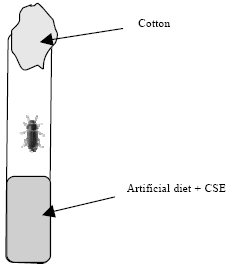
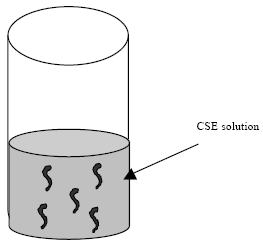
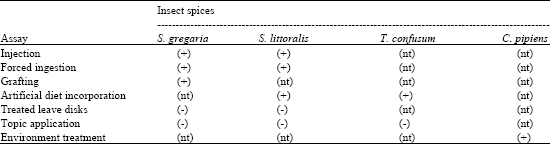
Research Article
Toxicity Experiments of the Saponic Extract of Cestrum Parqui (Solanaceae) on Some Insect Spices
Entomological Laboratory-Department of Biological Sciences and Plant Protection, High School of Horticulture, 4042Chott Mariem, Tunisia
Ben Halima Mounia
Entomological Laboratory-Department of Biological Sciences and Plant Protection, High School of Horticulture, 4042Chott Mariem, Tunisia
Ben Hamouda Habib
Entomological Laboratory-Department of Biological Sciences and Plant Protection, High School of Horticulture, 4042Chott Mariem, Tunisia