ABSTRACT
Lake Naivasha and its environs experienced increased levels of pesticide application due to the rapid expansion in floriculture farming. Previous studies had shown detectable levels of Organochlorine pesticide residues in water samples. The Lake is under threat since such occurrences are linked to the poor water quality. Moreover, Organochlorine pesticide residues persist in the environment while, the Organophosphate pesticides which had been adopted as an alternative to Organochlorine pesticides were highly toxic. The study investigated the occurrence and effects of Organochlorine and Organophosphate pesticide residues on water quality. The specific objectives of the study were: To determine water physico-chemical parameters and investigate pesticide residues concentration of Organophosphate and Organochlorine. Longitudinal study design was used during the months of February to July, 2011. The sampling sites were selected on the basis of their relevance as point sources of pesticide contamination. Three replicates of water samples were collected monthly, 18 water samples per site, totaling to 90 samples. These samples were subjected to water quality analysis and Gas Liquid Chromatography technique. The data generated were subjected to analysis while applying Statistical Package of Social Science using one way ANOVA at p<0.05. The results revealed that the water samples tested were in compliance with World Health Organisation and Kenya Bureau of Standards recommended guidelines while, Organochlorine and Organophosphate pesticide residues were not detected. It was concluded that the lack of these pesticide residues was due the current conservation measures preventing water pollution in the Lake. The study further, recommended continuous monitoring and conservation measure to be maintained.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpp.2013.105.113
URL: https://scialert.net/abstract/?doi=ajpp.2013.105.113
INTRODUCTION
Lake Naivasha and its environs has experienced rapid expansion in flower farming activities since early 1980’s and currently occupying 4,000 ha (Becht et al., 2005). This sector is linked to intensive irrigation and pesticide use. These pesticides have significant effect since over 98% of sprayed insecticides and 95% of herbicides reach destination other than the target namely: air, water, bottom sediments and food (Miller and Miller, 1998).
The increased level of pesticides application and their inappropriate use are widely documented (Appelgren, 1994). Although these pesticides are hazardous farmers still prefer to use them due to their effectiveness in pest and disease control and their application to a wide variety of crops (Wandiga, 1995). Their uses are restricted namely: Aldrin and Dieldrin for termite control in building industry, Organochlorine and Organophosphate pesticides is deemed inevitable despite their ban or restriction (Getenga et al., 2000). Farmers will not fully comply to abandon them so long as they were still available in the market for the simple reason that production will fall (IPCS, 2005). Lindane for seed dressing and ban on Dichlorodiphenyl-trichloroethene but, much has not changed. These pesticides are still in common use in the Lake’s vicinity (Gitahi et al., 2002). For instance, Lindane, Aldrin and Dieldrin are used for seed dressing while Dichlorodiphenyl-trichloroethene in public health especially for fight against Malaria (Thumbi et al., 2011) which explains their occurrence in the Lake. Currently, the situation in Lake Naivasha is delicate and needs urgent attention.
Furthermore, poor cultivation methods have made it easier for alluvial and loamy soils found in the Lake’s surrounding (Arusei et al., 2002) to be carried by erosion to the Lake. Some flower farms had extended their boundaries right down to the water bodies (Arusei et al., 2002). Further, accelerating drifting of pesticides due to the soils’ fine texture, high water retaining ability and high levels organic content (Becht et al., 2005). This makes it easier for Organochlorine and Organophosphate pesticide residues to be carried into the Lake without any treatment through surface run off. There is increased sediments and chemical residue reaching the Lake from flower farms. Farms far away are not spared as their waste water eventually ended up into the Lake since once these pesticides had been applied in the fields they were transported to the Lake by surface runoff, rivers and streams (Wandiga, 2001; Wandiga et al., 2002; Getenga et al., 2004). In addition, wind and rain also carried pesticides away from their point source, causing contamination of surface waters (Bouman et al., 2002; Shomar et al., 2005).
The broad objective of this study was to assess the effect of pesticide residues from the flower farms on water quality within the shores of Lake Naivasha, Kenya. Monitoring programs as suggested by Mavuti and Harper (2005) enabled us to determine with confidence the principal point source of Organochlorine and Organophosphate pesticide residues. Their study recommended continuous analysis to generate useful information that would be used to put in measures to maintain the level of pesticides to be below the maximum acceptable level in drinking water according to the standards set by WHO (1984a), CCME (1991) and Government of Kenya (2006).
MATERIALS AND METHOD
Research design: This was a longitudinal study involving laboratory investigations. The variables were namely: Water physico-chemical parameters and pesticide residues analysis. The first set of water samples were tested for physico-chemical parameters namely (1) Temperature, (2) Conductivity, (3) pH, (4) Turbidity, (5) Dissolved oxygen, (6) Chemical oxygen demand, (7) Biochemical oxygen demand and (8) Total suspended solids. The second set of water samples were investigated for pesticide residues analysis targeting five Organochlorine pesticides namely: (a) Endosulfan, (b) Lindane, (c) Aldrin, (d) Dieldrin and (e) Dichlorodiphenyl-trichloroethene and Organophophate pesticides namely (a) Nemacur, (b) Orthene, (c) Durban, (d) Diazol and (e) Fenitrothion.
The water samples were collected between February and July, 2011 at 10.00 a.m.-3.00 p.m. The duration of the study allowed for season variation to be investigated for it covered both dry and wet season. All the sampling sites were accessed using a motorboat together with research assistants to aid in water sample collection. Flexibility was paramount necessitating the use of three laboratories simultaneously considering that some variables were more likely to be affected by storage prior to analysis. For this reason determination of temperature was carried out in situ while water samples for Biochemical oxygen demand, Organochlorine and Organophophate were put under low temperature conditions until analysis to prevent deterioration and retard growth of micro-organisms.
Sample size determination: Three replicate of surface water samples were collected monthly, 18 water samples per site, totalling 90. There was no mathematical formula applied to calculate sample size due to the discrete nature of water. However, the sample size was sufficiently reasonable without compromising on the outcome yet making economic sense due to high cost involved in the chemicals analysis.
Water sample collection procedure: Sample bottles were rinsed out two or three times with the water to be sampled before collection. The three replicates of water samples with a capacity of 5 L were collected at each site sum total of 75 L of water per field visit and attached with appropriate labels. This volume was enough to take care of special determinations which required slightly larger quantities of water.
Data collection instruments: The instruments used for physico-chemical parameter were: (1) Conductivity metre, (2) Thermometer, (3) Photoelectric turbidimeter, (4) Glass cells with stopper, (5) pH meter, (6) Vacuum flask and pump, (7) Rubber adapter, (8) Filter, (9) Incubator and (10) Oxitop with magnetic stirrer, (11) Digestion vessels, (12) Pipette and (13) Burette guided by scientific procedures (Thomaz et al., 1992). Gas Liquid Chromatography set up had the following accessories: Syringe, analytical column, gases, suitable detector and a printer. Initially, before running any sample, it was confirmed that all parts of the equipment in contact with the samples and the reagents are interference free through analysis of method blank as a continuing check and safe guard against chronic laboratory contamination. The procedure was carried out at the Central Veterinary Laboratory, Kabete. A varian model equipped with ECD/NPD operating under the following conditions: Oven temperature 170°C for 1.5 min, ramp rate 15° min-1, oven No. II 170°C for 0.5, ramp rate@ 25° min-1 to hold for 1 min, column 3% OV 17, injector temperature 350°C ECD/NPD detector type and detector saturation below 1 (specific 0.4) in water bath.
Training of observers: The research assistants went through orientation programme at the initiation of the study to ensure that all the prepared water samples were registered in the laboratory with their details labelled.
Measurement of water physico-chemical parameters: These included water Temperature, Conductivity, Turbidity, pH, Total Suspended Solids, Dissolved Oxygen, Chemical Oxygen Demand and Biochemical Oxygen Demand using standard laboratory procedures (Thomaz et al., 1992; AOAC, 1990).
Organochlorine (OC) and organophosphate (OP) analysis: The pesticide residue analysis focussed on Organochlorine and Organophophate pesticide while targeting five molecules from each of these two groups of pesticides. The water samples were subjected to Gas Liquid Chromatography due to its accuracy in detecting low quantities of these analytes. The sampling was done using standard methods as described by Akerblom (1995). The water samples were analysed without prior filtering so that the residues which had been adsorbed on the surface of suspended particles, especially the organic particles like humus (Ramulu, 1985) could be detected. Unfiltered water samples (100 mL), previously preserved with 10% NaCl were extracted by Liquid-Liquid Extraction (LLE) method (Akerblom, 1995). The analytes were extracted using the mixed solvent (hexane/diethylether) in the ratio 85: 15% as described in the reference (Greenberg et al., 1992) and shaken vigorously using a mechanised shaker for one hour and subsequently supernatant layers extracted. The extracted supernatants appeared clean and thus were not subjected to further clean up. The supernatants were concentrated to 2 mL by using rotary evaporator with water bath (Sieber and Notling, 1982).
Further precautions were taken to ensure that there was no residual water left in the preparation. Once ready, 2 μL aliquots of both reference standards and the extracts were injected into the column and the comparison was made between the standards and the samples based on the retention times and the total area under the peak on the chromatograms.
Data analysis: The data generated by the study were recorded in a Microsoft Excel spread sheets and subjected to Statistical Package of Social Science (SPSS) using one way ANOVA at p<0.05 level of significance for probability. The descriptive statistics criterion was applied to summarized and evaluate significant findings.
RESULTS
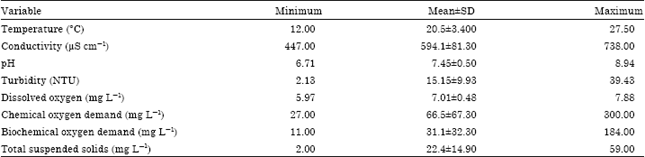
Water physico-chemical parameters: The results of the overall surface water physico-chemical parameters showing the mean and range values of variables measured in Lake Naivasha during a 6 month study (February-July, 2011) are shown in Table 1.
Water quality standards were used to evaluate Lake’s water aspects in regards to its compliance to surface water suitability. The results (Table 1) indicated that water physico-chemical parameters were within the limits in compliance with World Health Organisation and Kenya Bureau of Standards as shown in Table 2.
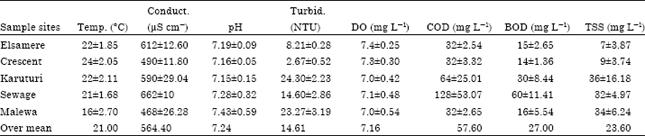
Seasonal effect on physico-chemical parameters: The results of the surface water physico-chemical parameters showing the mean and SD values of variables measured in Lake Naivasha during the dry season (February to April, 2011) are shown in Table 3.
| Table 1: | Physico-chemical parameters in Lake Naivasha from February-July, 2011 |
 | |
| Table 2: | World health organisation and Kenya bureau of standards |
 | |
| Table 3: | Water physico-chemical parameters (Mean±SD) across sites during dry season (n = 45) |
 | |
| Table 4: | Water physicochemical parameters (Mean±SD) across sites during wet season |
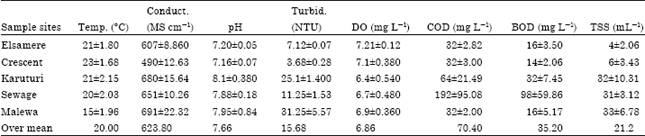 | |
Comparison was made between values of variables measured in Lake Naivasha during dry season (Table 3) and wet season (Table 4) to find out the range of difference between the seasons.
The results of the surface water physico-chemical parameters showing the mean and SD values of variables measured in Lake Naivasha during the wet season (May to July, 2011) are shown in Table 4.
It was imperative to conclude that the Lake’s overall water quality was not affected by seasonal variations. Since, results (Table 3 and 4) indicated that there were no significant changes in the water physico-chemical parameters between two seasons.
DISCUSSION
Physico-chemical parameter: The result revealed that water physico-chemical variables were significantly different between the sampling sites. The results revealed high range of water quality aspects within the shores off River Malewa and Karuturi Flower especially, high levels of BOD, COD and Total Suspended Solids which were attributed to the organic compounds carried along with the surface run off relative to other sampling sites. These were attributed to the changes in the land use and soil erosion resulting from human activities contributed to siltation leading to increased water turbidity. The results also revealed that a higher turbidity level at the Elsamere Conservation Centre which was due to wind and human perturbation.
Although, according to World Health Organization (WHO, 1984b, 1986, 1989, 2000) and Kenya Bureau of Standards (KEBS) recommended guidelines for surface water (Table 2). The comparisons of the results revealed that the mean values of water physico-chemical variables were within the recommended levels and were relatively stable despite the seasonal variations (Table 3 and 4) experienced during the study duration.
River Malewa inflow had the highest conductivity. This was attributed to clearance of the original forest, particularly the papyrus swamp in the northern delta of the inflow rivers (North Swamp) (Gaudet and Muthuri, 1981) which served as a filter retaining silt/sediment from upstream during rainy periods. Consequently, the inflow of rivers at the northern end of the Lake may contain a large amount of sediment and conductivity may change very rapidly in a few hours (Kitaka et al., 2002). Similarly, Kitaka et al. (2002) reported that northern shore of the Lake especially at the entry point of River Malewa had experienced intensive cultivation.
The results revealed high Dissolved Oxygen levels which was attributed to the Lake’s high altitude (1884 masl) (Harper et al., 2003). The Lake’s high altitude above sea level may have promoted the dissolution of oxygen in water but, this was limited to about 11 mg L-1 as this was a function of pressure (Bootsma and Hecky, 1993). The study noted that the control sites recorded significantly high levels of Dissolved Oxygen in comparison to the test sites an indication of their inflows carrying significant level of organic and inorganic matter.
In overall, the results revealed that turbidity, conductivity, pH and Dissolved Oxygen were significantly different across the sampling months. Mean conductivity ranged from 564.4 μS cm-1 (Table 3 ) to 623.8 μS cm-1 (Table 4) with seasonal variations. Harper et al. (1990) and Harper and Mavuti (2004) earlier reported a range of 233-480 μS cm-1 in their study of the Lake. This change could have been caused by surface run off with sediments, the drainage from irrigation, chemical residues and the Lake’s water level fluctuations which may have had a strong influence on water quality and its ecological biodiversity (Harper et al., 1990; Harped et al., 1995).
The result revealed moderate and stable pH values 6.71-8.94 (Table 1). This is in agreement with (Harper et al., 1990) who also observed a pH value 7.7-8.5. However, it was noted that the values of pH were slightly affected with seasonality. Furthermore, the wet season was accompanied with strong water currents and increased air movement (Walton, 2010) thus the significance monthly variations in the levels of Dissolved oxygen. Moreover, the decrease in the levels of Dissolve Oxygen was attributed to the decomposition of macrophytes that had inhabited the shores of the Lake during the wet season leading to high algae growth which had consumed large volume of oxygen (Wetzel, 1975).
In overall results revealed that there was no significant difference in temperature, chemical oxygen demand, biochemical oxygen demand and total suspended solids. The low temperatures of the Lake in the morning were attributed to heat losses from the Lake by specific conduction to the air (Verschuren et al., 2000) which was enhanced by evaporation. Therefore, to avoid high disparities involving temperature, the procedure was carried out at mid day in situ using thermometer.
Status of organochlorine (OC) and organophosphate (OP) pesticide residues: The targeted analytes of organochlorine and organophophate pesticides residue had experienced drastic decline in their use following the ban of production and usage in most developed countries (USGS, 2005). Therefore, these analytes were either banned or restricted for instance DDT banned in 1992 while Lindane, Aldrin and Dieldrin had been restricted (Thumbi et al., 2011). Despite the restrictions on Lindane to seed dressing only it is relatively biodegradable and less persistent (Tanabe et al., 1993) while Aldrin and Dieldrin are restricted to termite control in the building industry (PCPB, 2010, 1998).
Organophophate have short half-lives and relatively biodegradable (Lalah and Wandiga, 1996; Gitahi et al., 2002; Gitahi, 1999). Furthermore, the low deposition rate of Organophosphate was due to their high solubility in water and potential hydrolysis degradation (Wang et al., 2012). Simlarly, Lalah and Wandiga (1996) noted that water aided the photolytic process of Organophophate.
CONCLUSION AND RECOMMENDATION
The study noted relative stability in water physico-chemical parameters while analytes of Organochlorine and Organophosphate pesticide residues to be below detection limit. These were reflections of the various intervention measures employed by the government and stakeholders. The implementation of these approaches was good for the conservation of the Lake and its environs. All the indications were pointing that the Lake was on its pathway to restoration to its state of not being contaminated especially following the ban and restrictions. Therefore, the targeted analytes were not of any threat to the Lake’s water.
REFERENCES
- AOAC., 1990. Official Methods of Analysis. 15th Edn., Association of Official Analytical Chemists, Washington, DC., USA., Pages: 684.
Direct Link - Bouman, B.A.M., A.R. Castaneda and S.I. Bhuiyan, 2002. Nitrate and pesticide contamination of groundwater under rice-based cropping systems: Past and current evidence from the Philippines agriculture. Ecosyst. Environ., 92: 185-199.
CrossRef - Bootsma, H.A. and R.E. Hecky, 1993. Conservation of the African Great Lakes: A limnological perspective. Conserv. Biol., 7: 644-656.
CrossRef - Getenga, Z.M., J.I.O. Jondiko, S.O. Wandiga and E. Beck, 2000. Dissipation behavior of malathion and dimethoate residues from the soil and their uptake by garden pea (Pisum sativum). Bull. Environ. Contam. Toxicol., 64: 359-397.
CrossRef - Getenga, Z.M., F.O. Keng'ara and S.O. Wandiga, 2004. Determination of organochlorine pesticide residues in soil and water from river Nyando drainage system within Lake Victoria Basin, Kenya. Bull. Environ. Contam. Toxicol., 72: 335-343.
CrossRef - Gitahi, S.M., D.M. Harper, S.M. Muchiri, M.P. Tole and R.N. Ng'ang'a, 2002. Organochlorine and organophosphorus pesticide concentrations in water, sediment and selected organisms in Lake Naivasha (Kenya). Hydrobiologia, 488: 123-128.
CrossRef - Harper, D.M., K.M. Mavuti and S.M. Muchiria, 1990. Ecology and management of Lake Naivasha, Kenya, in relation to climatic change, alien species' introductions and agricultural development. Environ. Conserv., 17: 328 -336.
CrossRef - Harped, D.M., C. Adams and K. Mavuti, 1995. The aquatic plant communities of the Lake Naivasha wetland, Kenya: Pattern, dynamics and conservation. Wetlands Ecol. Manage., 3: 111-123.
CrossRef - Harper, D.M. and K.M. Mavuti, 2004. Lake Naivasha, Kenya: Ecohydrology to guide the management of a tropical protected area. Ecohydrol. Hydrobiol., 4: 287-305.
Direct Link - IPCS, 2005. Inventory of IPCS and other WHO pesticide evaluations and summary of toxicological evaluations and summary of toxicological evaluations performed by joint meeting of pesticide residue (JMPR) through 2002. The International Programme on Chemical Safety, World Health Organisation, Geneva, Switzerland.
- Kitaka, N., D.M. Harper and K.M. Mavuti, 2002. Phosphorus inputs to Lake Naivasha, Kenya, from its catchment and the trophic state of the lake. Hydrobiologia, 488: 73-80.
CrossRef - Lalah, J.O. and S.O. Wandiga, 1996. The persistence and fate of malathion residues in stored beans (Phaseolus vulgaris) and maize (Zea mays). Pestic. Sci., 46: 215-220.
Direct Link - Thomaz, S.M., F.A. Lansac Toha, M.C. Roberto, F.A. Esteves, A.F. Lima, 1992. Seasonal variation of some limnological factors of Lagoa do Guarana, a varzea lake of the Rio Parana State of Mato Groso do Sul, Brazil. Rev. Hydrobiol. Trop., 25: 269-276.
Direct Link - Tanabe, S., A. Subramanian, A. Ramesh, P.L. Kumaran, N. Miyazaki and R. Tatsukawa, 1993. Persistent organochlorine residues in dolphins from the bay of Bengal, South India. Mar. Pollut. Bull., 26: 311-316.
CrossRefDirect Link - Verschuren, D., K.R. Laird and B.F. Cumming, 2000. Rainfall and drought in equatorial East Africa during the past 1,100 years. Nature, 403: 410-414.
Direct Link - Wandiga, S.O., 2001. Use and distribution of organochlorine pesticides. The future in Africa. Pure Applied Chem., 73: 1147-1155.
CrossRef - Wandiga, S.O., P.O. Yugi, M.W. Barasa, I.O. Jumba and J.O. Lalah, 2002. The distribution of organochlorine pesticides in marine samples along the Indian Ocean coast of Kenya. Environ. Technol., 23: 1235-1246.
CrossRef








