triticum aestivum L. Plant Sci., 89: 209-214.
CrossRefDirect Link - McCormac, A.C., H.X. Wu, M.Z. Bao, Y.B. Wang, R.J. Xu and D.F. Chen, 1998. The use of visual marker genes as cell-specific reporters of agrobacterium-mediated T-DNA delivery to wheat (Triticum aestivum L.) and barley (Hordeum vulgare L.). Euphytica, 99: 17-25.
CrossRefDirect Link - Redway, F.A., V. Vasil, D. Lu and I.K. Vasil, 1990. Identification of callus types for long-term maintenance and regeneration from commercial cultivars of wheat (Triticum aestivum L.). Theoret. Applied Genet., 79: 609-617.
CrossRefDirect Link - Varshney, A., T. Kant, V.K. Sharma, A. Rao and S.L. Kothari, 1996. High frequency plant regeneration from immature embryo cultures of Triticum aestivum and T. durum. Cereal Res. Commun., 24: 409-416.
Direct Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Sayar, T.M., A.M. Birsin, H. Ulukan and M. Ozgen, 1999. Effect of seed size on the tissue culture response of callus from mature embryos of wheat species. Wheat Inform. Serv., 89: 1-6.
Direct Link








