ABSTRACT
Growth and ionic concentrations of alfalfa (Medicago sativa L.) seedlings in response to interactive effects of different concentrations of Na+ (10, 50, 100 and 200 mol m-3) and Ca2+ (0, 5 and 10 mM) in growth media were studied. Dry weights of roots, stems and leaves were substantially declined with NaCl concentration increase, however, Ca2+ supplementation to the growth media alleviated the deteriorative effects of NaCl. Na+ and Cl¯ were significantly increased with increasing NaCl concentrations in all plant organs, but addition of Ca2+ had markedly reduced Na+ and Cl¯ content, particularly in roots. K+ was significantly reduced with increasing salinity level in roots and no clear trend was noticed in shoots. Ca2+ supplementation significantly increased K+ contents, particularly in roots. Ca2+ content was unchanged or slightly increased with increasing NaCl. However, K/Na and Ca/Na ratios were significantly decreased with increasing NaCl, but addition of Ca2+ had clearly altered the ratios to a positive increasing trend.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ja.2006.175.181
URL: https://scialert.net/abstract/?doi=ja.2006.175.181
INTRODUCTION
More than 30% of the irrigated land world wide is affected by salinity (Carter, 1975) and new areas are increasingly affected in many regions of the world (Chauhan, 1987). Increasing demands for quality water for both industrial development and population is likely to entail the use of low quality, brackish water (Epstein and Rains, 1987). High salt concentration in root medium affects the growth and economic yield of many important crops (Maas and Hoffman, I977). Glycophytes, in order to overcome high salt concentration must be able to withstand potential water deficit, ion toxicity and nutrient imbalances (Greenway and Munns, 1980).
Alfalfa is moderately sensitive to salinity (Maas and Hoffman, I977) and is considered as the most important forage crop in the Kingdom of Saudi Arabia. Ion toxicity was usually associated with either excessive Cl¯ or Na+ intake (Groham et al., 1985; Munns and Termaat, 1986). Addition of Ca2+ has been reported to ameliorate the adverse effects of salinity on root elongation of plants (Ashraf and Naqvi, 1991; Cramer et al., 1989; Nakamura et al., 1990; Zidan et al., 1990) and shoot growth (Ashraf and Naqvi, 1991; Cramer et al., 1989; Zidan et al., 1990; Kent and Lauchli, 1985; Grieve and Fujiyama, 1987; Grieve and Mass, 1988; Subbarro et al., I990; Yeo et al., I991). Elevated Ca2+ levels in NaCl medium inhibited Na+ binding to cell walls (Stassart et al., 1981) and the plasma membrane (Cramer et al., 1985; Lynch and Cramer, 1987) and hence reduced leakiness of membranes (Cramer et al., 1985; Van Steveninck, 1985; Picchioni et al., 1991) however, Lynch and Lauchli (1985) did not observe this in salt stressed corn root protoplasts. Ca2+ also increased the uptake and transport of K+ (Nakamura et al., 1990; Cramer et al., 1985) and reduced Na+ accumulation in plants (Subbarro et al., I990; Cramer et al., 1987; Ehret et al., 1990; Zidan et al., 1991). The growth stimulation effect of Ca+ addition under saline conditions was attributed not only to the function of membranes, but also to cell elongation and cell division (Zidan et al., 1990; Kurth et al., 1986).
The present investigation was aimed to study the interactive effects of NaCl salinity and Ca/Na ratio on growth, ion uptake and translocation of alfalfa seedlings.
MATERIALS AND METHODS
Seeds of alfalfa (Medicago sativa L.) were sterilized with 0.5% sodium hypochlorite solution for one minute and then washed twice with distilled water. Seeds were germinated in petri-dishes with filter paper Watman No. 1. Three days after germination, seedlings developed their first pair of leaves. At this stage, five seedlings were transferred to several 100 mL conical flasks filled with 25 cm3 L-1. Twenty milli Liter of NaCl were pipetted into the conical flasks, which were then sealed with polyethylene sheets. The flasks were incubated in a programmed refrigerated incubation using 16 h light: 8 h dark at 25 to 15°C day: night. Seedlings were grown at combined concentrations of 10, 50, 100 and 200 mM NaCl and 0, 5 and 10 mM CaCl2. The twelve combined salt treatments were added to one-tenth strength modified Hoagland’s solution. The concentrations of Na+, K+ and Ca2+ were 0.005, 0.6 and 0.4 mM, respectively. A completely randomized design with factorial combinations of 4 levels of NaCl and 3 levels of CaCl2 replicated six times was used to conduct the experiment. Plant were harvested 21 days after treatment application. Plants were separated into leaves, stems and root for the determination of fresh and dry weights. Stem and leaves of alfalfa seedings were washed two times in distilled water while ions were removed from the free space of roots by washing 2 min in sorbitol solutions isotonic with the treatments concentration in which the plants had grown. Leaves, stems and roots were dried at 85°C for 48 h to determine their dry weights.
For the analysis of K+, Na+, Ca2+ and Cl¯, three samples each of ≈ 200 mg of fresh material of roots, stems and leaves were homogenized using a pestle and mortar and extracted in 25 mL of deionized water at 90°C for 4 h. The Na+, K+ and Ca2+ were determined with a GBS 905 Atomic Absorption Spectrophotometer. Cl¯ was determined using a chloride meter (Jenway, PCLLM3).
The data were statistically analyzed according to the technique of analysis of variance (ANOVA) for the factorial experiment in the completely randomized design as published by Gomez and Gomez (1984). The treatment means were compared using Baysian Least Significant Difference (BLSD) at 5% level of probability (Waller and Duncan, 1969). All statistical analysis were performed using the facility of computer and SAS software package (SAS Institute, 1996).
RESULTS AND DISCUSSION
Increasing NaCl concentration in the rooting media significantly reduced dry weights of root, stem and leaves (Fig. 1). This is consistent with the results obtained by other researchers (Chow et al., 1990; Bar-Tal et al., 1991; Khan et al., 1998; Kinraide, 1999; Al-Khateeb, 2005a). Adverse effects of increasing NaCl concentration were more pronounced on leaves than on stem and root, indicating that root growth was less affected by salinity (Greenway and Munns, 1980; Delane et al., 1982).
Ca2+ supplementation in the rooting media particularly at the highest NaCl concentration, significantly increased dry weight of root, stem and leaves. This is consistent with the results obtained by other researchers (Ashraf and Naqvi, 1991; Kent and Lauchli, 1985; Reid and Smith, 2000; Bonilla et al., 2004). This may indicate that under NaCl salinity Ca2+ supplementation decreases the inhibitory effects of salinity in stressed plant (Khavari-Nejad, 1988).
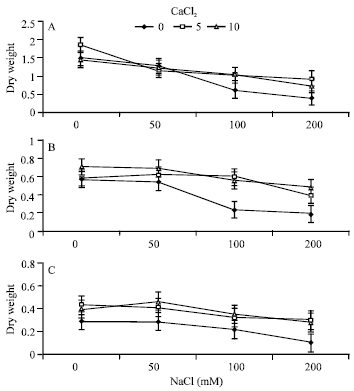 | |
| Fig. 1: | Effects of Ca2+ supplementation (mM) on dry weights of leaves (A), stem (B) and root (C) (mg/plant) of alfalfa seedlings grown under saline conditions (Bars = BLSD 0.05) |
Supplemental Ca2+ alleviates deleterious salt effects probably through mitigating the toxic effects of Na+ ions (Bliss et al., 1986; Qadir et al., 2002). However, no significant differences were detected between 5 and 10 mM CaCl2. Insignificant differences may be attributed to the ability of alfalfa seedlings to show almost constant growth pattern when Ca2+ is abundant to plants.
The Na+ content increased significantly in root, stem and leaves as NaCl concentration increased (Fig. 2). Ca+2 supply significantly decreased Na+ accumulation in root and leaves, particularly under high salinity levels. However, no significant differences were detected between 5 and 10 mM CaCl2. The reduction in Na+ concentration with the addition of Ca2+ was much pronounced on root.
The reduction in the accumulation of Na+ by alfalfa seedlings with the addition of Ca2+ supports the findings of others (Ashraf and Naqvi, 1991; Subbarro et al., I990; Cramer et al., 1987; Zidan et al., 1991; Muhammad et al., 1987; Rengel, 1992) who observed a decrease in Na+ uptake with Ca2+ supplementation.
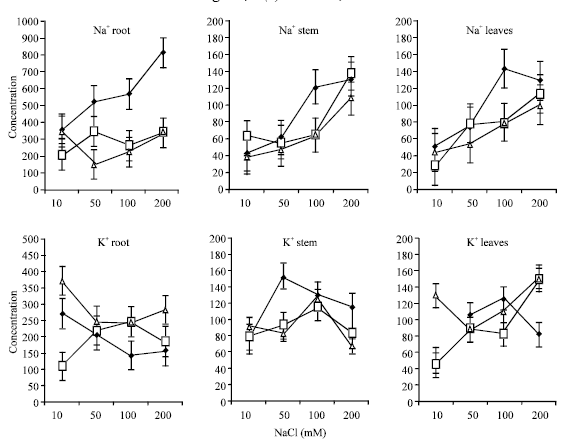 | |
| Fig. 2: | Effects of Ca2+ supplementation (mM) on Na+ and K+ concentrations (μM) in root, stem and leaves of alfalfa seedlings grown under saline conditions (Bars = BLSD 0.05) |
The reduction in Na+accumulation may be associated with the decrease of membrane permeability due to the reduction of Na+ binding to cell walls and the plasma membrane which alleviates membrane leakage (Cramer et al., 1985; Picchioni et al., 1991; Rengel, 1992). Na+concentration was higher in root compared to stem and leaves. Translocation of Na+from root to stem was most probable with Ca2+ supplementation while a mechanism that retains Na+in root might be operating under absence of Ca2+.
Supplemental Ca2+ at 200 mM NaCl had no significant effect in decreasing Na+in stem and to some extend in leaves. It is quite probable that the role of Ca2+ in maintenance of plasma membrane integrity and functioning are much disturbed under high level of salinity.
Root K+ content under no Ca2+ addition was decreased significantly as NaCl concentration increased, while with Ca2+ supplementation K+ concentration was significantly increased, particularly under high Ca2+ concentrations. Stem K+ contents seems to be less affected by high level NaCl in root media, since K+ contents was highest under Ca2+ deficient. In leaves, K+ content was also higher with no Ca2+ supplementation except under 200 mM where Ca2+ addition increased K+ contents (Fig. 2). Moreover, addition of 10 mol m-3 of Ca2+ significantly increased K+ concentration under the lowest NaCl concentration, however, a trend of increasing K+ concentration under Ca2+ supplementation with increasing NaCl concentration was clearly evident.
The lower internal K+ concentration with an increase in external Na+concentration in the absence of Ca2+ supply is normally expected (Esechie and Rodrignez, 1998). This could be attributed to the competition of Na+with the uptake of K+ (Salisbury and Ross, 1994).
There was higher K+ accumulation in root while leaves had generally the lowest concentration. Wolf and Jeschke (1987) reported that in shoot of salt stressed barley there was recirculation of K+ via phloem from shoot to root. Part of this recirculated K+ was transferred from phloem to xylem within the root and returned to the shoot via xylem. Similar results were reported in Lupinus albus (Jeschke et al., 1986). The lower K+ concentration with Ca2+ addition in stem and leaves may be attributed to the role of Ca2+ in decreasing membrane selectivity which may generate a selective ion transport mechanism similar to that reported for Na+. Similar results were also reported by Groham et al. (1985), while contradicting results were obtained by Kawasaki and Moritsuger (1979) and Ashraf and Naqvi (1991).
Interestingly, Ca2+ concentration in roots under Ca2+ deficient remained almost unchanged with increasing NaCl concentration, while its concentration at 50 and 100 mM NaCl was significantly lower with 10 mM CaCl2 (Fig. 3).
Stem Ca2+ concentration was generally higher with Ca2+ supplementation. This trend was distinctly obvious at the lowest two NaCl concentrations.
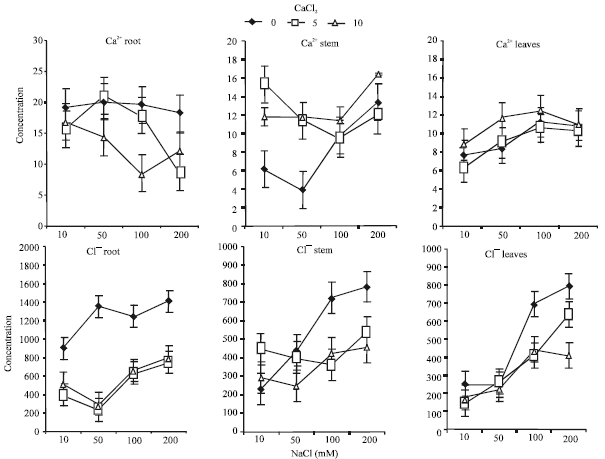 | |
| Fig. 3: | Effects of Ca2+ supplementation (mM) on Ca2+ and Cl¯ concentrations (μM) in root, stem and leaves of alfalfa seedlings grown under saline conditions (Bars = BLSD 0.05) |
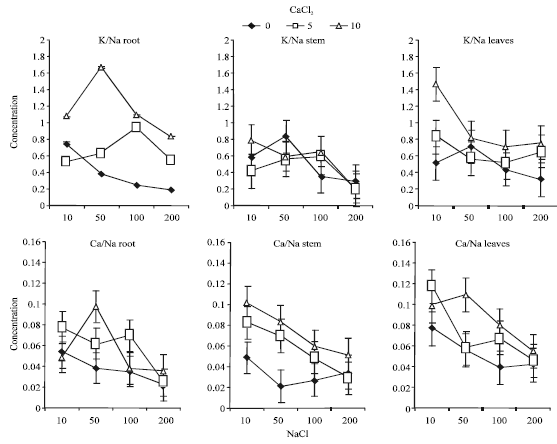 | |
| Fig. 4: | Effects of Ca2+ supplementation (mM) on K/Na and Ca/Na ratio in root, stem and leaves of alfalfa seedlings grown under saline conditions (Bars = BLSD 0.05) |
Under Ca2+ supplementation, Ca2+ concentration was a slightly changed with increasing NaCl concentration in the rooting media.
The concentration of Ca2+ in leaves was gradually increased with increasing NaCl concentration. However, the increase was only significant under 50 mM NaCl and 10 mM CaCl2.
Lower Ca2+ concentrations in plant tissues with increasing salinity levels have been reported in barley (Lynch and Lauchli 1985; Cramer et al., 1990; Lynch et al., 1988), Cotton (Cramer et al., 1987) and Alfalfa (Al-Khateeb, 2005b), but this was not true in Spergularia sp. (Olubukanla et al., 1984). Lower Ca2+ concentration particularly in roots even with Ca2+ supplementation could be attributed to the role of Na+in reducing adsorption of Ca2+ to root cell walls in competition for the same binding sites (Stassart et al., 1981). However, the role of Ca2+ in plant under saline conditions had been widely reported to be related to both membrane permeability which might reduce Na+accumulation in plant tissue as well as increasing the uptake and transport of K+ which was clearly evident in the present study. It is worth mentioning that the slight change in concentration of Ca2+ in leaves with Ca2+ addition could be attributed to the reduction of Ca2+ transport to the shoot particularly under the highest NaCl concentration (Lynch and Lauchli 1985; Wolf et al., 1991). However, a mechanism that retained Ca2+ in stem is highly operative under these circumstances. Davenport et al. (I997) reported that in salt sensitive wheat cultivars, Na+ significantly inhibited Ca2+ translocation, reflecting probably differences in Ca2+ transport processes, which may be partially due to greater salt sensitivity of alfalfa seedings.
Cl¯ concentration in roots was significantly higher as NaCl concentration increased. Supplementation of Ca2+ significantly reduced Cl¯ uptake even under the highest NaCl concentrations. Similar trend was also reported in stem, except under the lowest NaCl concentration where Ca2+ addition increased Cl¯ concentration. Concentration of Cl¯ in Stem was much lower than those observed in roots. In leaves, Cl¯ concentration was higher under the highest NaCl concentration, while Ca2+ supplementation reduced Cl¯ concentration in leaf tissues of alfalfa seedlings (Fig. 3).
The higher Cl¯ concentration in plant organs with increasing NaCl concentration is normally expected (Esechie and Rodrignez, 1998; Al-Khateeb, 2005b) Olubukanla et al. (1984) reported that increasing both salinity and Ca2+ concentration had positively affected Cl¯ absorption, while this phenomenon was not observed by Ashraf and Oleary (1997). Cl¯ seems to have a significant role in decreasing membrane selectivity that may generate a selective ion transport mechanism similar to that reported for Na+and K+. Moreover, Cl¯ translocation from root to shoot seems to be much inhibited with Ca2+ supplementation. It is much likely that when external Cl¯ concentration are high, the tonoplast rather than plasmalemma becomes a barrier to Cl¯ absorption (Cram, 1973) which may explain the higher Cl¯ concentration in the present study with increasing Cl¯ concentration in the media.
K/Na ratio was significantly decreased as NaCl concentration increased in all plant organs. Increasing Ca2+ concentration in the media showed significant increase in K/Na ratio in root and lower increase in leaves. Roots were able to accumulate one fold higher Na+and K+ concentration, compared to that of stem and leaves (Fig. 4). It was clearly shown that alfalfa seedlings were able to maintain relatively constant K/Na ratio between 0.2 to 1.4 for all plant organs with the lower values in stems. It seems that the ability to maintain higher K/Na ratio particularly in roots are much related to salt tolerance.
Ca/Na ratio decreased significantly with increasing NaCl concentration in all plant organs. Ca2+ supplementation significantly increased Ca/Na ratio, particularly under the lowest NaCl concentration (Fig. 4). Alfalfa seedlings maintained higher Ca/Na ratio in all plant organs, but not under 200 mM NaCl salinity. This trend may indicate that alfalfa seedlings can readly tolerate higher NaCl concentration less than 200 mM if sufficient Ca2+ concentration is available for plant. K+ and Ca2+ concentrations in alfalfa seedling organs seem to be much related to their K/Na and Ca/Na ratios, respectively, rather than their concentrations in plant organs or media. However Ca2+ appeared to play a significant role in mitigating the deleterious effects of salinity (i.e., Na+and ClG) probably by controlling Na+, K+ and Cl- absorption through a generating ion selective mechanism.
REFERENCES
- Ashraf, M. and J.W. Oleary, 1997. Response of salt-tolerant and a salt- sensitive line of sunflower to varying sodium/calcium ratios in saline sand culture. J. Plant Nutr., 20: 361-377.
Direct Link - Bar-Tal, A., S. Feigenbaum and D.L. Sparks, 1991. Potassium-salinity interactions in irrigated com. Irrig. Sci., 12: 27-35.
CrossRefDirect Link - Bonilla, I., A. El-Hamdaoui and L. Bolanos, 2004. Boron and calcium increase Pisum sativum seed germination and seedling development under salt stress. Plant Soil, 267: 97-107.
CrossRefDirect Link - Chow, W.S., M.C. Ball and J.M. Anderson, 1990. Growth and photosynthetic responses of spinach to salinity: Implications of K+ nutrition for salt tolerance. Aust. J. Plant Physiol., 17: 563-578.
CrossRefDirect Link - Cramer, G.R., A. Lauchi and V.S. Polito, 1985. Displacement of Ca2+ by Na+from the plasmalemma of root cells. A primary response of salt stress. Plant Physiol., 79: 207-211.
PubMedDirect Link - Cramer, G.R., J. Lynch, A. Liiuchii and E. Epstein, 1987. Influx of Na+, K+ and Ca2+ into roots of salt-stressed cotton seedlings: Effects of supplemental Ca2+. Plant Physiol., 83: 510-516.
Direct Link - Cramer, G.R., R. Abdel Basset and J.R. Seeman, 1990. Salinity-calcium interactions on root growth and osmotic adjustment of two corn cultivars differing in salt tolerance. J. Plant Nutr., 13: 1453-1462.
Direct Link - Davenport, R.J., R.J. Reid, F.A. Smith and C.H. Bornman, I997. Sodium-Calcium interactions in two wheat species differing in salinity tolerance. Physiol. Plant., 99: 323-327.
Direct Link - Delane, R., H. Greenway, R. Munnus and J. Gibbs, 1982. Ion concentration and carbohydrate status of the elongating leaf tissue of Hordeum vulgare growing at high external NaCl. 1. Relationship between solute concentration and growth J. Exp. Bot., 33: 557-573.
Direct Link - Ehret, D.L., R.E. Redmann, B.L. Harvey and A. Cipywnyk, 1990. Salinity-induced calcium deficiencies in wheat and barley. Plant Soil, 128: 143-151.
CrossRefDirect Link - Epstein, E. and D.W. Rains, 1987. Advances in salt tolerance. Plant Soil, 99: 17-29.
CrossRefDirect Link - Esechie, H.A. and V. Rodrignez, 1998. Ion compartmentation in salinity-stressed alfalfa seedlings growing under different temperature regimes. Commun. Soil Sci. Plant Anal., 29: 2607-2618.
Direct Link - Gomez, K.A. and A.A. Gomez, 1984. Statistical Procedures for Agricultural Research. 2nd Edn., John Wiley and Sons Inc., Hoboken, New Jersey, ISBN: 978-0-471-87092-0, Pages: 704.
Direct Link - Grieve, C.M. and H. Fujiyama, 1987. The response of two rice cultivars to external Na/Ca ratio. Plant Soil, 103: 245-250.
CrossRefDirect Link - Grieve, C.M. and E.V. Maas, 1988. Differential effects of sodium/calcium ratio on Sorghum genotypes. Crop Sci., 28: 659-665.
CrossRefDirect Link - Gorham, J., R.G.W. Jones and E. McDonnell, 1985. Some mechanisms of salt tolerance in crop plants. Plant Soil, 89: 15-40.
CrossRefDirect Link - Kent, L.M. and A. Lauchli, 1985. Germination and seedling growth of cotton: Salinity-calcium interactions. Plant Cell Environ., 8: 155-159.
CrossRefDirect Link - Khan, M.G., M. Silberbush and S.H. Lips, 1998. Response of Alfalfa to potassium, calcium and nitrogen under stress induced by sodium chloride. Biol. Plant, 40: 251-259.
CrossRef - Kinraide, T., 1999. Interactions among Ca2+, Na+and K+ in salinity toxicity: Quantitative resolution of multiple toxic and ameliorative effects. J. Exp. Bot., 50: 1495-1505.
Direct Link - Kurth, E., G.R. Cramer, A. Lauchli and E. Epstein, 1986. Effects of NaCl and CaCl2 on cell enlargement and cell production in cotton roots. Plant Physiol., 82: 1102-1106.
CrossRef - Lynch, J. and A. Lauchli, 1985. Salt stress disturbs the calcium nutrition of barley (Hordeum vulgare L.). New Phytol., 99: 345-354.
Direct Link - Lynch, J., G.R. Cramer and A. Lauchli, 1987. Salinity reduces membrane-associated calcium on corn root protoplasts. Plant Physiol., 83: 390-394.
PubMedDirect Link - Maas, E.V. and G.J. Hoffman, 1977. Crop salt tolerance-current assessment. J. Irrig. Drainage Div., 103: 115-134.
Direct Link - Muhammad, S., M. Akbar and U.H. Neue, 1987. Effect of Na/Ca and Na/K ratios in saline culture solution on the growth and mineral nutrition of rice (Oryza sativa L.). Plant Soil, 104: 57-62.
CrossRefDirect Link - Munns, R. and A. Termaat, 1986. Whole-plant responses to salinity. Aust. J. Plant Physiol., 13: 143-160.
Direct Link - Nakamura, Y., K. Tanak, E. Ohta and M. Sakata, 1990. Protective effects of external Ca2+ on elongation and intracellular concentration of K+ in intact mung bean roots under high NaCl stress. Plant Cell Physiol., 31: 815-821.
Direct Link - Picchioni, G.A., S.M.T. Arnoto and J.B. Storey, 1991. Rapid testing of salinity effects on Pistachio seedling rootstocks. J. Am. Soc. Hortic. Sci., 6: 555-559.
Direct Link - Qadir, M., R.H. Qureshi and N. Ahmad, 2002. Amelioration of calcareous saline sodic soils through phytoremediation and chemical strategies. Soil Use Manage., 18: 381-385.
Direct Link - Reid, R.J. and F.A. Smith, 2000. The limits of sodium/calcium interactions in plant growth. Aust. J. Plant Physiol., 27: 709-715.
Direct Link - Rengel, Z., 1992. The role of calcium in salt toxicity. Plant Cell Environ., 15: 625-632.
CrossRefDirect Link - Salisburry, F.B. and C.W. Ross, 1991. Plant Physiology. 4th Edn., Wadsworth Publishing Company, Belmont, California.
Direct Link - Wolf, O., R. Munns, M.L. Tonnet and W.D. Jeschke, 1991. The role of the stem in the partitioning of Na+ and K+ in salt-treated barley J. Exp. Bot., 42: 697-704.
Direct Link - Yeo, A.R., A.S. Lee, P. Izard, P.J. Boursier and T.J. Flowers, I991. Short- and long-term effects of salinity on leaf growth in rice (Oryza sativa L.). J. Exp. Bot., 42: 881-889.
CrossRefDirect Link - Zidan, I., H. Azaizeh and P.M. Neumann, 1990. Does salinity reduce growth in maize root epidermal cells by inhibiting their capacity for cell wall acidification?. Plant Physiol., 93: 7-11.
Direct Link - Zidan, I., B. Jacoby, I. Ravina and P.M. Neumann, 1991. Sodium does not compete with calcium in saturating plasma membrane sites regulating 2 Na+ influx in salinized maize roots. Plant Physiol., 96: 331-334.
Direct Link - Waller, R.A. and D.B. Duncan, 1969. A bayes rule for the symmetric multiple comparisons problem. J. Am. Stat. Assoc., 64: 1484-1503.
CrossRefDirect Link - Bliss, R.D., K.A. Platt-Aloia and W.W. Thomson, 1986. Osmotic sensitivity in relation to salt sensitivity in germinating barley seeds. Plant Cell Environ., 9: 721-725.
CrossRef








