Research Article
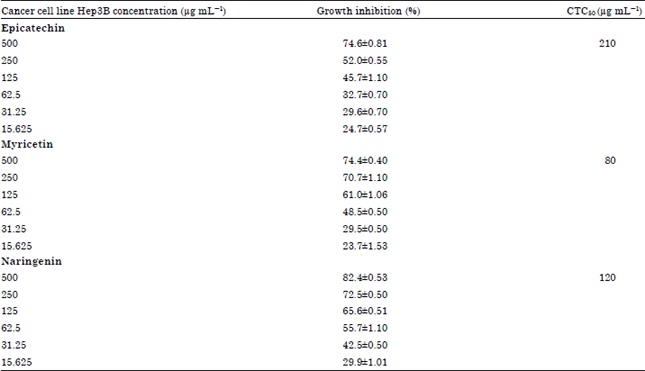
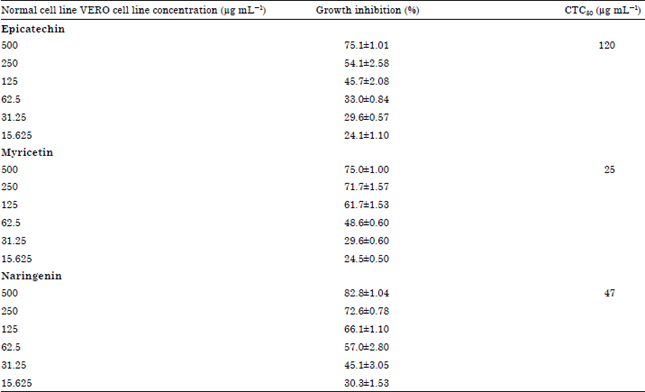
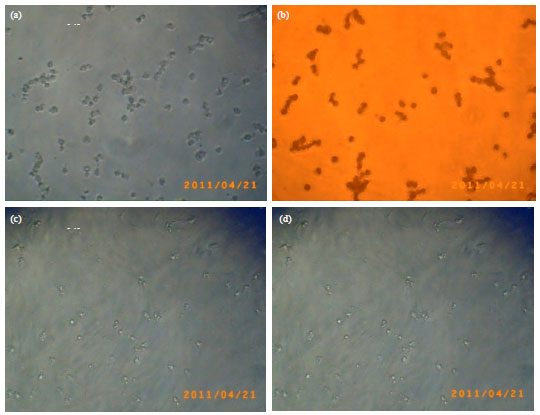
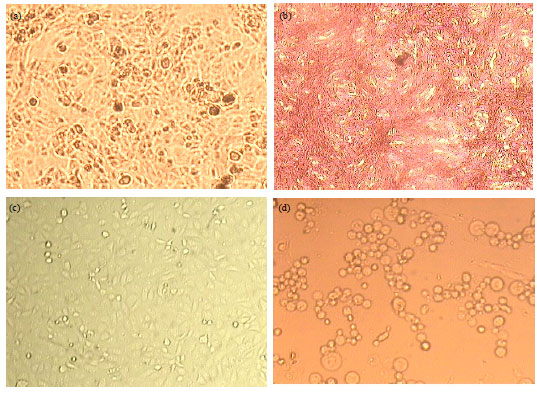
In vitro Analysis of Phytochemicals Epicatechin, Naringenin, Myricetin Against Hep3B Cell Lines and VERO Cell Lines
Department of Biochemistry and Bioinformatics, Karpagam University, Coimbatore, Tamil Nadu, India
V.K. Gopalakrishnan
Department of Biochemistry and Bioinformatics, Karpagam University, Coimbatore, Tamil Nadu, India