Research Article
Symptomatic and Asymptomatic Cryptosporidiosis in Young Children in Iran
Department of Microbiology, Faculty of Veterinary Medicine,Razi University, Postal Code 67156-8-5414, P.O. Box 1451, Kermanshah, Iran
Over the past decade, there has been a dramatic rise in the number of reports of C. parvum infections and outbreaks from all over the world (Smith and Rose, 1998). Estimates from United States public health records suggest that, 2% of all stools tested by health care providers are positive for C. parvum (Mead et al., 1999). Estimating, 15 million annual visits for diarrhea, infection with C. parvum might be expected in 300000 persons annually (Mead et al., 1999).
After ingesting C. parvum, immunocompetent humans can experience asymptomatic infection or self-limited diarrhea (DuPont et al., 1995). However, those with defects in innate (Kelly et al., 2000), humoral (Levy et al., 1997) or cellular immunity (Gomez Morales et al., 1996) can experience severe or prolonged illness. The life-threatening potential of C. parvum in immunocompromised and immunosuppressed individuals has increased the importance of Cryptosporidiosis as a global public health problem. Although C. parvum infections are usually of short duration and self- limiting in individuals with an intact immune system the lack of effective anticryptosporidial drugs (Reynoldson, 1999) means the very young and elderly may be at risk of severe disease as a result of C. parvum infection.
Though AIDS, immunosuppressive infectious diseases, diarrhea and enteric diseases are not uncommon in Iran, the present study was conducted to determine the presence and probably prevalence of C. parvum among young children in Iran.
Study population: The study was conducted during the period May to October 2003 in 515 children less than three years of age with diarrhea diseases presenting at the pediatric clinic of teaching hospitals in Kermanshah, Iran. The control group included 99 comparable children presenting at the same hospitals with complaints other than gastrointestinal symptoms. These hospitals cover a large area of population living in urban as well as in the rural areas. The parents of the children were interviewed to complete a questionnaire on demographic information: age, sex, area of residence, contact with animals and health status of the child.
Stool collection and examination: Single fecal samples were collected in plastic cups within two days of hospital admission or when the child presented at the hospital. All stool specimens were concentrated using the formalin-ethyl acetate method by centrifugation at 800 g in a fecal parasite concentrator (Zierdt, 1984) and two smears were made from the resulting pellet. The slides prepared with fecal pellet were used to detect Cryptosporidium parvum oocysts by a modified Ziehl Neelsen technique (Henriksen and Pohlemz, 1981). Briefly, slides were stained for 2 min with carbol fucshin and were destained for 30-60 sec with a 10% solution of sulfuric acid. After being washed, the slides were counterstained with methylene blue for 1 min. Red-stained Cryptosporidium parvum oocysts were observed microscopically using a 40x objective.
Statistical analyses: Data were analyzed using Chi-square and Fisher exact tests. Results were considered significant when the p-value was less than 0.05.
As shown in Table 1, 10.4% of stool samples were positive for Cryptosporidium infection. Cryptosporidium infection rates in diarrheic children (11.6%) were more than in non-diarrheic children (4.0%; p<0.05). Cryptosporidium parvum oocysts was observed more frequently in stool samples of children who lived in rural areas (15.2%) than those who lived in urban areas (7.2%; p<0.05). Shedding of oocysts were significantly higher in both diarrheic (15.1%) and non-diarrheic (16.0%) children who lived in rural areas than diarrheic (9.0%) and non-diarrheic (0.0%) children who lived in urban areas (p<0.05). In regard to the presence of animals, the infection rate of children who lived in association with animals (18.5%) was more than those who lived in compounds with no animals (8.2%; p<0.05). Shedding of oocysts was significantly higher (p<0.05) in diarrheic children (21.0%) who had contact with animals in comparison with those (9.1%) who had not contact with animals, the difference was not significant in non-diarrheic children with (5.0%) and without (3.8%) contact with animals (p>0.05). The infection rate was similar (p>0.05) between boys (10.5%) and girls (10.3%). The difference between diarrheic male (11.6%) and female (11.6%) and non diarrheic male (4.8%) and female (2.9%) was not significant (p>0.05).
As shown in Table 2, the majority of Cryptosporidium parvum cases occurred in children between the ages of 0-12 months (11.9%), followed by in children between the ages of 13-24 months (9.2%) and in children between the ages of 25-36 months (3.0%). The difference among children between ages of 0-12 and 13-24 months as well as 13-24 and 25-36 months was not significant (p>0.05), but more oocyst were isolated in age group of 0-12 months (p<0.05) in comparison with age group of 25-60 months. The prevalence rates for cryptosporidium infections in diarrheic children in age groups of 0-12 months (14.2%) and 13-28 months (9.0%) were similar (p>0.05), but in both groups were higher than the age group of 25-60 months (0.0%; p<0.05). In non-diarrheic children, infection rates was similar between age groups of 13-24 months (10.5%) and 25-60 months (20.0%) and in both groups were higher than (p<0.05) the age group of 0-12 months (0.0%).
| Table 1: | Socio-demographic data of children with and without diarrhea |
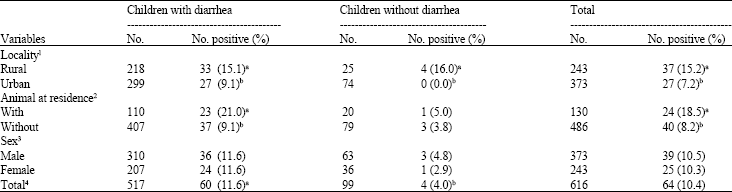 | |
| 1, 2, 3: Means within the same column with different letter(s) are significantly different (p< 0.05), 4: Means within the same row with different letter(s) are significantly different (p<0.05) | |
| Table 2: | Age-wise distribution of Cryptosporidium parvum in children with and without diarrhea |
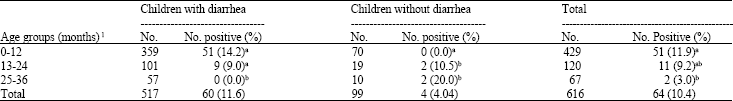 | |
| 1: Means within the same column with different letter(s) are significantly different (p<0.05) | |
The present study revealed existence of Cryptosporidium infection among children with and without diarrhea in Kermanshah Iran. Current et al. (1991) reported that acute infection with Cryptosporidium sp. among children and infants is more prevalent in developing countries (5% to >10%) than in developed countries (1%). The worldwide prevalence of Cryptosporidium among asymptomatic children is less than 0.5% (Vuorio et al., 1991). The overall percentage of diarrheic individuals positive for cryptosporidium infection in this study (11.6%) was lower than reported figures of 45 and 32% from Peru (Checkley et al., 1997) and Guatemala (Laubach et al., 2004), respectively, but was higher than corresponding figure of 5.6% reported from India (Pal et al., 1989). The proportion of Cryptosporidium infection associated with diarrhea in this population is similar to that reported from Venezuela (Chacin-Bonilla et al., 1997), Mexico (Enriquez et al., 1997) and Pakistan (Iqbal et al., 1999). The relatively high percentage of asymptomatic infection detected in this study (4.0%) is consistent with studies from developing countries (IQbal et al., 1999; Palit et al., 2005). In this study Cryptosporidium was isolated significantly more frequently from children with diarrhea than from those without diarrhea, suggesting that children with diarrhea are more likely to have Cryptosporidium oocysts identified in their stools; furthermore it appears that Cryptosporidium may be an unrecognized pathogen responsible for diarrhea and other gastrointestinal disorders in the pediatric population. It has been predicted that the proportion of asymptomatic carriers of important diarrheagenic pathogens like Cryptosporidium sp. may be high in area of low socio-economic status in developing countries (Chacin-Bonilla et al., 1993) which is very much evident in the present study. This is attributed to the unsanitary living condition, paucity of clean drinking water supplies, mixed dwelling habit (i.e., domestic/Pet animals are kept near or inside the houses), improper sewage or waste disposal facilities, intake of contaminated food, etc. However, over-crowding and previously unnoticed diarrhea in the family may also become positive factors, as found in a study in Mexico (Solorzano-Santos et al., 2000).
Our results confirm the fact that diarrheic children even symptomless individuals can have been colonised by Cryptosporidium, may be in very early in life, as has also been reported from a study in southern India (Mathan et al., 1985). This fact may be more alarming because it has already been shown (Checkley et al., 1998) that even asymptomatic cryptosporidial infection are associated with significant growth shortfalls in children in developing countries. In Africa, cryptosporidial diarrhea in the first year of life was associated with diminished linear growth effect with blunting of catch-up growth in young children (Molbak et al., 1997). Thus, cryptosporidial infection even without overt diarrhea may be an emerging cause of malnutrion in the developing world.
In the present study; the prevalence of Cryptosporidium infection in males and females were not statistically significant (p>0.05, Table 1), a finding that supports observation made in studies from Peru (Xiao et al., 2001), Korea (Jae-Ran et al., 2004) and Jordan (Mahgoub et al., 2004). Other studies showed higher percentage of Cryptosporidium infection amongst females as compared to that in males (Smith et al., 1989; Laubach et al., 2004). Smith et al. (1989) have stated that the reason for this higher proportion of females than males were unknown. However Laubach et al. (2004) have suggested that the male’s exposure to Cryptosporidium was affected by life style risk factors that increased the exposure of the females to untreated water.
In the present study, the infection rate in children who lived in rural areas was significantly (p<0.05) higher than those lived in urban areas, a finding that supports observation made in studies from Korea (Chai et al., 1996) and Jordan (Mahgoub et al., 2004). In this study the positive rate in rural areas was much lower than the figures of 43.5 and 32% reported from Jordan (Mahgoub et al., 2004) and Guatemala (Laubach et al., 2004), respectively, but was higher than corresponding figure of 3.3% reported by Jae-Ran et al. (2004) from Korea. The cryptosporidium infection rate in urban areas in this study is similar to figure of 8.64% reported from Pakistan (Iqbal et al., 1999) and lower than the corresponding figures of 32.1 and 24.67% reported from Jordan (Mahgoub et al., 2004) and Ghana (Adjei et al., 2004), respectively. An epidemiological study has indicated a remarkable difference in positive rates between rural area and urban area, from 10.6 to 0.5% (Chai et al., 1996). Most people in rural areas depend on wells and stored water (tanks) as a source of drinking water. Transmission of Cryptosporidium sp. by consumption of contaminated surface or ground water is well documented and waterborne outbreaks have been reported (D’Antonio et al., 1986; Rush et al., 1987). However, in spite of the low socioeconomic status of pediatric population in rural areas in this study, there was no significant difference between cryptosporidiosis and living condition such as presence of domestic animal at house. Although the positive rate in children with contact with animal was higher than those without contact; but the difference was not significant (p>0.05). In the present study, the majority of Cryptosporidium parvum cases occurred in children below two year of age (Table 2). This result is consistent with those of previous reports (Iqbal et al., 1999; Adjei et al., 2004; Palit et al., 2005).
In conclusion, the data presented here demonstrate the frequent occurrence of Cryptosporidium among children with and without diarrhea. Since Cryptosporidiosis appears to be more prevalent in developing countries than in the developed world and asymptomatic and symptomatic Cryptosporidial infections are associated with significant growth shortfalls in children (Molbak et al., 1997; Checkley et al., 1998), it would be logical to emphasize that the inclusion of a search for Cryptosporidium oocysts be considered part of the routine clinical microbiological examination of cases of diarrhea. Additionally, with the continuous rise in the number of HIV/AIDS-infected persons in Iran and the designation of Cryptosporidiosis as an AIDS-defining opportunistic infection, the routine examination for oocyst of cryptosporidium in diarrhea stools may be used as a first-line screening measure for clinically suspected AIDS patients in Iran.