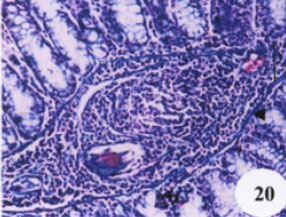
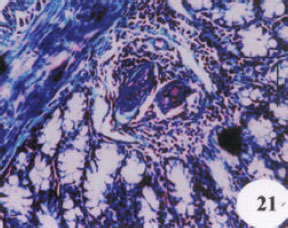
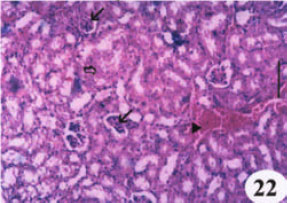
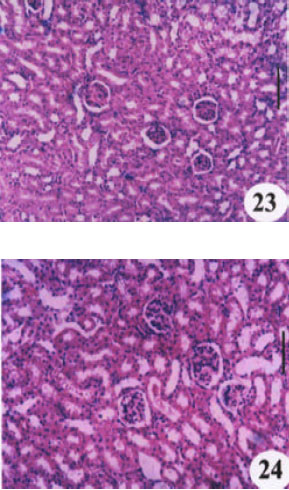
Research Article
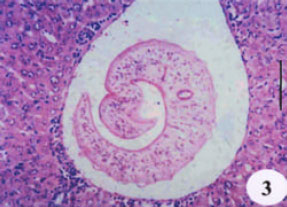
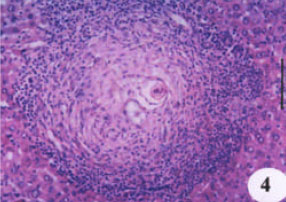
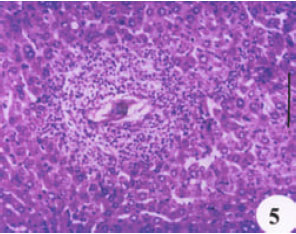
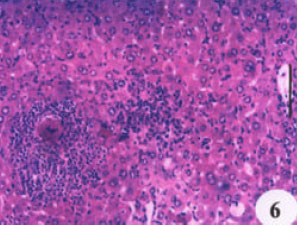
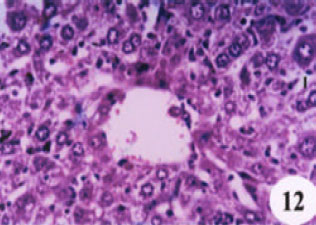
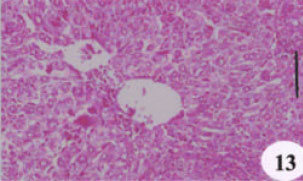
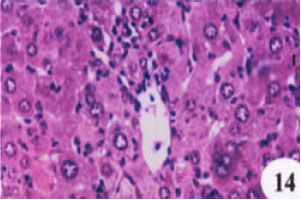
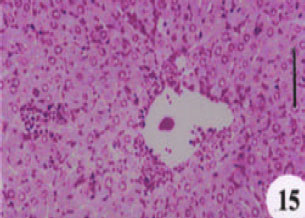
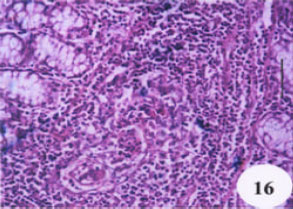
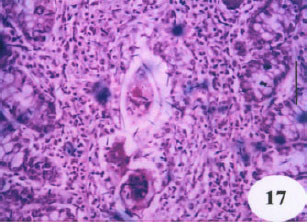
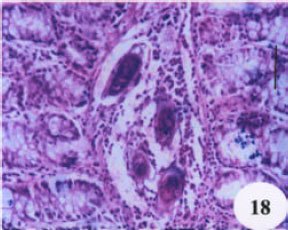
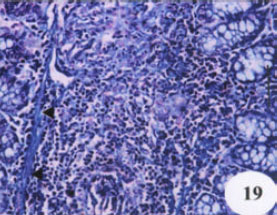
Evaluation of the Protective Effect of Two Antioxidative Agents in Mice Experimentally Infected with Schistosoma mansoni: Haematological and Histopathological Aspects
Zoology Department, Faculty of Science, Suez Canal University, Ismailia, Egypt
Nahla S. El-Shenawy
Zoology Department, Faculty of Science, Suez Canal University, Ismailia, Egypt