Research Article
Hepatoprotective Influence of Vitamin C on Thioacetamide-induced Liver Cirrhosis in Wistar Male Rats
Department of Biological Sciences, Faculty of Sciences, King Abdul Aziz University, P.O. Box 139109, Jeddah 21323, Saudi Arabia
The liver is a large and complex vital organ, and considered to be one of the most vital organs that functions as a centre of metabolism of nutrients such as carbohydrates, proteins and lipids, and excretion of waste metabolites. Additionally, it is also handling the metabolism and excretion of drugs and other xenobiotics from the body thereby providing protection against foreign substances by detoxifying and eliminating them (Meyer and Kulkarni, 2001; Saleem et al., 2010). Because the liver performs so many vital functions, liver damage can impact almost all body systems. In today's world, the liver has to work harder than ever before, and all over the world we find that liver problems are increasing. Globally, one in every ten persons suffers with some type of liver, bile duct or gall bladder disease. Liver cancer (hepatocellular carcinoma) is one of the ten most common cancers in the world with over 2,50,000 new cases each years and has a poor outlook (Gupta and Misra, 2006). Three hundred and fifty million people worldwide suffer from hepatitis B which kills more than 2 million annually. Hepatitis C is the most rapidly spreading infectious disease in many countries and is a time bomb waiting to explode. These problems are increasing, and thousands of people are waiting anxiously for liver transplants that many of them will never be lucky enough to receive. The liver disorders are a world problem. Despite its frequent occurrence, high morbidity and high mortality, its medical management is currently in adequate, no therapy has successfully prevented the progression of hepatic diseases, even though newly developed drugs have been used to treat chronic liver disorders these drugs have often side effects (Rao et al., 2006). Hence, there is an ever increasing need for safe hepatoprotective agents (Agarwal, 2001; Rao et al., 2006; Kuriakose and Kurup, 2010).
Chronic liver disease is responsible for over 1.4 million deaths annually according to data from the World Health Organization Mortality Database (WHO, World Health Report; http://www.who.int/en/) and in the western world this disease is among the top ten of disease-related causes of death (CDC, 2005). Overall there has been reported a 13% increase in the death rate from liver-related disease per year (Mocroft et al., 2005). Of the liver-related deaths, 77% were associated with viral hepatitis, 14% with alcohol abuse and 9% with hepatocellular carcinoma (Weber et al., 2006). Many etiological factors cause fibrosis and eventually lead to cirrhosis. It has been estimated that excessive alcohol consumption is a major contributor in 41-95% of deaths from cirrhosis in some countries (Yoon et al., 2005). The level and duration of alcohol consumption are important determinants in the development of liver pathology. As the primary site for detoxification of alcohol and its metabolites, the liver can go through the following pathological stages: fatty liver, alcoholic hepatitis, fibrosis and cirrhosis. Hepatic fibrosis is one of the processes that occur when the liver is damaged through viral activity, toxins, autoimmune diseases, metabolic disorder or genetic defects. It is a result of chronic liver injury that ultimately leads to cirrhosis and its complications of portal hypertension, liver failure and hepatocellular carcinoma. Millions of people die each year worldwide (Lai et al., 2003; Poynard et al., 2003; Lin and Kirchner, 2004; McClain et al., 2004; McHutchison, 2004). Hepatic fibrosis represents the response of the liver to diverse chronic insults such as parasitic disease, chronic viral infection (hepatitis B and C), immunologic attack (autoimmune hepatitis), hereditary metal overload, toxic damage, etc. Because of the worldwide prevalence of these insults, liver fibrosis is common and is associated with significant morbidity and mortality. The elucidation of the cellular and molecular mechanisms responsible for the development and progression of the liver fibrosis (Pinzani, 2000) has provided a sound basis for development of pharmacologic strategies able to modulate the course of the disease (Fujimoto, 2000; Kawada, 2004; Li et al., 2008). The cellular mechanisms of hepatic fibrosis are shared among the various insults and in many aspects, mirrors the scarring and wound-healing processes of other tissues (Friedman, 2000; Henderson and Iredale, 2007; Iredale, 2007). Hepatic fibrosis, regardless of the cause, is characterized by an increase in extracellular matrix (ECM) constituents, although the relative distribution within the liver lobule varies with the site and nature of the insult. In the injured liver, the Hepatic Stellate Cells (HSC) lying in the space of Disse beneath the endothelial cell layer, constitute the major source of the ECM proteins (Hautekeete and Geerts, 1997; Friedman, 2000; Luo et al., 2004; Lv et al., 2007). These cells are usually quiescent, with a low proliferation rate; on activation, probably because of hepatocyte injury, (Alcolado et al., 1997), they differentiate into myofibroblast- like cells, with high proliferative capacity (Saile et al., 2002). The predominant ECM protein synthesized by the HSC in fibrosis is collagen type I, which increases from approximately 2% of total proteins in a normal human liver to 10% to 30% in a cirrhotic liver. In experimental models of fibrosis of various types, the increased deposition of type I collagen results primarily from increased transcription of the type I collagen genes. In hepatic fibrosis, increases in the gene expression of other types of collagens such as III and IV and other matrix proteins have also been reported (Bedossa and Paradis, 2003; Hui and Friedman, 2003; Campbell et al., 2005; Takahara et al., 2006; Gnainsky et al., 2007). In addition to being caused by increased ECM synthesis, liver fibrosis may also result from relative imbalance between production and degradation of matrix proteins. It has been shown that activated stellate cells constitute the source of various collagenases and tissue inhibitors of metalloproteinases (TIMPs), which are necessary for the ECM remodeling (Arthur et al., 1998; Li and Friedman, 1999; Friedman, 2003; Lin et al., 2006). The lack of a specific inhibitor(s) of any component of the ECM in general and of collagen type I in particular, limits the success of prevention and treatment of hepatic fibrosis, although numerous agents have been tried (Dufour et al., 1998; Spira et al., 2002; Gieling et al., 2008; Pinzani and Vizzutti, 2008; Ramachandran and Iredale, 2009).Thioacetamide (TAA), also known as thioacetimidic acid, or acetothioamide (CH3CSNH2), is a widely used sulfur-containing compound both in the laboratory and in various technical applications (Mane et al., 2000; Sharma et al., 2000; Farawati and van den Berg, 2001; Spataru et al., 2005; Dodge et al., 2006; Manea et al., 2006; Houskova et al., 2007; Zaleska et al., 2007; Stengel et al., 2008) and can also be present in the environment as organic sulfur compounds (Sharma et al., 2000; Farawati and van den Berg, 2001; Dodge et al., 2006). TAA is used as a replacement for hydrogen sulfide in qualitative analyses and in hospital practice, as an accelerator in rubber vulcanization, as a reductant additive in the leather, textile and paper industries, generally having a similar role to thiourea (Sharma et al., 2000; Spataru et al., 2005; Manea et al., 2006). The most important recent technical applications and research studies concerning TAA use now, also draw attention to the usefulness of TAA in the preparation of photocatalytic nanocomposites, mesoporous nanomaterials and metal chalcogenide thin films (Mane et al., 2000; Houskova et al., 2007; Zaleska et al., 2007). The widespread uses of TAA are unavoidably accompanied by its toxicity and a number of ecotoxic effects in environmental pollution can result from TAA manipulation and its consequent presence in waste waters. Additionally, The primary routes of potential human exposure to TAA are inhalation and dermal contact. Potential consumer exposure to TAA residues could have occurred from contact with products where TAA was used as a solvent. Potential occupational exposure may occur during production and packaging of the chemical as a laboratory reagent. Chemists and laboratory technicians are at greatest risk of possible exposure. These aspects become increasingly important due to high degree of hepatic and neurotoxic injury, and carcinogenity, of TAA, which has been demonstrated under various experimental conditions (Wang et al., 2000; Li et al., 2002; Rahman and Hodgson, 2003; Yeh et al., 2004; Tunez et al., 2005; Constantinou et al., 2007; Huang et al., 2007). TAA, a well-known hepatotoxin, has been considered to be an inducer of liver cirrhosis (Eroglu et al., 2002; Kumar et al., 2004; Aydin et al., 2010). The bioactive metabolites of TAA namely TAA-sulfoxide and TAA-sulfdioxide, are well known hepatotoxins (Novosyadlyy et al., 2005) which causes hepatocellular necrosis in perivenous areas of the liver acinus. Prolonged administration of TAA leads to hyperplastic liver nodules, liver cell adenomas and hepatocarcinomas (Yeh et al., 2004). It has been demonstrated in rats that regenerative nodules and liver fibrosis are more prominent in the cirrhotic model induced by TAA and that the histology of the TAA model more closely resembles that of human liver cirrhosis (Li et al., 2002; Perez et al., 2002; An et al., 2006).
Vitamin C (C6H8O6), chemical names: ascorbic acid and ascorbate, is a water soluble antioxidant that was firstly isolated and characterized by Albert Szent-Györgyi in 1928, who was awarded the Nobel Prize in Medicine or Physiology in 1937 (Von Szent-Gyorgyi, 2005). Vitamin C is a six-carbon lactone which is synthesized from glucose by many animals. Vitamin C is synthesized in the liver in some mammals and in the kidney in birds and reptiles. However, several species including humans, non-human primates, guinea pigs, Indian fruit bats, and Nepalese red-vented bulbuls-are unable to synthesize vitamin C. When there is insufficient vitamin C in the diet, humans suffer from the potentially lethal deficiency disease scurvy (Pimentel, 2003; Larralde et al., 2007). Humans and primates do not synthesize ascorbic acid due to lack the terminal enzyme in the biosynthetic pathway of ascorbic acid, l-gulonolactone oxidase, because the gene encoding for the enzyme has undergone substantial mutation so that no protein is produced (Iqbal et al., 2004; Mandl et al., 2009). Vitamin C has several antioxidant properties. It is an essential cofactor involved in many biochemical functions and it acts as an electron donor or reducing agent, it is said to have ascorbate oxidant activity (Seghrouchni et al., 2002). Ascorbate effectively scavenge singlet oxygen, superoxide, hydroxyl and water soluble peroxyl radical and hypochlorous acid (Smirnoff and Wheeler, 2000). The goal of the present study was to evaluate the possibility of using vitamin C both to prevent and to treat hepatic cirrhosis induced by TAA.
Experimental rats: Healthy Wistar male rats weighing 120-150 g were used in this study. The rats were obtained from the Experimental Animal Unit of King Fahd Medical Research Center, King Abdul Aziz University, Jeddah, Saudi Arabia. Upon arrival to the experimental animal room, the rats were allowed to acclimatize for one week before starting the experimental investigations. The experimental rats were kept at 20±1°C with enough humidity and under controlled light periods. They were fed ad libitum on normal commercial chow and had free access to water.
Experimental design: Following the acclimatization period, the animals were divided into five groups, each of which contained 15 rats and they randomized into the following groups:
| • | Group 1: Control rats were given saline solution (0.9% NaCl) by intraperitoneal injection, twice weekly, for 10 weeks |
| • | Group 2: Rats were given 300 mg kg-1 body weight (b.wt.) of TAA (Sigma-Aldrich Corp., St. Louis, MO, USA) by intraperitoneal injection, twice weekly, for 10 weeks |
| • | Group 3: Rats were intraperitoneally injected with TAA at the same dose given to group 2 and after one hour given 100 mg kg-1 b.wt. of vitamin C (Loba Chemie Pvt. Ltd. India) by intraperitoneal injection, twice weekly, for 10 weeks |
| • | Group 4: Rats were intraperitoneally injected with vitamin C at the same dose given to group 3 and after 1 h exposed to TAA at the same dose given to group 2, twice weekly, for 10 weeks |
| • | Group 5: Rats were given 100 mg kg-1 b.wt. of vitamin C by intraperitoneal injection, twice weekly, for 10 weeks |
Biochemical and hematological estimations: After 10 weeks, rats were anaesthetized with diethyl ether. Blood samples were collected from orbital venous plexus in non-heparinized and heparinized tubes. Blood specimens in non-heparinized tubes were centrifuged at 2000 rpm for 20 min and the clear supernatants sera were obtained for biochemical estimations. Serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), creatine kinase (CK), glucose, triglycerides and cholesterol were measured using an automatic analyzer (Reflotron® Plus System, Roche, Germany). Serum gamma glutamyl transferase (GGT), total bilirubin, total protein, albumin and lactate dehydrogenase (LDH) was measured using Automated Clinical Chemistry Analysis System, Dimension® type RXL Max (Dade Behring Delaware, DE 19714, USA). Blood specimens in the heparinized tubes were immediately used for hematological evaluation including red blood corpuscles (RBC) count, hemoglobin (Hb) concentration, hematocrit (Hct) value and white blood corpuscles (WBC) count. These hematological parameters were determined using ADVIA Hematology Automatic System (USA).
Histological evaluation: After blood sampling, rats were dissected and the liver tissues were preserved in 10% formalin immediately after removal from the animals. The liver tissues were dehydrated by ascending grades of isopropyl alcohol by immersing in 80% isopropanol overnight and 100% isopropyl alcohol for 1 h. The dehydrated tissues were cleared in two changes of xylene, 1 h each. The wax impregnated tissues were embedded in paraffin blocks using the same grade wax. The paraffin blocks were morented and cut with rotary microtome at 4 micron thickness. The sections were floated on a tissue floatation bath at 40°C and taken on glass slides and smeared with equal parts of egg albumin and glycerol. The sections were then melted in an incubator at 60°C and after 5 min the sections were allowed to cool. The sections were deparaffinised by immersing in xylene for 10 min in horizontal staining jar. The deparaffinised sections were washed in 100% isopropyl alcohol and stained in Ehrlich’s hematoxylin for 8 min in horizontal staining jar. After staining in hematoxylin, the sections were washed in tap water and dipped in acid alcohol to remove excess stain (8.3% HCl in 70% alcohol). The sections were then placed in running tap water for 10 min for blueing (slow alkalization). The sections were counter stained in 1% aqueous eosin for 1 min and the excess stain was washed in tap water and the sections were allowed to dry. Complete dehydration of stained sections was ensured by placing the sections in the incubator at 60°C for 5 min. When the sections were cooled, they were mounted in DPX mount having the optical index of glass, the sections were wetted in xylene and inverted on to the mount and placed on the cover slip (Dunn, 1974). All liver sections were examined using light microscope and photographed.
Statistical analysis: Data were expressed as the mean±SD. Data were analyzed by one-way analysis of variance (ANOVA). Multiple comparative analyses were conducted between all experimental groups using Tukey's test. Results were considered statistically significant at p<0.05.
As shown in Fig. 1a-f, 2a-f and 3a-d, the chronic exposure to TAA increased the levels of serum total bilirubin (517.7%), triglycerides (107.8%), cholesterol (121.3%), ALT (60.9%), AST (108.9%), ALP (46.3%), GGT (444.1%), CK (73.4%) and LDH (130.5%), while the levels of glucose (27.2%), total protein (14.2%), albumin (17.3%), RBC (26.1%), Hb (25.2%), Hct (13.7%) and WBC (17.1%) were statistically decreased in rats of group 2 compared with control group.
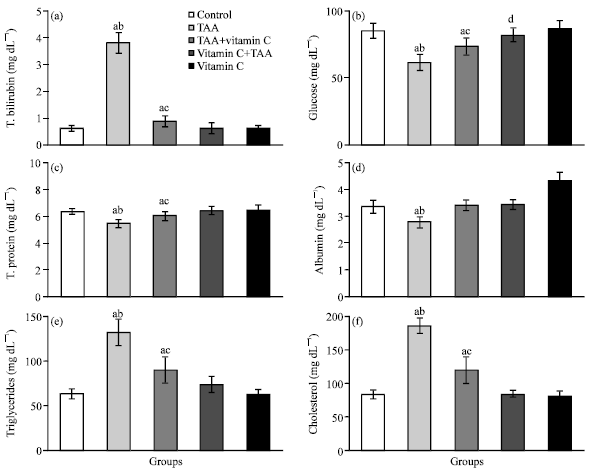 | |
| Fig. 1: | (a-f) The values of serum total bilirubin, glucose, total protein, albumin, triglycerides and cholesterol of control (group 1), TAA (group 2), TAA plus vitamin C (group 3), vitamin C plus TAA (group 4) and vitamin C (group 5) treated rats after 10 weeks (n = 6). a: Indicates a significant difference between control and treated groups. b: Indicates a significant difference between TAA treated group and groups treated with TAA plus vitamin C, vitamin C plus TAA and vitamin C. c: Indicates significant difference between TAA plus vitamin C treated group and groups treated with vitamin C plus TAA and vitamin C. d: Indicates significant difference between vitamin C plus TAA treated group and group treated with vitamin C |
In rats treated with TAA plus vitamin C (group 3), the levels of serum total bilirubin (43.6%), triglycerides (42.4%), cholesterol (43.2%), ALT (13.4%), AST (30.3%) ALP (12.8%) and GGT (83.6%) were statistically increased, while the values of glucose (13.4%), total protein (5.5%), RBC (6.1%) and WBC (13.1%) were declined compared with control values. Also, the levels of CK, LDH, albumin, Hb and Hct were significantly unchanged in rats of group 3. The value of AST (4.9%) was notably increased in rats treated with vitamin C plus TAA (group 4) compared with control group, while the level of serum glucose (2.8%) was decreased compared with rats treated with only vitamin C (group 5). The values of total bilirubin, total protein, albumin, triglycerides, cholesterol, ALT, ALP, GGT, CK, LDH, RBC, Hb, Hct and WBC were remarkably unchanged in rats of group 4. Additionally, the values of all biochemical and hematological parameters were statistically unchanged in rats treated with only vitamin C (group 5).
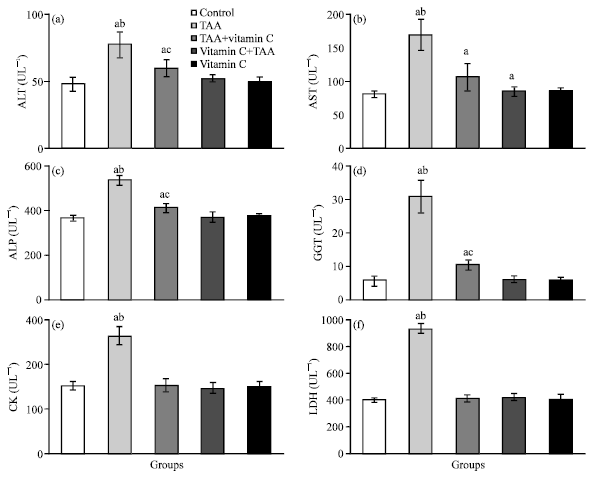 | |
| Fig. 2: | (a-f) The activities of serum ALT, AST, ALP, GGT, CK and LDH of control (group 1), TAA (group 2), TAA plus vitamin C (group 3), vitamin C plus TAA (group 4) and vitamin C (group 5) treated rats after 10 weeks (n = 6). a: Indicates a significant difference between control and treated groups. b: Indicates a significant difference between TAA treated group and groups treated with TAA plus vitamin C, vitamin C plus TAA and vitamin C. c: Indicates significant difference between TAA plus vitamin C treated group and groups treated with vitamin C plus TAA and vitamin C |
The liver histology of control group was found to be normal (Fig. 4a), that is, with normal architecture and without abnormal changes. Intraperitoneal injections of TAA for 10 weeks resulted in a uniform nodularity of the surface of the rat livers in group 2. As shown in Fig. 4b-e, the administration of TAA induced nodular transformations in liver parenchyma similar to those found in human nodular cirrhosis. Also, the parenchyma nodules surrounded by fibrous septae which divided the liver into pseudolobules. TAA administration induced centrilobular necrosis, hepatic cells surrounding central vei showed various degenerative changes like cloudy swelling, hydropic degeneration and necrosis with loss of nucleus. Livers of rats treated with TAA plus vitamin C (group 3) showed an appreciably histological regeneration (Fig. 4f) compared to those of TTA treated rats (group 2). They showed a reduced extent and development of fibrous septa and an increase in the extension of normal hepatic parenchyma, and in no case led to the nodular parenchyma transformation own to the cirrhotic process. The histopathological examination of the liver sections of rats treated with vitamin C plus TTA (group 4) and only vitamin C (group 5) showed normal hepatic lobular architectures with central veins and radiating hepatic cords (Fig. 4g and h).
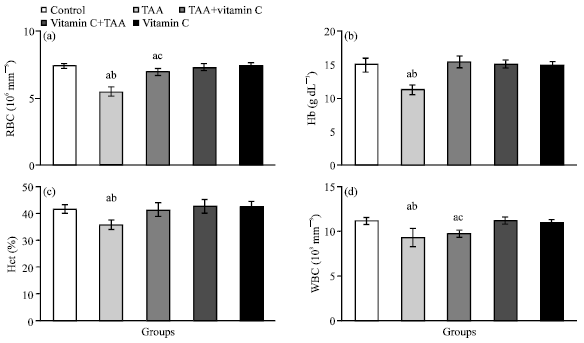 | |
| Fig. 3: | (a-d) The values of RBC, Hb, Hct and WBC of control (group 1), TAA (group 2), TAA plus vitamin C (group 3), vitamin C plus TAA (group 4) and vitamin C (group 5) treated rats after 10 weeks (n = 6). a: Indicates a significant difference between control and treated groups. b: Indicates a significant difference between TAA treated group and groups treated with TAA plus vitamin C, vitamin C plus TAA and vitamin C. c: Indicates significant difference between TAA plus vitamin C treated group and groups treated with vitamin C plus TAA and vitamin C |
Liver diseases are considered as one of the serious health problems. Steroids, vaccines and antiviral drugs that are employed as therapy for liver diseases have potential adverse effects especially when administered for long periods (Sehrawat et al., 2006). Additionally, a number of drugs, chemicals and viruses have been reported to cause severe liver necrosis, which sometimes becomes difficult to manage by medical therapies. It is important to search for compounds that can be used for better management of the hepatic failure due to severe necrosis. In the present study, the chronic administration of TAA caused liver cirrhosis as indicated by histopathological alterations and markedly elevated levels of serum total bilirubin, ALT, AST, ALP and GGT. These results are in accordance with many investigations on TTA-induced liver fibrosis and cirrhosis in experimental animals (Tripathi et al., 2003; Dai et al., 2005; Galisteo et al., 2006; Madani et al., 2008; Guerra et al., 2009; Aydin et al., 2010). As a result of necrosis, the levels of diagnostic indicators of myocardial infarction, such as CK and LDH, increase in the serum (Padmanabhan et al., 2005; Nigam, 2007). Also, it is well known that diagnosis of cardiac enzymes is important. Serum CK activity is a more sensitive indicator in early stage of myocardial ischemia, while peak rises in LDH is roughly proportional to the extent of injury to the myocardial tissue (Chatterjea and Shinde, 2002). These enzymes are tightly bound to the contractile apparatus of the cardiac muscle tissue and any serious insult to the heart muscle will evoke the release of these enzymes into the serum. The present results also demonstrated that the chronic administration of TAA induced cardiotoxicity manifested by a significant increase in serum of CK and LDH activities. This is due to leakage from the heart as a result of TAA induced necrosis. The present decline of serum glucose, albumin and total protein; and the increases of triglycerides and cholesterol levels indicate disturbances in carbohydrate, protein and lipid metabolism induced by TAA intoxication.
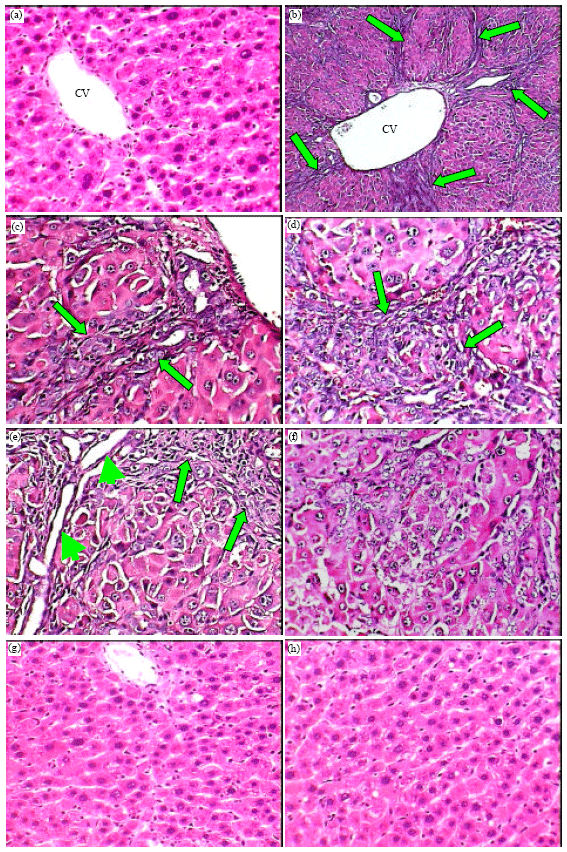 | |
| Fig. 4: | (a-h) Photomicrographs of liver sections of normal control rats (a, X400) showing normal histological structure; TAA treated rats (b, X100; c, d and e, X400) showing severe damage of liver structure including the formation of pseudolobules with fibrotic septae (arrows) and necrotic areas (head arrows); TAA plus vitamin C treated rats (f, X400) showing disarrangement of hepatic strands and an absence of fibrotic septae; vitamin C plus TAA (g, X400) and vitamin C treated rats (h, X400) showing normal structures |
Blood glucose concentration is known to depend on the ability of the liver to absorb or produce glucose. The liver performs its glucostatic function owing to its ability to synthesize or degrade glycogen according to the needs of the organism, as well as via gluconeogenesis (Ahmed et al., 2006). Kruszynska and McIntyre (1991) reported that the blood sugar level after overnight fasting in cirrhotic patients is believed to decrease only in severe hepatic failure. Trennery and Waring (1983) and Galisteo et al. (2006) reported that TAA intoxication models registered significantly lower plasma total protein levels compared to those of the healthy models. This alteration could be related to the induction of ubiquitin-associated protein degradation by TAA toxic stress (Andersen et al., 1981). Tripathi et al. (2003) showed that the levels of serum cholesterol were decreased in adult male albino rats intoxicated with TAA, carbon tetrachloride or paracetamol. The present results demonstrated that the chronic TAA exposure was associated with low levels of RBC, Hb, Hct and WBC compared to control rats. Hemopoietic and leukocytic are two dynamic systems which react quickly to chemical intoxications, and condition the maintenance of homeostasis by an organism. However, the observed effects of TAA, which represented by decline levels of RBC, Hb, and Hct are generally in agreement with the results of several investigations on the animals treated with different chemical factors (Rafatullah et al., 1991; Al-Attar, 2007; Krishna and Ramachandran, 2009; Salah et al., 2009; Al-Attar and Al-Taisan, 2010). From the present study, it is obviously that the administration of vitamin C after TAA intoxication led to decrease the severe biochemical, hematological and histopathological alterations. But more overt protections were observed in vitamin C plus TAA treated rats. The pretreatment of rats with vitamin C prevented the rise in the serum levels of total bilirubin, ALT, AST, ALP, GGT, CK, LDH, triglycerides and cholesterol, the decline in the serum levels of glucose, albumin and total protein, and the reduction of blood RBC, Hb, Hct and WBC values. Also, the liver sections of the rat treated with the vitamin C followed by TAA intoxication exhibited a significant protection as it was evident by normal cellular architecture with distinct hepatic cells and the absence of fibrosis, necrosis, tissue damage and vacuoles.
The possible mechanism of vitamin C as hepatoprotective factor may be due to its antioxidant effect which impair the activation of TAA into the reactive form. Bruck et al. (2004) stated that the chronic of TAA administration induced liver cirrhosis and oxidative stress. Cruz et al. (2005) reported that TAA induced liver fibrosis, oxidative stress with extensive tissue damage and enhanced alpha-smooth muscle actin expression in rats liver. Also, Aydin et al. (2010) showed that TAA administration resulted in hepatic fibrosis, significant increases in plasma transaminase activities as well as hepatic hydroxyproline and lipid peroxide levels, while liver glutathione (GSH), superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) protein expressions and activities decreased. It was reported that the administration of vitamin C ameliorated the necrotic and fibrotic changes in carbon tetrachloride-induced liver damage (Halim et al., 1997). Ascorbic acid is a potent scavenger of reactive oxygen species in plasma and extracellular compartments of the liver (Inoue, 2001). Ascorbic acid scavenges and destroys free radicals in combination with vitamin E and glutathione. George (2003) reported that the drastic decrease of ascorbic acid in dimethylnitrosamine (DMN)-induced hepatic fibrosis may indicate increased oxidative stress, free radical formation, and simultaneous damage of the liver plasma membrane lipid bilayers. The increased oxidative stress and the formation of reactive oxygen species cause extensive necrosis of the liver, which finally contributes to the development of fibrosis and cirrhosis. After scavenging the reactive oxygen species, an amount of ascorbic acid could regenerate either enzymatically using monodehydroascorbate reductase, or nonenzymatically by spontaneous dismutation. The enzymatic regeneration of the ascorbic acid may occur principally intracellularly at the expense of reduced glutathione. The highly reduced concentrations of glutathione in liver diseases (Bianchi et al., 1997; Jain et al., 2002) may affect the enzymatic regeneration of the ascorbic acid, which results in the sacrifice of ascorbic acid during scavenging of reactive oxygen species in liver diseases, including DMN-induced hepatic fibrosis. Under physiologic conditions, the regenerating system of ascorbic acid may function normally, but during extreme necrosis of the liver, it may be impaired, which contributes to a decreased ascorbic acid concentration in both liver and circulating system (George, 2003).On the light of the results obtained from this study, it should be pointed out that the pretreatment of vitamin C has a hepatoprotective action on TAA-induced hepatic cirrhosis and that is possibly due to its antioxidant and free radical scavenging effects. Finally, these results warrant further investigation on vitamin C as a potential treatment in other models of hepatic cirrhosis induced by different pathogens.