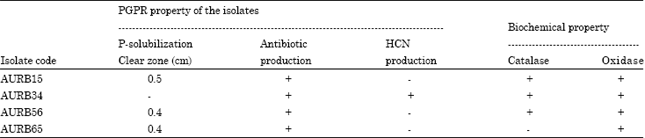
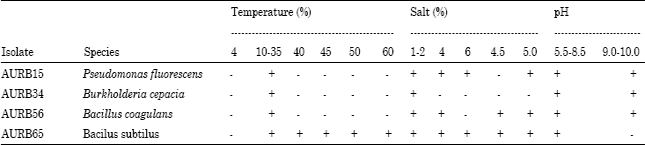
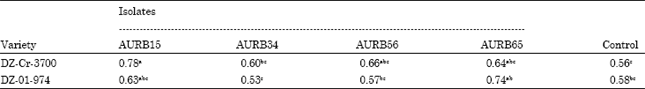
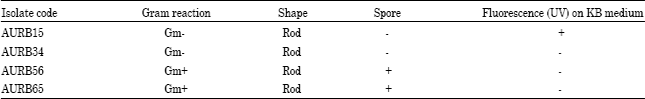Research Article
Effects of Plant Growth Promoting Rhizobaceria on Growth and Yield of Tef (Eragrostis tef Zucc. Trotter) under Greenhouse Condition
Department of Biology, Jimma University, College of Natural Sciences, P.O. Box 378, Jimma, Ethiopia
Fassil Assefa
Department of Biology, Addis Ababa University, P.O. Box 1176, Addis Ababa, Ethiopia