Research Article
The Influence of Feeding Ration on the Acute Stress Response of Beluga (Huso huso)
Islamic Azad University, Savadkooh Branch, Mazandaran, Iran
A. Kousha
Department of Fisheries, Islamic Azad University, Ghaemshahr Branch, Iran
The chondrostean fishes, comprised of the sturgeons and paddlefishes, apparently originated as a group in fresh water in the early Triassic Period (Barton et al., 2000; Bahmani et al., 2001). They are Anadromous and Potamodromous species of the Northern Hemisphere (Asadi et al., 2006). The evolutionary history of which goes back to 100 million years, apparently originated as a group in fresh water in the early Triassic Period and includes 28 species, of which 6 species inhabit the Caspian basin (Barton et al., 2000; Bahmani et al., 2001).
The complex life cycles of many fish species together with their varying environmental conditions often mean that periods of fasting or reduced food availability are a natural part of the life cycle (Ruane et al., 2002). Even in intensive rearing facilities fish. Even in intensive rearing facilities fish may be exposed to conditions of low feeding levels once they have attained the required market size, or before stressful procedures such as transport or grading. Although fish are well adapted to such periods of low food intake, these procedures can influence many aspects of the fishes` physiology (Ali and Wootten, 1999; Jobling et al., 1999; Ruane et al., 2002). From studies on starvation in a number of fish species (Ruane et al., 2002), two physiological states are known to occur. An initial short-term phase involves the mobilization of metabolites and a subsequent reduction in energy (glycogen) stores, while a second phase is associated with weight loss and increased lipid and protein catabolism, conserving energy stores (Farbridge and Leatherland, 1992; Davis et al., 2001; Ruane et al., 2002). The pancreatic hormones insulin and glucagon regulate these responses through modifications of glycolysis, gluconeogenesis and lipolysis during these periods of limited or low food availability (Sundby et al., 1991; Ruane et al., 2002).
Beluga undergoes a stress response with a release of glucocorticoids during management practices (Bahmani et al., 2001). Stress is not inherently detrimental, but it does jeopardize the animal`s welfare if the stress causes a significant biological cost (Belanger et al., 2001).
Cortisol, an important stress-related hormone in fish, which combines both glucocorticoid and mineral corticoid functions (Weerd and Komen, 1998), has been shown to exert an important influence on energy partitioning during stress (Vijayan and Moon, 1992; Ruane et al., 2002). As the ability to cope with and respond to stress involves a metabolic cost (Blom et al., 2000; Ruane et al., 2002; Belanger et al., 2001), it is therefore conceivable that the energy status of an animal, primarily its prior feeding history, may affect its response to stressful stimuli. Little is known about the influence of high or low food rations on stress responses in fish and relatively few studies have examined the effects of starvation on the stress response of fish. Barton et al. (2000) demonstrated that chinook salmon Oncorhynchus tshawytscha, fed daily had a higher glucose response to stress than fish starved for 20 days (Blom et al., 2000). In contrast, rainbow trout O. mykiss, deprived of food for 30 days had generally higher post-stress levels of cortisol and glucose than fed fish (Vijayan and Moon, 1992). Ruane et al. (2002) illustrated that feeding history influenced the onset of the stress response with stressor-induced elevations of plasma cortisol, glucose and free fatty acids being higher in common carp fed a high ration compared with those fed a low ration prior to sampling.
The Beluga sturgeon, Huso huso, is an anadromous species endemic to the waters of Caspian Sea and rivers following into it. Different aspects about ecology and biology of this primitive bony fish are well studied. The stocks of this valuable species have been greatly reduced by the impact of over fishing and habitat deterioration (Bahmani et al., 2001). Interest in this species was recently aroused by successful attempts at artificial reproduction, specially because of its high growth rate (Bahmani et al., 2001), which suggest that Huso huso may be suitable for fish farming and restocking. Sturgeon culture systems are intensive and involve various management stressors such as handling, crowding, transportation and biopsy and hormonally induced spawning. Knowledge of the stress response in these fishes may help to improve management, production and animal welfare.
The aim of this study was to determine to which extent prior feeding history, exerts an influence on growth and the ability of the beluga, Huso huso, to respond to an acute stressor.
Origin and Maintenance of Fish
Forty five one year old reared beluga; Huso huso reared beluga sturgeon (Huso huso), derived from artificial reproduction and were reared at the International Sturgeon Research Institute, Rasht, Gilan province, Iran from March to Jun 2006 for 90 days. Fish were maintained in Square fiberglass tanks, with volume of 1,000 L, supplied with fresh water at 15°, 7.3 pH, 7 ppm dissolved oxygen and 0.1 L sec-1 discharge. Fish were acclimated for 15 days before starting experiment.
Feeding Regimes
Sturgeons were fed on commercial dry pellets (45% protein, 14% fat, 10% carbohydrate, Chine Co, Iran) and were divided into three triplicate treatment groups based on control and high (H) or low (L) feeding levels as follows:
| • | Sturgeons in control treatment raised on an optimal feeding level of 4% of body weight per day during the experiment. |
| • | The fishes in High treatment (H>L) was fed 4% of body weight per day for 60 days and food ration was reduced to 1% of body weight per days in the last month of experiment. |
| • | The fishes in Low treatment (L>H) was fed 1% of body weight per day for 60 days and food ration was suddenly increased to 4% of body weight per days in the last month of experiment. |
Growth was checked every 20 days by measuring weight (Wt.) and Total Length (TL) and condition factor (K-factor), Growth Rate (GR), Specific Growth Rate (SGR) and Food Consumption Ratio (FCR) were calculated according to the formula (Kissil et al., 2001):
K = (Weight (g)/Length (cm)3) x100 SGR = (ln W2 – W1) (g)/t2 – t1 (day) GR = W2 – W1 (g)/t2 – t1 (day) |
Netting Procedure
For the application of the netting stressor, only two tanks of each treatment group were netted. Briefly, all fish were caught in a single net and five fish were immediately anaesthetized as time zero controls. The remaining fish were quickly placed in 5 L nets (five fish per net) which were suspended in identical fiberglass tanks. Each experimental tank had one corresponding confinement tank. The confinement tanks received a flow of water at 20 L min-1 and were well aerated. One net was removed from each confinement tank after 0.5, 1 and 3 h. After the 3 h sample point, the remaining two groups were placed in recovery tanks and sampled after 3 h of recovery.
Sampling Condition
Sturgeon were sampled after anesthesia in buffered solution of tricaine methane sulphonate (MS 222), which induced the cessation of body and opercular movements within 4-8 min after exposure. Fishes were fasted for 12 h before sampling. Two milliliters of blood specimens were collected from the caudal vein. Clotted blood was centrifuged at 3000 x g for 10 min and serum was stored at -20 °C for further analysis.
Plasma Analysis
Plasma cortisol levels were measured by radioimmunoassay. Plasma glucose was determined by the GOD-PeridR method (Boehringer, Mannheim). Plasma cholesterol and triglyceride were determined enzymatically using Sigma Diagnostic Kits.
Statistical Analysis
Range values, means and standard error of mean (SEM) were determined for all the parameters examined. Differences between groups was determined using t-test. Significance was accepted at p<0.05.
Growth Parameters and Physiological Indices
Growth of the fish during the experimental period is shown in Table 1. Fish when fed at the higher feeding level grew faster, as seen from the higher growth rates and lower food conversion values for these groups.
| Table 1: | Effect of feeding rate on growth indices in beluga sturgeon (n = 15 in each treatment) |
 | |
| Table 2: | Effect of feeding rate on growth indices in beluga sturgeon (n = 15 in each treatment) |
 | |
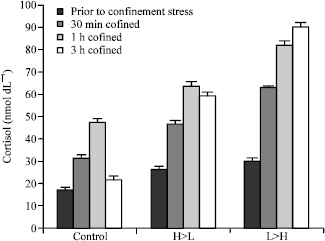 | |
| Fig. 1: | Effect of 3 h net confinement on cortisol levels in 3 groups of beluga sturgeon with different feeding rate on (n = 15 in each treatment) |
Feeding level was found to significantly affect (p<0.05) the total length, weight, FCR, SGR and GR that fish from groups of control and H>L are bigger than L>H. No difference in growth indices was found between the H>L and control groups.
Influence of Feeding Levels on Basal Cortisol and Metabolite Levels
Plasma cortisol and metabolite levels prior to confinement are shown in Table 2. A significant effect of feeding rate on stress and biochemical levels was with fish on a control treatment prior to sampling having significantly lower values than the other groups (p<0.05). Feeding level also had a significant effect on plasma triglyceride with fish from the H>L group having significantly (p<0.05) lower values.
Influence of Feeding History on the Cortisol, Glucose and Fish Response to Confinement
All groups showed a significant (p<0.05) increase in plasma cortisol levels at 0.5 and 1 h confinement with a gradual return to basal levels in control and H>L group after 3 h confinement (Fig. 1). During confinement, fish fed a normal feeding level prior to sampling (control) had significantly lower (p<0.05) post-stress cortisol levels than L>H and H>L fishes at 30 min confinement. Significant difference was found between H>L and L>H fishes at 30 min confinement (p<0.05). No difference between H>L and L>H groups was noted after 1 and 3 h confinement, but significant difference (p<0.05) was found between these groups with control treatment after 1 and 3 h confinement. The levels of cortisol decreased significantly to basal levels in control treatment after 3 h confinement. Insignificant decrease of cortisol after 3 h confinement was also observed in H>L group.
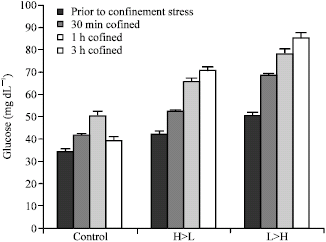 | |
| Fig. 2: | Effect of 3 h net confinement on glucose levels in 3 groups of beluga sturgeon with different feeding rate on (n = 15 in each treatment) |
 | |
| Fig. 3: | Effect of 3 h net confinement on triglyceride levels in 3 groups of beluga sturgeon with different feeding rate on (n = 15 in each treatment) |
Glucose significantly increased (p<0.05) with time in both L>H and H>L groups (Fig. 2). L>H and H>L fishes showed significant levels of hyperglycemia at all times during the confinement in comparison to control. Fish from the control group had significantly higher glucose levels after 30 min and 1 h confinement than prior to confinement, but returned to pre-stress values after 3 h. Compared with the L>H group, fish fed a low ration (H>L) had significantly lower (p<0.05) plasma glucose levels after 0.5, 1 and 3 h of confinement. Confinement significantly increased (p<0.05).
All groups showed a significant (p<0.05) increase in plasma cholesterol and triglyceride levels at 0.5, 1 and 3 h confinement with a slow return to basal levels after 3 h confinement in control and H>L group (Fig. 3, 4). During confinement, fish fed a normal feeding level prior to sampling (control) had significantly lower (p<0.05) cholesterol and triglyceride levels than L>H and H>L fishes at 30 min confinement.
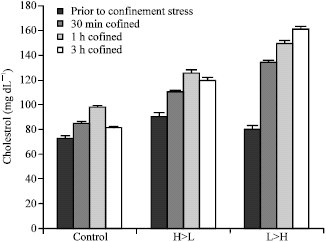 | |
| Fig. 4: | Effect of 3 h net confinement on cholesterol levels in 3 groups of beluga sturgeon with different feeding rate on (n = 15 in each treatment) |
Significant difference was found between H>L and L>H fishes at 30 min confinement (p<0.05). No difference between H>L and L>H groups was noted after 1 and 3 h confinement, but significant difference (p<0.05) was found between these groups with control treatment after 1 and 3 h confinement. The levels of cholesterol and triglyceride decreased significantly to basal levels in control treatment after 3 h confinement. Insignificant decrease of cortisol after 3 h confinement was also observed in H>L group.
Very few studies have investigated the effect of maintenance feeding levels or starvation on the acute stress response in fish.
The fact that growth efficiencies increase as the feeding level increases from maintenance to the optimal level is well known for many fish species (Huisman, 1976; Hogendoorn, 1983; Ruane et al., 2002) and is illustrated by the growth of the experimental groups on different feeding levels in this study. The results from this study further indicate the importance of environmental influences on the stress response in fish (Weerd and Komen, 1998) and demonstrate that fish fed a suitable food ration responded with less severity to an acute stressor. As physiological indicators of stress may not always correlate with the severity of the stressor (Davis et al., 2001), it is not known whether this lower response reflects an actual reduced stressed state of the fish or whether it is an impaired response induced by high, low or suitable food intake. Vijayan et al. (1990), Ruane et al. (2002) and Belanger et al. (2001) stated that fish under chronic stress with lower energy reserves have an impaired ability to compensate for additional stressors.
This study demonstrates that beluga maintained on a low feeding level showed a lower cortisol, glucose, cholesterol and triglyceride response to stress compared with fish fed a higher feeding level prior to confinement. Furthermore significant increase of cortisol was reported in control group after 0.5 and 1 h confinement and return to basal level quickly. The same result was reported by Ruane et al. (2002) on common carp. Conflicting results have been reported regarding starvation studies, with increased post-stress cortisol levels found in rainbow trout (Vijayan and Moon, 1992) or no effect in white suckers, Catostomus commersoni (Bandeen and Leatherland, 1997). Barton et al. (2000) found that starved chinook salmon also had lower cortisol levels 12 and 24 h after a brief handling stressor compared with fed fish (Blom et al., 2000). Although these findings may reflect experimental or species differences, it may be possible that a restricted feeding level can affect the Hypothalamo Pituitary Interrenal (HPI) response through effects on brain neuropeptide concentrations. A number of these neuropeptides such as adrenocorticotropin hormone (ACTH), α-melanophore stimulating hormone (α-MSH), β-endorphin (β-END) and serotonin (5-HT) are known to be involved in feeding behavior (Lin et al., 2000) and in the regulation of the stress response (Pottinger and Moran, 1993; Weerd and Komen, 1998; Winberg et al., 1997). Winberg et al. (1997) demonstrated lower concentrations of serotonin 5-HT in starved Arctic charr, Salvelinus alpinus, although Jobling et al. (1999) found no consistent effect of feed restriction on brain 5-HT concentrations in whitefish, Coregonus lavaretus. The peptide leptin, which is involved in the maintenance of energy balance, is known to act via its hypothalamic receptor on the regulation of these neuropeptides in mammals. Although leptin has been identified in fish and is known to be affected by feeding levels (Johnson et al., 2000), a link with the stress response in fish is as yet unknown.
The mobilization of glucose and triglyceride as major energy substrates during confinement in different species has been demonstrated previously by Ruane et al. (2001, 2002). Plasma glucose levels prior to the stressor were similar between the groups in this study as levels were maintained possibly by glycogenolysis and gluconeogenesis as is often seen in food deprived fish (Ruane et al., 2001, 2002). The hyperglycaemic stress response of H>L fish was lower, particularly at the initial stages of confinement. Barton et al. (2000) also showed a lower glucose response to stress in chinook salmon starved for 20 days compared with fed fish and suggested that this was related to the depleted stores of glycogen found in food deprived fish (Blom et al., 2000) and the same result was reported by Ruane et al. (2002) on common carp. As reduced glycogen stores in the hepatopancreas of fishes during food restriction and starvation has been shown by Shimeno et al. (1997), it is possible that reduced glycogen in fish fed a low ration in this study resulted in the lower level of stress-induced hyperglycaemia.
Plasma triglyceride and crude fat levels in the hepatopancreatic tissue and whole body of fish were also found to decrease with reduced food rations (Shimeno et al., 1997). As the mobilization of glucose and triglyceride are controlled by a number of enzymes, the activity of these enzymes becomes an important factor during stress. Many of these enzymes (such as pyruvate kinase, glycogen phosphorylase, phosphofructokinase) are down-regulated during periods of food deprivation (Mendez and Weiser, 1993; Shimeno et al., 1997), which may further contribute to the lower level of substrate mobilization in low-ration fish.
The fact that gross growth efficiencies increase as the feeding level increases from maintenance to the optimal level is well known for many fish species (Ruane et al., 2002) and is illustrated by the growth of the experimental groups on different feeding levels in this study. The results from this study further indicate the importance of environmental influences on the stress response in fish (Belanger et al., 2001) and demonstrate that fish fed a lower food ration responded with less severity to an acute stressor. As physiological indicators of stress may not always correlate with the severity of the stressor (Davis et al., 2001), it is not known whether this ‘lower` response reflects an actual reduced stressed state of the fish or whether it is an impaired response induced by a low food intake. Vijayan et al. (1990) stated that fish under chronic stress with lower energy reserves have an impaired ability to compensate for additional stressors. The results of this research reveal that rearing condition of fish used for experimental purposes has an important influence on the subsequent results, a fact which is often given little attention in many experiments.
The authors would like to thanks colleagues at Islamic Azad University, Savad Kooh Branch, for their helps and financial supports by grant No. 14/94500.