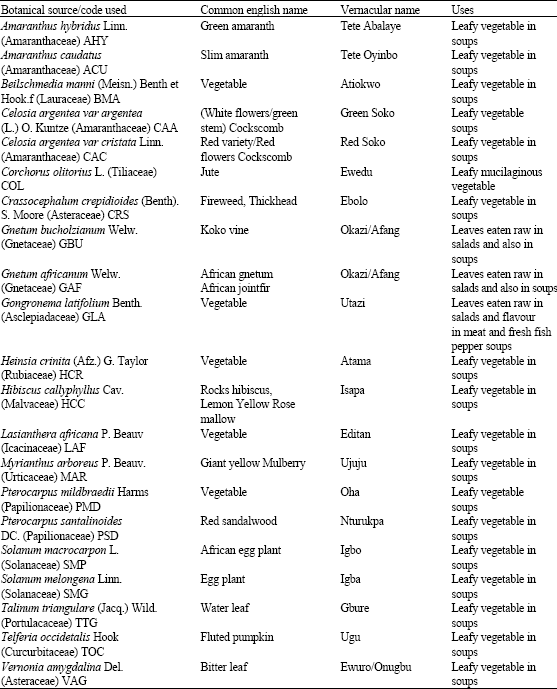
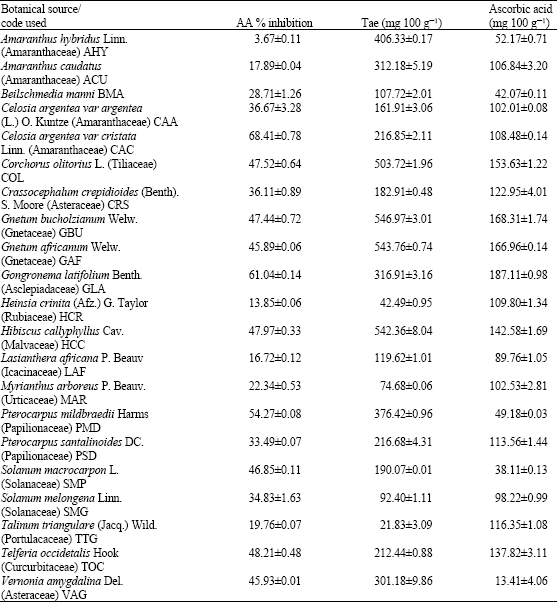
Research Article
Antioxidant Activity of Selected Nigerian Green Leafy Vegetables
Department of Pharmacognosy, Faculty of Pharmacy, University of Lagos, Nigeria
S.I. Inya-Agha
Department of Pharmacognosy, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, Nigeria
F.I. Segun
Department of Pharmacognosy, Faculty of Pharmacy, University of Lagos, Nigeria
M.O. Sofidiya
Department of Pharmacognosy, Faculty of Pharmacy, University of Lagos, Nigeria
O.O. Ilori
Department of Pharmacognosy, Faculty of Pharmacy, University of Lagos, Nigeria