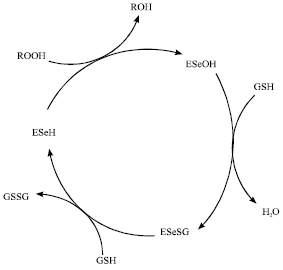
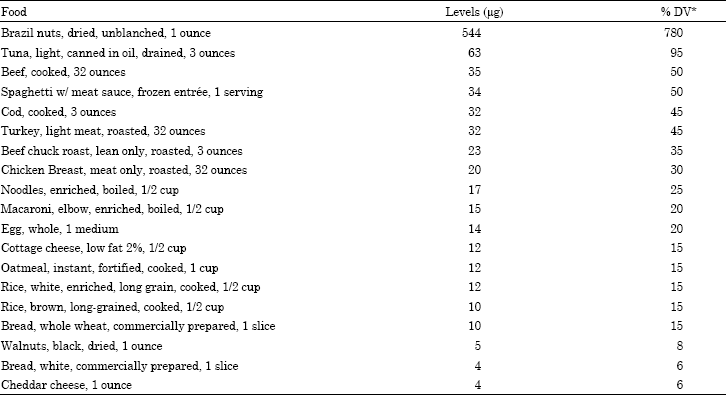
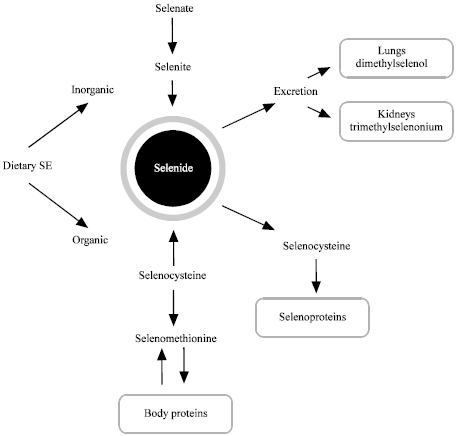
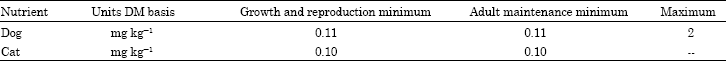Research Article
Role of Selenium in Pets Health and Nutrition: A Review
Provimi India Innovation Centre, Bangalore, India
B. Purushotham
Provimi India Innovation Centre, Bangalore, India
P.M. Radhakrishna
Provimi India Innovation Centre, Bangalore, India
P.M. Abhilekha
Provimi India Innovation Centre, Bangalore, India
H.M. Vagdevi
Sahyadri Science College (Autonomous), Shimoga, Karnataka, India