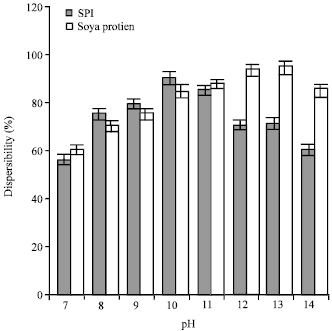
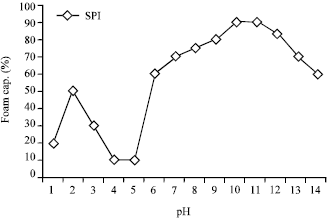
Research Article
Sesame Protein 11: Functional Properties of Sesame (Sesamum indicum L.) Protein Isolate as Influenced by pH, Temperature, Time and Ratio of Flour to Water During its Production
School of Food Science and Technology, Southern Yangtze University, No.l800, Lihu Road, Wuxi 214122, Jiangsu, People’s Republic of China
Zhu Kerui
School of Food Science and Technology, Southern Yangtze University, No.l800, Lihu Road, Wuxi 214122, Jiangsu, People’s Republic of China
Zhou Hui Ming
School of Food Science and Technology, Southern Yangtze University, No.l800, Lihu Road, Wuxi 214122, Jiangsu, People’s Republic of China
Qian Haifeng
School of Food Science and Technology, Southern Yangtze University, No.l800, Lihu Road, Wuxi 214122, Jiangsu, People’s Republic of China
Jestina Baby Kanu
Milton Margai College of Education and Technology, University of Sierra Leone, Goderich Campus, Freetown, Sierra Leone
Zhu Kexue
School of Food Science and Technology, Southern Yangtze University, No.l800, Lihu Road, Wuxi 214122, Jiangsu, People’s Republic of China