Research Article
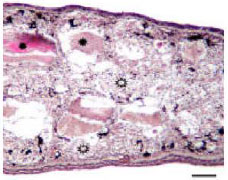
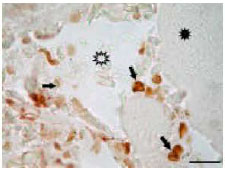
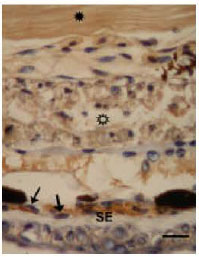
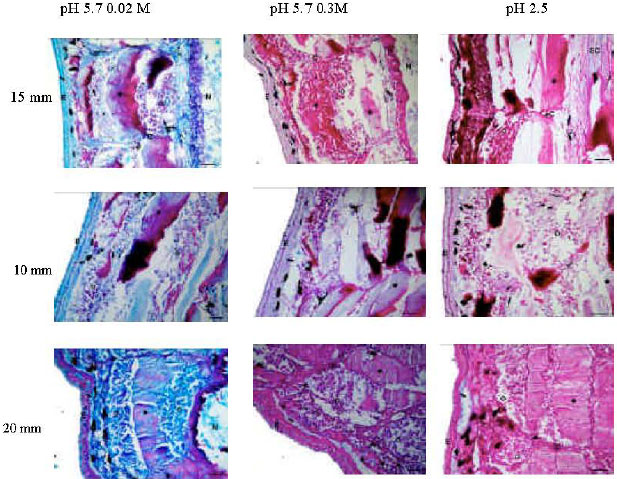
The Glycoconjugate Changes of Apoptotic Skeletal Muscle Tissues in Regressing Eurasian Green Toad, Bufo viridis (Amphibia: Anura) Tadpole Tail
Department of Biology, Molecular Biology Section, Faculty of Science and Art, Celal Bayar University, Muradiye Campus, 45047, Manisa, Turkey
Erdal Balcan
Department of Biology, Molecular Biology Section, Faculty of Science and Art, Celal Bayar University, Muradiye Campus, 45047, Manisa, Turkey