ABSTRACT
Study on the soil fertility status of University of Port Harcourt, Nigeria was carried out with respect to its land use types. The aim was to establish preliminary data for further studies in the area. Four soil samples were collected randomly from eight plots (10x10 m) length; air dried and analysed to assess the fertility status of the soils. The land use and dominant plant species were also noted. The average concentrations of the heavy metals and physico-chemical parameters showed that within the study area Potassium, K (0.94±1.19 mg kg-1), Lead, Pb (0.22±0.21 mg kg-1), Cadmium, Cd (0.11±0.16 mg kg-1), Manganese, Mn (0.54±0.40 mg kg-1), Chromium, Cr (0.22±0.21 mg kg-1), pH (4.58±0.10), Conductivity, (23.94±10.84 μS cm-1), Available Phosphorus PO43¯ (5.78±4.81 mg kg-1), Nitrate, NO3¯ (2.47±0.82 mk kg-1), organic matter, OM (6.46±2.18%) and Nitrogen, N (0.50±0.44%). Soils within the University Park were generally good for agricultural purposes. However, a little variation exists based on land cover and land-use types. The study establishes that the land-use and land cover system support the fertility of the soil and hence support growth of plant community structure which maintains the ecosystem. The data obtained would serve as preliminary information for further studies.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjb.2013.24.30
INTRODUCTION
Plants and animals depend on soil for their survival. In various parts of the world, different human activities have changed the status of the soils. These activities include; road construction, farming, oil exploration and exploitation, construction of houses and industries, etc. which introduces some of these pollutants to the soil. Heavy metals are toxic to plants and animals if an excessive concentration is absorbed. Uzoukwu and Onomake (2003) have reported comparison between heavy metals status of soil samples collected at different distances (5-200 cm) form the River bank situated where there are industrial activities and elsewhere. Also, Adekola et al. (2002) showed that pollutants released into the atmosphere could be absorbed by plants and animals. Changes in land-use are relatively easy to observe and quantify and several recent studies have focused on the prediction of land-use changes (Veldkamp and Lambin, 2001; Louisa and Antonio, 2002). According to Hartemink (2003), soil fertility decline and nutrient mining are perceived to be an important form of soil degradation in the agricultural land-use systems of the tropics. Lal (1997) earlier reported that soil degradation is the loss of actual potential productivity and utility, and soil degradation implies a decline in the soil’s inherent capacity to produce economic goods and perform environmental regulatory functions. The ability of a region of the world to produce or access food is determined access to an adequate amount of productive cropland; the ability to maintain high crop yields on that land; or the ability to purchase and import food from other regions (Ramankutty et al., 2002).
In University of Port Harcourt, there is an increase in the rate of deforestation associated with construction of buildings and other developmental projects. However, there is no preliminary data or report on the fertility status of soil and land-use within the University Park. Therefore, this study is aimed at establishing the soil fertility status of the University Park taking into consideration the land use and land cover types. The information obtained from the study would serve as preliminary data source for further studies.
MATERIALS AND METHODS
Sample collection: Eight plots (10x10 m) were mapped out within University of Port Harcourt Park (Table 1) and four samples (top, 0-15 cm and bottom, 15-30 cm depth) collected randomly from each of the plots using a stainless steel soil auger totaling eight (8) samples per plot. The soil samples were air dried and analysed to assess the fertility status of the soils.
Determination of pH: About 5 g of the air-dry soil was taken in a glass beaker and 10 mL of distilled water was added. The contents were mixed thoroughly with a glass rod and allowed to stand for 30 min. The soil pH was measured using EQUIP-TRONICS Digital pH meter model EQ-610.
Determination of soil conductivity: A Jenway 4010 conductivity meter was used for the determination of the electrical conductivity. The platinum electrode was cleaned using chromic acid and distilled water, as necessary, to keep the conducting surface free of contaminants. The samples were brought to room temperature (25°C). The meter and the electrode were set up to stabilize.
| Table 1: | Sample location/Land use/land cover of the study area |
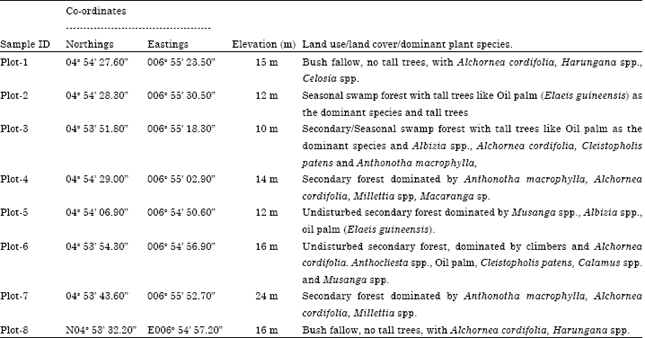 | |
Then, the electrode was immersed in the test sample containing 1:1 volume of soil-water suspension. The set-up was allowed to stand for 15 min to stabilize before taking the reading. Each of the samples followed similar treatment before the measurement.
Determination of total organic carbon: Two gram of soil was taken in a 500 mL Erlenmeyer flask, 10 mL of 1 N K2CrO7 was added and the flask was swirled to mix the contents. Twenty mL of conc. H2SO4 was added to the soil suspension; flask was swirled again for 1 min and allowed to stand for 30 min. After this, 200 mL of water, 10 mL of H3PO4 and 1 mL of diphenylamine indicator were added and the contents were titrated against 0.5 N FeSO4.7H2O until the colour changed from blue to red. The organic matter was obtained by multiplying total organic carbon values by a conversion factor of 1.27 (AOAC, 1990).
Nitrate determination (Brucine Method): The Brucine method was used for the determination of nitrate content of the soil samples the method involves colorimetric determination by the use of UV/Visible spectrometry as described by Uzoukwu and Onomake (2003) and Uzoukwu (1992) in accordance with EPA 352.1 and APHA 4500-NO3¯B.
Procedure: Two millimetres of water extract of the soil sample was used for nitrate determination and from a nitrate stock solution of 100 ppm prepared from NaNO3, standard solutions were made. Spectrophotometric determination of the analyses was carried out using a UV/Visible spectrophotometer at 410 nm against water blank. The absorbance values of the working solutions as well as those of the samples were measured. Water was used as reference. The concentrations of nitrate present in the samples were determined from a calibration curve.
Determination of Phosphate (Molybdenum Blue Method): The molybdenum blue method was used for the determination of phosphate content of the soil samples. This method is in accordance with APHA 4500-P D (APHA, 1989).
Procedure: Five gram of each of the samples was weighed and 15 mL of 2.5% acetic acid added. The solution was mixed thoroughly using stirring rod and left to stand for 24 h. The extracts obtained by decantation were used for determination of phosphate contents of the samples. Into each of the standards and sample extracts, 1.5 mL of the molybdenum reagent was added, followed by ascorbic acid and the colour allowed to develop within 10 min. The absorbance of the characteristic molybdenum blue colour of both sample and standards were measured using a spectrophotometer at 880 nm. The concentrations of the samples were determined using a calibration curve.
Sample digestion: The soil samples were digested on a labcon digester at 300°C in a mixture of hydrogen peroxide, sulphuric acid, selenium and salicyclic acid (Okalebo et al., 2002). The digests were analysed for total N, K, Pb, Cd, Mn and Cr. The total N content in the digests were determined by Kjeldahl method, 10 mL of the digest solution was taken in the distillation flask, 20 mL of 40% NaOH was added and the NH3 evolved was collected in a flask containing 4% H3BO3. Finally, the distillate was titrated against 0.1 N H2SO4.
RESULTS AND DISCUSSION
The results of the soil characteristics in the study area are presented in Table 1 to 6. The summary of the soil characteristics within University of Port Harcourt is presented in Table 2. The average concentrations of the heavy metals and physico-chemical parameters recorded in the study area are as follows: Potassium, K (0.94±1.19 mg kg-1), Lead, Pb (0.22±0.21 mg kg-1), Cadmium, Cd (0.11±0.16 mg kg-1), Manganese, Mn (0.54±0.40 mg kg-1), Chromium, Cr (0.22±0.21 mg kg-1), pH (4.58±0.10), Conductivity, (23.94±10.84 μS cm-1), Available Phosphorus PO43- (5.78±4.81 mg kg-1), Nitrate, NO3- (2.47±0.82 mk kg-1), organic matter, OM (6.46±2.18%) and Nitrogen, N (0.50±0.44%). The total nitrogen and available phosphorus were quite high except pH and K contents of the soil when compared with the work of Okonwu and Mensah (2012). The soil pH is slightly acidic with the mean value of 4.58. Similarly, the organic matter and organic carbon were consistent with the work of Ibeawuchi et al. (2007) although it was low. The lower organic carbon observed in some study sites can be attributed to low plant cover. Brady and Weil (2002) reported that vegetation removal reduces the soil organic matter and affects soil structure and functions of soil. It indicated disturbance factors such as collection of fuel wood, encroachment by agriculture and tree falling for construction which was more intensive than other land use systems. Feyssa et al. (2011) also reported that the higher soil organic matter content could be attributed to low value of bulk density in settled farmers land use systems.
| Table 2: | General characteristics of the soil within the study area |
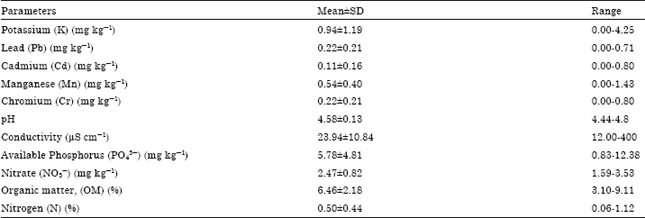 | |
| Table 3: | Characteristics of soil within Bush fallow/rain forest |
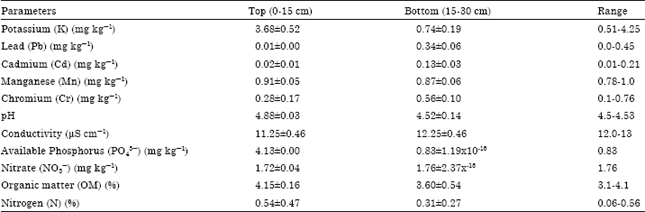 | |
Soils within the University Park is generally good for agricultural purposes however, based on land cover and land use types, the characteristics of soils samples with in the area showed little variation as shown in Table 2.
The soil characteristics within the bush fallow areas are presented in Table 3. Within these areas, the available phosphorus and nitrate contents of the bottom soil were low as compared to other areas studied. This is as a result of the previous farming activities in the areas. However, the ranges for parameters tested were Potassium, K (0.51-4.25 mk kg-1), Lead, Pb (0.0-0.45), Cadmium, Cd (0.01-0.21 mg kg-1), Manganese, Mn (0.78-1.0 mg kg-1), Chromium, Cr (0.1-0.76), pH (4.5-4.53), conductivity, (12.0-13.0 μS cm-1), Available Phosphorus, PO43¯ (0.83 mg kg-1), Nitrate, NO3¯ (1.76 mg kg-1), Organic matter, OM (3.1-4.1%) and Nitrogen, N (0.06-0.56%).
In the same vein, the results of the soils within the seasonal swamp forests are shown in Table 4. Potassium, K (0.0-0.65 mg kg-1), Lead, Pb (0.0-0.71 mg k g-1), Cadmium, Cd (0.01-0.08 mg kg-1), Manganese, Mn (0.0 – 0.24 mg kg-1), Chromium, Cr (0.0-0.80 mg kg-1), pH (4.65-4.80), conductivity, (14.0-28.0 μS cm-1), Available Phosphorus, PO43¯ (1.65-12.38 mg kg-1), Nitrate, NO3¯ (2.47-3.53 mg kg-1), Organic matter, OM (5.5-9.11%) and Nitrogen, N (0.11-1.12%).
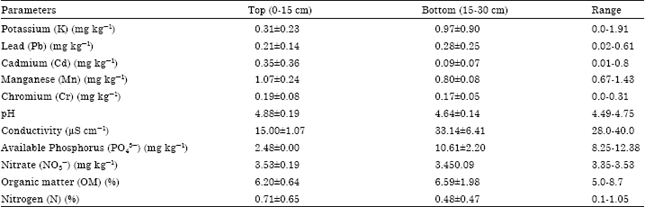
In the secondary rain forest areas of the study area, the concentrations of these different parameters ranges are Potassium, K (0.0-1.91 mg kg-1), Lead, Pb (0.02-0.61 mg kg-1), Cadmium, Cd (0.01-0.80 mg kg-1), Manganese, Mn (0.67–1.43 mg kg-1), Chromium, Cr (0.67-1.43), pH (4.49-4.75), conductivity, (28.0-40.0 μS cm-1), Available Phosphorus, PO43- (8.25-12.38 mg kg-1), Nitrate, NO3¯ (3.35-3.53 mg kg-1), Organic matter, OM (5.0-8.7%) and Nitrogen, N (0.1-1.05%) (Table 5).
| Table 4: | Characteristics of soil within seasonal swamp forests |
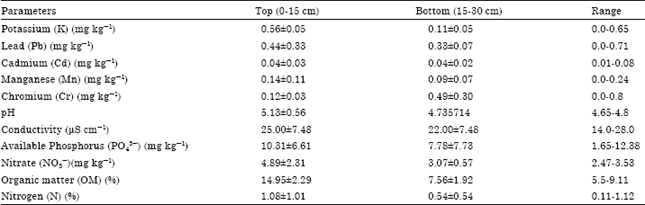 | |
| Table 5: | Characteristics of soil within secondary rainforests |
 | |
| Table 6: | Characteristics of soil within primary/undisturbed rainforests |
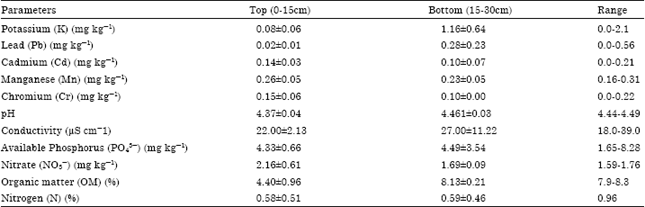 | |
The concentrations of Potassium (K), Lead (Pb), Cadmium (Cd), Manganese (Mn), Chromium (Cr), pH, conductivity, Available Phosphorus (PO43¯), Nitrate (NO3¯), Organic matter (OM) and Nitrogen (N) in the primary/undisturbed rain forests as shown in Table 6 ranged 0.0-2.1, 0.0-0.56, 0.0-0.21, 0.16-0.31, 0.0-0.22, 4.44-4.49, 18.0-39.0, 1.65-8.28, 1.59-1.76, 7.9-8.3 and 0.96 mg kg-1, respectively.
This result gives general characteristics of the soils within the University environment and is in line with Onwuka and Uzoukwu (2008) who analysed soil samples from the University Garden alone.
CONCLUSION
Maintaining the soil chemical fertility is a key prerequisite to sustaining crop productivity and the soil organic matter depends on the quantity of organic material added to the soil either by natural returns through roots, stubble, slough off roots nodules and root exudates. The study establishes that the land-use and land cover support the fertility of the soil and hence support growth of plant community structure which maintains the ecosystem. The data obtained would therefore serve as preliminary information for further studies.
REFERENCES
- Adekola, F.A., O.A. Eletta and S.A. Attanda, 2002. Determination of the levels of some heavy metals in urban run-off sediments in Ilorin and Lagos, Nigeria. J. Applied Sci. Environ. Manage., 6: 23-26.
Direct Link - Brady, N.C. and R.R. Weil, 2002. The Nature and Properties of Soils. 13th Edn., Prentice Hall, Inc. New York, USA, Pages: 976.
Direct Link - Ibeawuchi, I.I., F.A. Opara, C.T. Tom and J.C. Obiefuna, 2007. Graded replacement of inorganic fertilizer with organic manure for sustainable maize production in Owerri Imo State, Nigeria. Life Sci. J., 4: 82-87.
Direct Link - Jansen, L.J.M. and A. Di Gregorio, 2002. Parametric land cover and land-use classifications as tools for environmental change detection. Agric. Ecosyst. Environ., 91: 89-100.
CrossRefDirect Link - Okonwu, K. and S.I. Mensah, 2012. Effects of NPK (15:15:15) fertilizer on some growth indices of pumpkin Asian J. Agric. Res., 6: 137-143.
CrossRef - Ramankutty, N., J.A. Foley and N.J. Olejniczak, 2002. People on the land: Changes in global population and croplands during the 20th century. Ambio, 31: 251-257.
PubMedDirect Link - Uzoukwu, B.A. and O.R. Onomake, 2003. Physico-chemical studies of soil along the bank of Milli Shallow Stream in Nnewi, Anambra state, Nigeria for evidence of industrial pollution. Global J. Pure Applied Sci., 10: 111-119.
Direct Link - Veldkamp, A. and E.F. Lambin, 2001. Editorial: Predicting land-use change. Agric. Ecosyst. Environ., 85: 1-6.
Direct Link








