Research Article
Microbial Polycyclic Aromatic Hydrocarbons Degradation in Soil
College of Agriculture and Veterinary Medicine, Qassim University,51452 Buraidah, P.O. Box 6622, Saudi Arabia
Polycyclic Aromatic Hydrocarbons (PAHs), a group of staple chemicals, are wide spread pollutants in the environment, such as soil and sediments (Jones, 1991; Wild et al., 1994). They have attracted increasing public and academic concern because some of them are known or suspected of having mutagenic, carcinogenic and tetratogenic effects on animals (Perera, 1997). This has led to intensive research in their biological and chemical properties in the environment and mechanisms by which these physiological effects are produced.
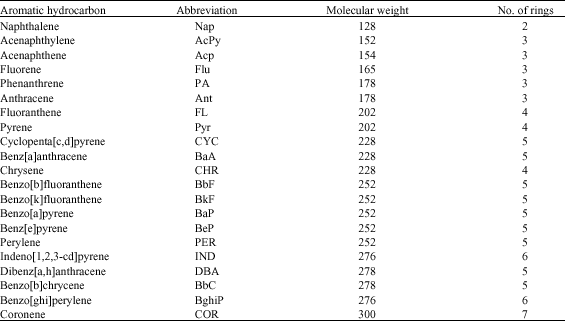
The major sources of PAHs contamination in the environment are from coking of coal, distillation of wood, operation of gas works and oil refineries, runoff from asphalt pavements, vehicular emission, petroleum spills and incomplete combustion of fossil fuels and organic matter (Al-Turki and Dick, 2003; Tang et al., 2005). PAHs of interest range from benzo(a) pyrene and benzo (ghi) perylene with 5 and 6 rings, respectively. Table 1 shows representative PAHs.
Fluoranthene and pyrene are the main representative PAHs because they are predominant in air, sediment and water and were often exist together in contaminated environment (Tang et al., 2005). These two 4-ring compounds are therefore frequently chosen as model compounds for ex situ biodegradation studies of high molecular weight PAHs. They have the same molecular weight and octanol-water partition coefficient, but the solubility of fluoranthene (0.26 mg L-1) was nearly double of that of pyrene (0.14 mg L-1).
Since, PAHs are habitual soil contaminants their presence in soil represents a public health hazard. The clean- up of soil can be accomplished using different remediation technologies.
General PAHs Fate in the Soil
Once PAHs are found in the soil, they are subjected to a number of natural weathering processes. Included in these processes is photooxidation. This process is of limited value because once the source of PAHs spilled in the soil, it usually seeps into the soil away from sunlight (Cofield et al., 2007). Another process that may take place is dissolution. This process is not significant because much of PAHs have only minimal solubility in water. Another mechanism that may determine the fate of PAHs is the adsorption of PAHs to the soil. PAHs are characterized by their palpable hydrophobic nature. Consequently, these species tend to be adsorbed on soil particulates, especially on the organic fraction of solids (Al-Turki et al., 2004). This binding can make the PAHs unavailable to biological systems hence there is reduction in toxicity but inhibition in biodegradation. The reason for this inhibition is that they are partially unavailable for microbial degradation since bacteria are known to degrade chemicals only when they are dissolved in water (Sticher et al.,1997; Al-Turki and Dick, 2003).
| Table 1: | Abbreviations, molecular weights and number of rings of major pollutant PAHs |
 | |
Intimately related to the adsorption process is sequestration phenomenon. The sequestrations are an aging stage in which hydrophobic adsorbed substances show a declining availability to both remediation and chemical extraction. The occurrence of sequestration is of significant importance since the toxicity values of sequestered molecules are, obviously, quite different from those assigned to dissolved substances or weakly adsorbed molecules (Brion and Pelletier, 2005).
Microorganisms Catalyzing Hydrocarbon Biodegradation
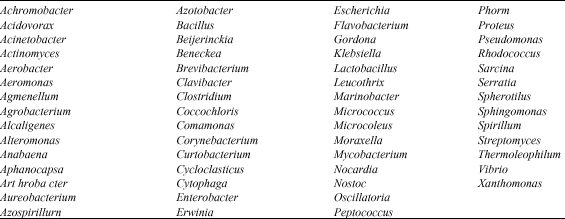
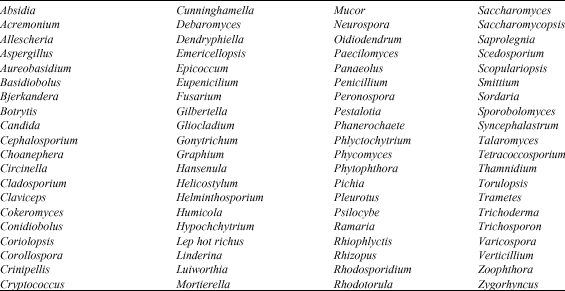
Biodegradation of hydrocarbon can be executed by various microorganisms (Whyte et al., 1996; El-Hadidi and Al-Turki, 2007). As shown in Table 2 and 3 more than 60 genera of bacteria and 80 genera of fungi contain species capable of degrading hydrocarbons.
These microorganisms can degrade hydrocarbons in a broad range of habitats and under both aerobic and anaerobic condition (Al-Turki, 2007). Some of these species are capable of degrading aliphatic hydrocarbons, others aromatic molecules but few are able to degrade both classes of molecules (Whyte et al., 1996). However, the complete degradation of PAHs may require a community of organisms that sequentially exchange and then transform excreted metabolites as the molecules is gradually broken down (Kelley et al., 1993).
Several micro algae species were found to degrade PAHs (Kirso and Irha, 1998; Lei et al., 2003). Species such Chlorella vulgaris, Scenedesmus quadricauda, Scenedesmus platydiscus and Selenanatrum capricornutum were found to remove fluoranthene and pyrene at different degrees (Lei et al., 2007).
| Table 2: | Bacterial genera that contain PAHs-degrading species |
 | |
| Table 3: | Fungal genera that contain PAHs-degrading species |
 | |
Microorganisms capable of degrading hydrocarbons anaerobically are less authenticated and most of the information available concerns the degradation of small aromatic molecules such as benzene, toluene and xylene (Colberg and Young, 1985). Anaerobic degradation of PAHs is currently becoming of interest to many researchers.
Most of the microorganisms in soil are located in the thin layers of water surrounding soil particles and biodegradation can be slower in exposed drier soils on hillocks than in sheltered moist area. Degradation is slower in cold climates than in more temperate or tropical zones and the degradation rates generally decrease with depth through soil profile (Al-Turki and Dick, 2003) Degradation of PAHs in the presence of oxygen is well documented by laboratory experiments.
Microorganisms cultivated through growth on a medium containing PAHs are typically used in laboratory experiments. However, naturally occurring microorganisms are normally isolated from PAHs-contaminated soil and ground water.
In optimum laboratory conditions, half-life values of less than three days for two-rings naphthalene have been seen in the soil/water mixture in aerobic conditions. Half-life values for multi-ringed PAHs, such as benzo(a)pyrene are 30-300 days. In anaerobic conditions, degradation is generally slower. In degradation experiments with soil from sites contaminated by PAHS, field trials have revealed half-life values of 6-16 years compared with 0.5-5 years in laboratory scale trials.
Biochemical Mechanisms for the Biodegradation of PAHs in Soil
The biological degradation of PAHs, as outlined by Johnsen et al. (2005), can serve three different functions: (a) assimilative biodegradation that yields carbon and energy for the microorganism and goes along with the mineralization of the compound, (b) intercellular mechanism that render the PAHs water-soluble and hence excreted and (c) cometabolism, which is the degradation of PAHs without production of energy and carbon for the organism metabolism (Boonchan et al., 2000).
Cometabolism is defined as a non-specific enzymatic reaction, with a substrate competing with the structurally similar primary substrate for the enzymes` active site. Cometabolism (co oxidation) may be defined also as a term used to describe the process in which a microorganism oxidizes or dehalogenate a substrate without being able to utilize the energy derived from these reactions to support growth.
The initial step in PAHs biodegradation is that PAHs must be taken by the microorganism. In aerobic bacteria the initial reaction is the incorporation of both atoms of an oxygen molecule. Aerobic activation of PAHs as outlined by Sutherland et al. (1995) is as follows: (a) activation by dioxygenases to produce cis-dihydrodiols by bacteria and green algae, (b) activation by methane monooxygenases to produce phenols by methanotrophic bacteria, (c) activation by cytochrome P450 monooxygenases to produce arene oxides by many fungi and few bacteria and (d) activation by lignin-degrading enzymes to quinones by lignolytic fungi.
Activation by Dioxygenases to Produce CIS-Dihydrodiols
In this reaction, both oxygen atoms are inserted in the substrate. Usually, naphthalene oxidation is taken as an example of this process because it has been well studied (Ensley et al., 1982; Torok et al., 1995). Although, the insertion of oxygen is thermodynamically favored, there is requirement for a reductant, in the form of NADH. There should be strong selection pressure on the microorganism to prevent the partially oxidized molecule from escaping the cell and this may be the reason for not detecting metabolic intermediates of PAHs biodegradation in the environment.
Sutherland et al. (1995) outlined the initial pathway for naphthalene oxidation in Pseudomonas and the most likely C-C bonds to be activated are equivalent to that between C1and C2 in naphthalene. The oxidation in this case is catalyzed by naphthalene dioxygenases(Ensley et al., 1982). It should be mentioned in this respect that naphthalene dioxygenases and toluene dioxygenases have been found to oxidize a wide range of substrate analogs to active products (Boyd et al., 1993). Many of these compounds have been used to synthesize a number of compounds of interest to the pharmaceutical and chemical industries. It has also been found that bacteria capable of degrading the larger PAHs compounds are often able to degrade the smaller ones and the reverse is not generally true (Kiyohara et al., 1992). It is clear that PAHs degradation in soil is most effective when there is a broad diversity of degrading microorganisms.
Activation by Methane Monooxygenases to Produce Phenols
The initial enzyme of methane oxidation, methane monooxygenase, has very broad substrate specificity. Methane monooxygenase has been shown to convert naphthalene to 1 and 2- naphtha`s (Stirling and Dalton, 1981) but the importance of this reaction in the environment is not seen.
Activation by Cytochrome P450 Monooxygenase to Produce Arene Oxides
The enzyme responsible for conversion to arene oxide is cytochrome P450. PAHs are oxidized to epoxide and then epoxide is converted non-enzymatically to phenols which can be conjugated (Cerniglia and Heitkamp, 1990). Alternatively, the epoxide is converted to trans-dihydrodiol by epoxide hydrolases.
The produced oxides are hydrolyzed to trans-dihydrodiol or to phenols which are subsequently conjugated with sulphate, glucuronic acid or glucose. The fungal cytochromeP450 transformation of PAHs may mobilize PAHs for further degradation by bacteria.
The ubiquitous co-existence of bacteria and fungi in the soil and their known catabolic co-operation suggest that physical interactions between them may be of importance for PAHs degradation.
Activation by Lignin-Degrading Enzymes to Produce Quinones
Lignolytic fungi oxidize lignin extracellularly by the action of lignin peroxidases, Mn-dependant peroxidases and laccases. These enzymes are non-specific and oxidize a wide variety of organic compounds. The lack of selectivity is reasonable, considering the random structure of lignin (Hammel et al., 1992). The peroxidases require the presence of peroxides (e.g., hydrogen peroxide) for activity. Electrons are removed from lignin aromatic rings to give cationic radicals, which are stabilized by cleavage of C-C bonds in lignin skeleton. The radicals then react with oxygen or water to give hydroxyl or keto derivatives.
The products of peroxidase-catalysed PAHs oxidation are PAHs-quinones (Hammel et al., 1992). Laccases use molecular oxygen to oxidize phenolic compounds to very reactive, free radicals. The presence of primary, mediating substrates extends the substrate range of laccases to non-phenolic aromatics by forming potent radicals which co-oxidize non-phenolic lignin compounds and also PAHs (Pickard et al., 1999). PAHs oxidized by fungi range in size from naphthalene to benz(a)pyrene. Variety of white rot fungal genera are capable of oxidizing these PAHs (de Jong et al.,1992).
The initial attack on high molecular weight PAHs in soil by fungal exo-enzymes appear to be more likely than attack by bacterial intracellular enzymes. The advantages of fungal oxidation is that fungal exoenzymes can diffuse to the immobile PAHs while bacterial PAHs-diooxygenases are generally cell bound because they require NADH as cofactor. Oxidation products of PAHs are more soluble than the parent compounds and hence more available to the microbial community.
One way to increase the extent of PAHs bioremediation in soil is to mix PAHs contaminated soil with organic matter containing mycelia of white rot fungi (Lestan and Lamar, 1996).
Anaerobic Biodegradation of PAHs
Recent investigations have demonstrated that several classes of PAHs were degraded in the absence of oxygen with nitrate, ferrous irons or sulphate as an electron acceptor or under conditions of methanogenesis (Widdle and Rabus, 2001). The sulphate-reducing bacterium, strain Hxd3, which is capable of oxidizing alkanes to CO2 under sulphate-reducing conditions, has been isolated from soil. This strain was closely related to the genus Desulfococcus (Aeckersberg et al., 1991). It was found that strain Hxd3 anaerobically transformed an alkane to fatty acid through sub-terminal carboxylation at C3 position of the alkane followed by elimination of the two adjacent terminal carbon atoms (So et al., 2003).
The anaerobic degradation of toluene and naphthalene has been reviewed (Widdle and Rabus, 2001). In contrast to the degradation of simple aromatic hydrocarbons (monoaromatic compounds) little information exists on the anaerobic degradation of PAHs. However, anaerobic PAHs biodegradation has been demonstrated only in many enrichment cultures, including those derived from marine sediments. Little is known about the initial activation reactions of these PAHs. This is another area of intense current research activity.
Fate of the Carbon of PAHs During Biodegradation
After reviewing the initial reactions involved in the activation of PAHs, a question regarding the fate of the hydrocarbons should be answered. Various studies have demonstrated that eventual fate is mineralization to CO2 and H2O but how complete is this oxidation? Using pure cultures of PAH-degrading bacteria and a variety of PAHs as a sole source of carbon and energy, Bouchez et al. (1999) found that: (a) between 56 and 77% of the carbon was converted to CO2 and (b) bout 16 to 35% of the carbon was converted to microbial biomass and mall but significant 5 to 23% of the carbon accumulated as metabolites.
In contaminated soil there should be other microorganisms capable of degrading intermediate metabolites. Partially oxidized metabolites may become bound to organic and inorganic material in the soil matrix.
Bioremediation
Bioremediation, the microbial decontamination of PAHs contaminated soil, is claimed to be efficient, economic and versatile alternative to physiochemical treatment (Atlas, 1992).
The rate of microbial degradation of PAHs in the soil is affected by several physiochemical and biological parameters including the number and species of microorganisms present, the conditions for microbial degradation activity (presence of nutrients, oxygen, pH and temperature). Aerobic biodegradation is far more widespread and far more rapid. Other limiting factors for microbial degradation of PAHs in the soil include the quantity, quality and bioavailability of the contaminants, soil characteristics (e.g., particle size distribution). PAHs degrading microorganisms have been found in all soils searched. PAHs can serve as the sole source of carbon and energy for the microorganisms. It should be noted that high levels of PAHs can inhibit all microbial growth. In this connection it should be mentioned that fertilizers that provide N, P, Fe and other trace elements are essential for bioremediation if the soil is deficient in these nutrients. Under certain circumstances, PAHs can become tightly adsorbed to the soil and become less available for the microbial degradation (Postma et al., 1990). Under such circumstances, a bacterial strategy to overcome such situations is to release a biosurfactant. Biosurfactants are small detergent-like molecules with a hydrophilic head and lipophilic tail. They form spherical or lamellar micelles when the surfactant concentration exceeds a compound-specific, critical micelles concentration. Hydrophobic compounds become solubilized in the hydrophobic cores of the micelles that lead to transfer of PAHs from solid to liquid (Volkering et al., 1997). Moreover, biosurfactants seem to influence the bacterial uptake of solubilized compounds (Beal and Betts, 2000).
It can be concluded that the potential of biodegradative microorganisms in the development of bioremediation applications e.g. for biomineralization or organic pollutants has not yet been fully realized. The role of chemo taxis in biodegradation is becoming more apparent and might be a factor in choosing strains for bioaugmentation purposes. It is also expected that microorganisms` biodegradation pathways will yield useful enzymes for biotechnology. Information obtained from genome sequences of soil microorganisms can lead to identification of useful reactions and pathways.