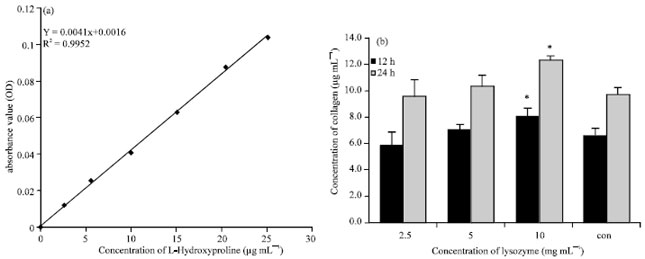
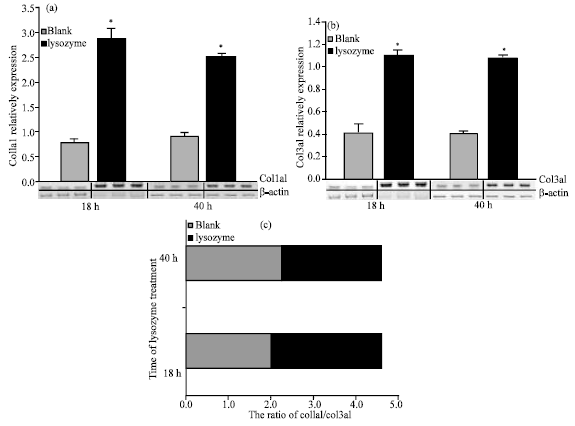
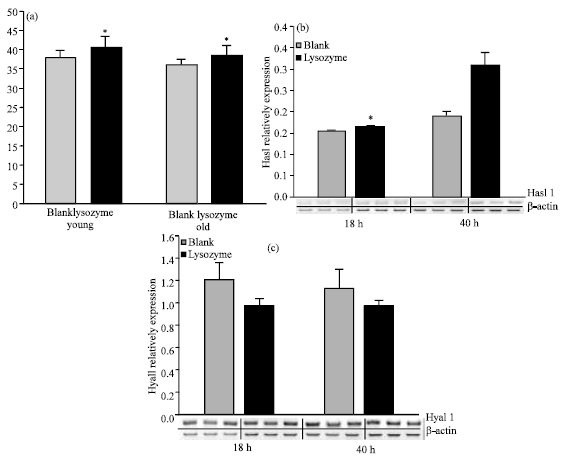
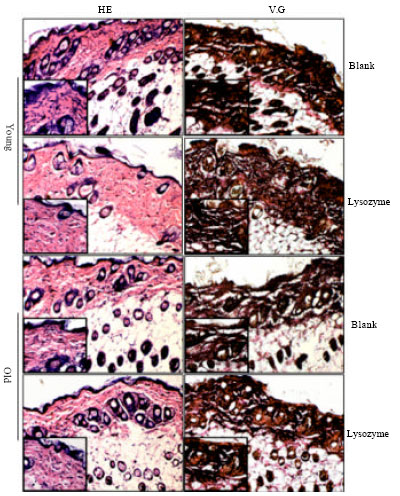
Research Article
Egg White Lysozyme Promoted Collagen Secreting of Dermal Fibroblasts in Mice
College of Animal Science and Technology, China Agricultural University, Beijing 100193, People Republic of China
X. Li
College of Animal Science and Technology, China Agricultural University, Beijing 100193, People Republic of China
K. Yu
College of Animal Science and Technology, China Agricultural University, Beijing 100193, People Republic of China
W.Z. Ma
College of Animal Science and Technology, China Agricultural University, Beijing 100193, People Republic of China
Z.C. Cao
College of Animal Science and Technology, China Agricultural University, Beijing 100193, People Republic of China
G.F. An
Zhong Ke Yi Nuo Biological Technology Co., Ltd, Beijing, P.R. China
Z.X. Lian
State Key Laboratory of Agrobiotechnolog, China Agricultural University Beijing 100193, People Republic of China