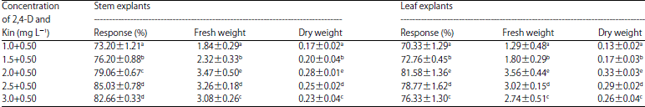
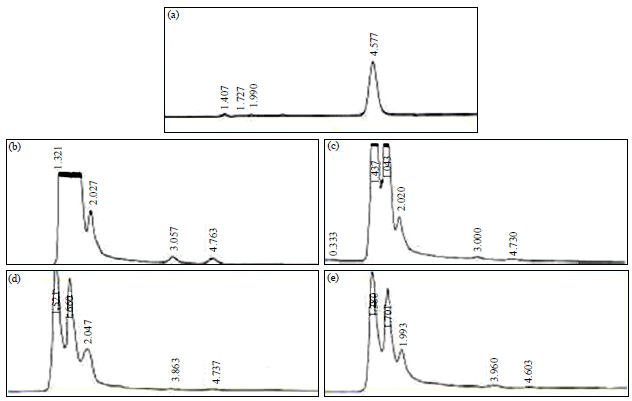
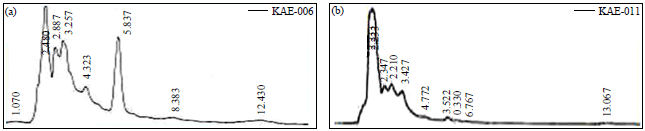
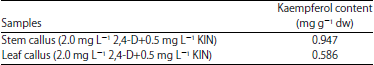Research Article
Effect of Yeast Elicitor on the Enhancement of Kaempferol from in vivo and in vitro Callus Cultures of Dregea volubilis Benth
Department of Biotechnology, Mohamed Sathak College of Arts and Science, Sholinganallur, Chennai, India
LiveDNA: 91.2037
R. Bhakyaraj
Department of Biosciences, SNMV College of Arts and Science, Coimbatore, India
LiveDNA: 91.4419
M. Syed Ali
Department of Biotechnology, Mohamed Sathak College of Arts and Science, Sholinganallur, Chennai, India
LiveDNA: 91.17314
R. Muthezhilan
Department of Marine Biotechnology, AMET University, Kanathur, Chennai, Tamil Nadu, India
LiveDNA: 91.1730