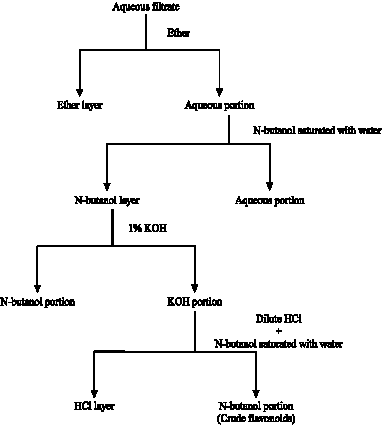
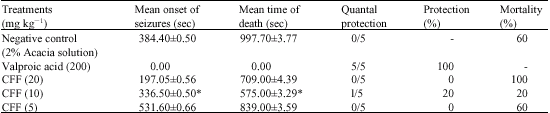
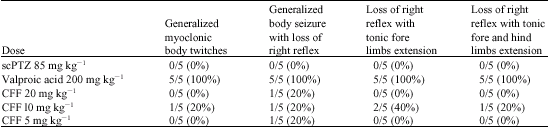
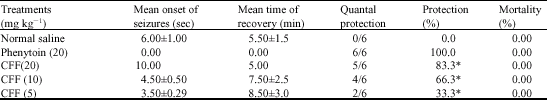Research Article
Anticonvulsant Activities of Crude Flavonoid Fraction of the Stem Bark of Ficus sycomorus (Moraceae)
Department of Pharmacognosy and Drug Development, Ahmadu Bello University, Zaria, Nigeria
S. Abdulmumin
Department of Pharmacognosy and Drug Development, Ahmadu Bello University, Zaria, Nigeria
K.Y. Musa
Department of Pharmacology, Bayero University, Kano, Nigeria
A.H. Yaro
Department of Pharmacology, Bayero University, Kano, Nigeria