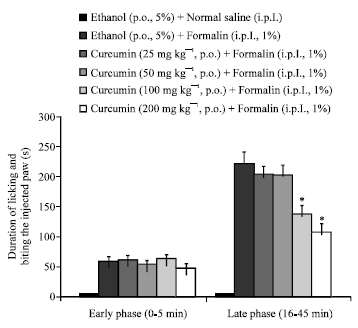
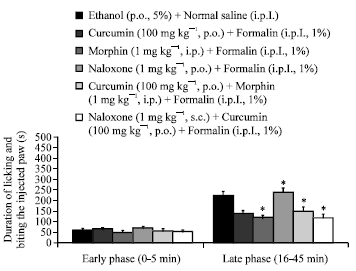
Research Article
Interaction Between Curcumin and Opioid System in the Formalin Test of Rats
Department of Food Hygiene
Esmaeal Tamaddonfard
Department of Physiology, College of Veterinary Medicine, Urmia University, Urmia, Iran
Nasrin Hamzeh-Gooshchi
Department of Physiology, College of Veterinary Medicine, Urmia University, Urmia, Iran