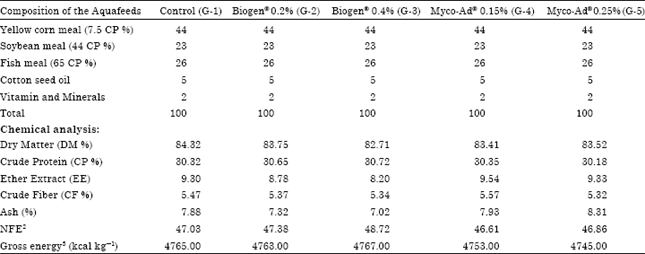
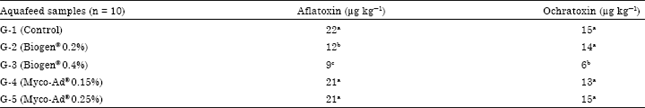
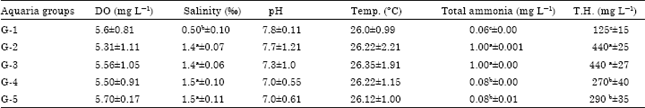
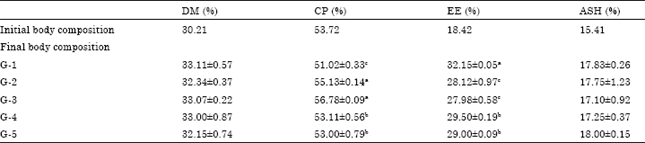
Research Article
Effect of Biogen® and Myco-Ad® on the Growth Performance of Common Carp (Cyprinus carpio) Fed a Mycotoxin Contaminated Aquafeed
Central Laboratory of Agriculture Research, Abbassa, Sharkia, Agricultural Research Center, Egypt
W. Anwer
Department of Veternairy Hygiene and Management, Faculty of Veternairy Medicine, Cairo University, Egypt