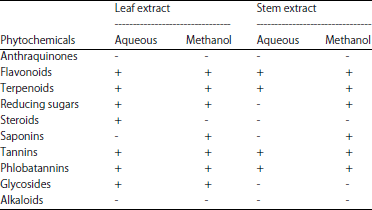
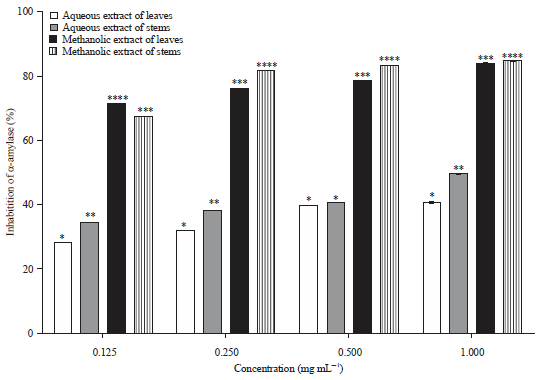
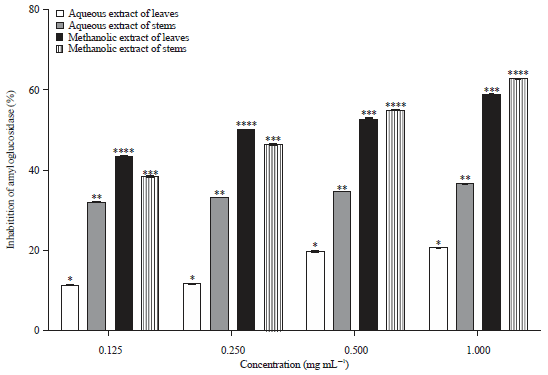
Research Article
Phytochemical Analysis and in vitro Evaluation of Antidiabetic Activity of Diospyros buxifolia
Bio Industrial Technology Programme, Faculty of Agro-based Industry, Universiti Malaysia Kelantan, 17600 Campus Jeli, Malaysia
LiveDNA: 91.3622
Elawarasi Krishinasamy
Bio Industrial Technology Programme, Faculty of Agro-based Industry, Universiti Malaysia Kelantan, 17600 Campus Jeli, Malaysia
Indireddy Rama Manohar Reddy
Department of Biotechnology, Sri Venkateswara University, 517502 Tirupati, India
Malepati Dhananjaya Naidu
Department of Zoology, Yogi Vemana University, 516003 Kadapa, Andhra Pradesh, India
Siew Hua Gan
Human Genome Centre, School of Medical Sciences, Universiti Sains Malaysia, 16150 Kelantan, Kubang Kerian, Malaysia