Research Article
Seasonal Changes in Gonadotropins, Prolactin and Sex Steroid Hormones in Captive Hill Mynah Gracula religiosa
Department of Biology, Faculty of Science, Ramkhamhaeng University, Bangkok 10240, Thailand
Hormones are critical factors for the activation of many aspects of reproductive behaviors in birds. Most studies about the roles of hormones in birds have focused on species that inhabited in temperate zones. A few studies have been carried out on tropical birds (Hau, 2001) which were in South America (Hau et al., 2004), India (Krishnaprasadan et al., 1988; Sailaja et al., 1988) and Australia (Wiley and Goldizen, 2003). Seasonal changes in photoperiod in temperate habitats are known to influence reproductive activities and the physiological mechanisms that regulate reproduction. In other words, most temperate birds rely on a high degree of photoperiodic stimuli for controlling reproduction. An increase in the photoperiod in spring switches on the secretion of gonadotropin-releasing hormones which then stimulate the secretion of hypophyseal hormones such as follicle-stimulating hormone and luteinizing hormone and in turn induce gonad growth and steroid hormone production (Murton and Westwood, 1977; Ball, 1993; Wingfield and Farner, 1993). Gonadal steroids regulate reproductive behaviors and physiological changes concerning reproduction (Balthazart, 1983; Wingfield and Ramenofsky, 1985; Archawaranon, 1987; Archawaranon and Wiley, 1988; Archawaranon et al., 1991; Wingfield et al., 1999). The reproductive period is usually ended by a mechanism called “photorefractoriness”? (Farner et al., 1983; Follett et al., 1985; Nicholls et al., 1988; Wilson and Donham, 1988; Bentley, 1997).
Seasonally controlled reproductive mechanisms in the tropical habitats where photoperiodic changes are minimal are rarely investigated. This study on reproductive endocrinology of Hill Mynah is, therefore, the first attempt to examine seasonal changes of plasma hormone levels in birds in Thailand.
Blood was drawn from the wing region of 30 male and 30 female (Archawaranon, 2004a) northern Hill Mynahs, Gracula religiosa intermedia, from October 1995 to September 1996. These adult birds were usually kept in eight outdoor aviaries, six to eight birds in each aviary, 4x5x3 m3 in dimension, at the Zoological Research Station, Ramkhamhaeng University, Bangna Campus, Bangkok, Thailand (Archawaranon, 2002, 2004b). Birds were captured from aviaries and kept overnight in an individual cage, 50x60x60 cm3 in dimension, for relaxation before blood drawing the next day. Blood was drawn in the morning during 09.00-12.00 h every two weeks for a year.
Plasma was employed to measure six hormone levels including follicle-stimulating hormone (FSH), luteinizing hormone (LH), testosterone, estradiol, progesterone and prolactin by Enzyme-linked Immunosorbent Assay (ELISA) (Archawaranon, 2005a). Seasonal changes of plasma hormone levels were compared using one-way ANOVA and Duncan`s New Multiple Range Test.
The hormone levels of these 60 birds which were kept in eight aviaries were also compared within the same sex with 10 couples kept separately in 10 breeding cages (Archawaranon, 2005b), 1x2x2 m3 in dimension. In order to examine whether social cues such as being with mates or being in flocks affected hormone levels, Student`s t-test was used to test the differences.
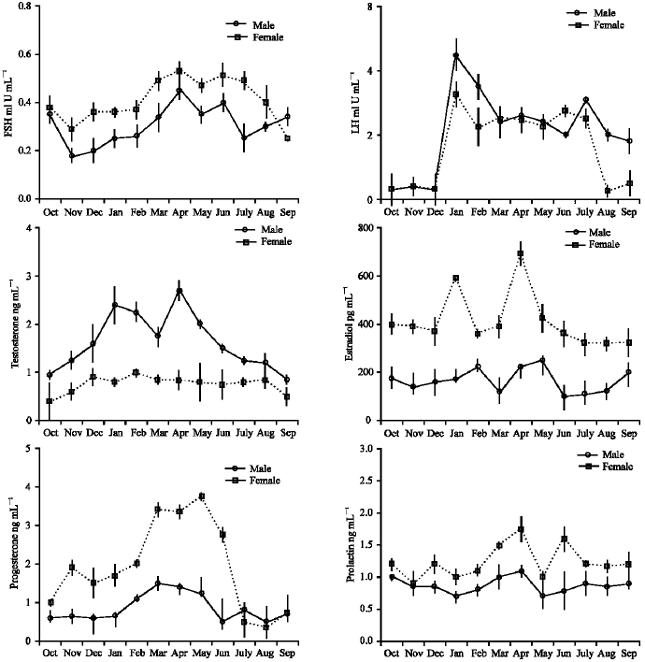
Seasonal changes in circulating gonadotropins, prolactin and sex steroid hormones: Hill Mynahs are regular seasonal breeders, like temperate birds. Both males and females showed regular seasonal changes in gonad sizes (Archawaranon, 1991) and reproductive hormones (Fig. 1). Generally the breeding season of Hill Mynah is January to July followed by the non-breeding season from August to December.
FSH levels in both males and females during the breeding season were significantly higher than those in
 | |
| Fig. 1: | Seasonal changes of hormone levels in captive Hill Mynah (M±SD) during 1995-1996 |
 | |
| Fig. 2: | The comparison of circulating hormone levels in birds kept with mates and birds kept in flocks (M = Males, F = Females) |
the non-breeding season (p<0.01). LH levels in both sexes were high in January and very low in October to December (p<0.01). Circulating levels of testosterone in male birds were significantly higher in the breeding season especially in January and April than in the non-breeding season (p<0.01). Circulating estradiol in females rose in January and April and was significantly higher than in the non-breeding season (p<0.01). But in males, the levels were high in February and May. Plasma levels of progesterone in females tended to be high in the breeding season especially in March, April and May than in the non-breeding season (p<0.01).
As for the plasma levels of prolactin, there was an increase during the breeding season (p<0.01) reaching a maximum in females in April and June.
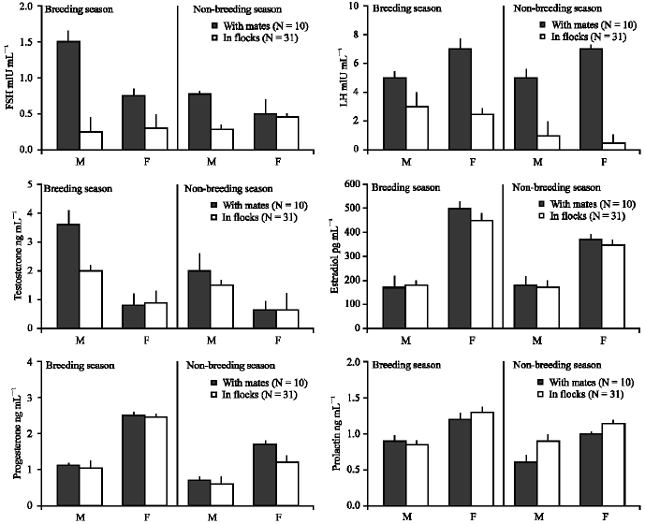
The influence of social cues on the reproductive endocrinology: During the breeding season, males which were kept with mates in the breeding cages increased plasma FSH to a higher level than those in flocks (t = 2.45, p<0.05). LH levels of males which were kept in pairs were significantly higher than the ones kept in flocks in the breeding season (t = 2.48, p<0.05) and in the non-breeding season (t = 2.32, p<0.05). Along the same line, plasma levels of LH in females with mates during the breeding season were higher than those in flocks (t = 3.84, p<0.0005) and during the non-breeding season (t = 5.67, p<0.001). Plasma testosterone levels paired males were significantly higher than in their counterparts kept in flocks (t = 3.74, p<0.005).
There were no significant differences in plasma levels of estradiol, progesterone and prolactin throughout the year between birds kept with mates and those kept in flocks (Fig. 2).
Hill Mynahs live in tropical regions where a small annual variation in photoperiod takes place. Seasonal breeding in this habitat is probably made possible by having high sensitivity to the slight seasonal changes in photoperiod (Hau, 2001). It is postulated that these birds can detect a slight photoperiodic increase and use it to initiate their reproductive activity. As a response to slight photoperiodic changes, these birds are physiologically prepared for the coming breeding season.
The breeding season of Hill Mynahs starts in January and continues into summer and the beginning of rainy season. In Thailand, most of the northern Hill Mynahs G. r. intermedia, breed during February to April while most of the southern birds G. r. religiosa breed during April to June (Archawaranon, 2003). The peak of breeding in this species is mostly during the summer time when ambient temperature is 35 °-40 °C and day length is approximately 12 h.
In both sexes, plasma levels of FSH rose in December, prior to the onset of breeding and remained elevated throughout the breeding season except in May. Circulating LH, testosterone and estradiol rose in January, the onset of breeding. Changes in circulating concentrations of estradiol correlated with FSH levels whereas elevated levels of progesterone varied with LH (Sakai and Ishii, 1986). The maximum levels of LH were accompanied by significant increases of circulating testosterone and were coincident with development of the gonads, secondary sex characters, periods of heightened territorial aggression and periods of heightened sexual behavior (Krishnaprasadan et al. 1988; Sailaja et al., 1988; Dittami and Reyer, 1984; Wingfield and Farner, 1993) Plasma levels of LH and estradiol seemed to be highest during the period of yolk formation, deposition and oviduct development (Donham, 1979; Schwabl et al., 1980; Dittami, 1981; Péczely and Pethes, 1982; Bluhm et al., 1983; Dawson, 1983; Rehder et al; 1986; Cockrem and Potter, 1991). Plasma levels of gonadotropins and sex steroids were parallel with gonadal development and reproductive behavior. Nevertheless, the temporal distribution of plasma testosterone and estradiol titers of male and female Hill Mynahs respectively were clearly bimodal with peaks in January and April. It indicated that the seasonal fluctuations of hormone levels corresponded exactly to the reproductive activity of Hill Mynahs, especially this species, as evidenced by the normally two clutches per breeding season (Archawaranon, 2003).
Previous studies show that plasma levels of progesterone in the female Canvasback Aythya valisinaria, are high at egg-laying and again in incubation period (Bluhm et al., 1983) whereas in other female birds such as European Starling Sternus vulgalis, Collared Dove Streptopelia decaocto and Rook Corvus fregilegus, progesterone levels are high only during egg-laying (Péczely and Pethes, 1979, 1982; Dawson, 1983) or in the development of the oviduct and in ovulation (Sharp, 1980). In the case of female Hill Mynahs, progesterone levels increase in March, April and May which are the period of egg-laying and incubation (Archawaranon, 2003).
There are distinct declines in plasma levels of FSH, LH and sex steroid hormones during the parental phase of the breeding cycle in many monogamous bird species (Wingfield and Farner, 1993). However, the prolactin has been implicated in the regulation of parental phase (Lehrman, 1965; Silver, 1978; Cheng, 1979; Scanes and Harvey, 1981, Cheng and Burke, 1983, Goldsmith, 1983, 1991; Lea and Sharp, 1989, 1991; Silverin and Goldsmith, 1990; Garcia et al., 1996; Schoech et al., 1998; Richard-Yris et al., 1998; Van Roo et al., 2003). In Hill Mynahs there is an increase in plasma levels of prolactin during the parental phase in April and June which are found in many monogamous species.
It is asserted that not only the hormones which regulate reproductive behaviors in Hill Mynahs but also social cues such as sexual partner or external stimuli such as eggs or nestlings that stimulate the next step in breeding activity. Although plasma levels of FSH, LH and testosterone in Hill Mynahs kept in couples were higher than in those kept in flocks, circulating estradiol, progesterone and prolactin in the former were not different from the latter. In sum, social cues such as sexual partner plays a role in the rise of FSH, LH and testosterone levels (Dufty and Wingfield, 1986; Shields et al., 1989; Silverin and Westin, 1995; Gwinner et al., 2002; Pinxten et al., 2003; Wiley and Goldizen, 2003). Physical contact with eggs stimulates incubation and that with chicks induces brooding behavior (Richard-Yris et al., 1998). In this study, Hill Mynah couples which were kept in breeding cages did not breed at the time under investigation, therefore, there was no eggs and nestlings in the nest. This probably explained why plasma levels of estradiol, progesterone and prolactin were the same in coupled birds and those in flocks.
This study gave the perspective of reproductive hormones in avian species in tropical regions. An examination of hormones in wild Hill Mynahs encompassing natural reproductive activities in the future will contribute more to our knowledge of avian reproductive endocrinology.
This research were supported by National Center for Genetics Engineering and Biotechnology, National Science and Technology Development Agency, Thailand and Ramkhamhaeng University.