Research Article
Effects of Chlorophyll on Body Functioning and Blood Glucose Levels
Department of Nutrition and Food Science , College of Home Economics, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
Nora Mohammed ALkehayez
Department of Nutrition and Food Science , College of Home Economics, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
Amal Hassan Alshawi
Department of Nutrition and Food Science , College of Home Economics, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
Nora Abdullah Al-Faris
Department of Nutrition and Food Science , College of Home Economics, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
LiveDNA: 966.16382









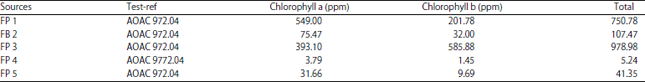
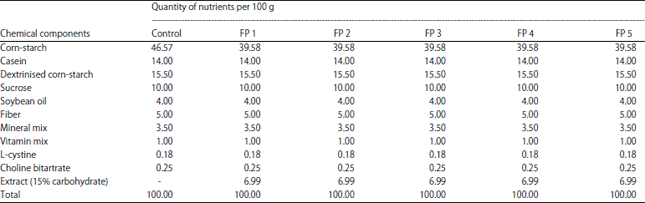
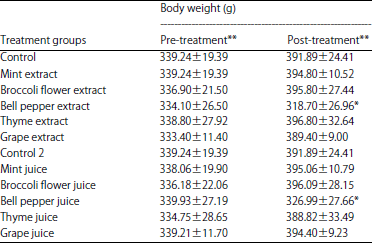
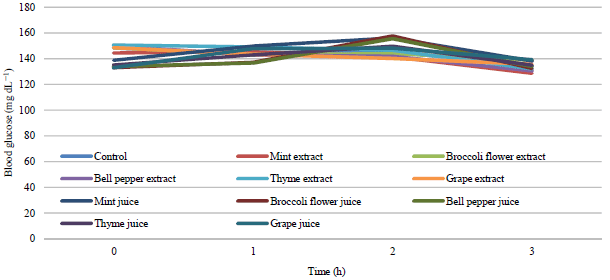
Salah Omar Reply
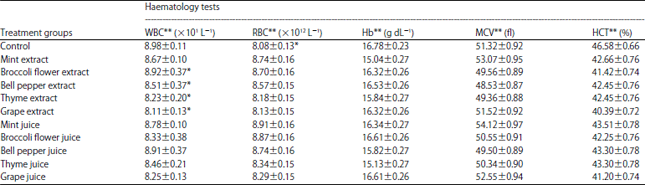
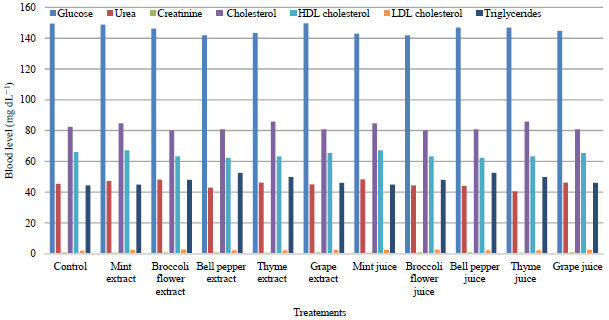
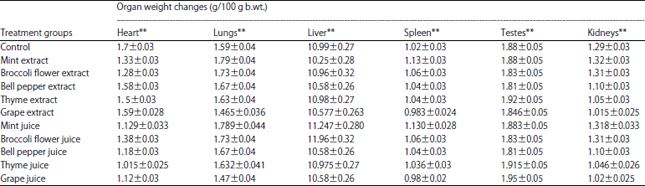
What is the significance of the slight decrease in white blood cells, haematocrit, and haemoglobin and the increase in red blood cells after the intake of chlorophyll in this study?