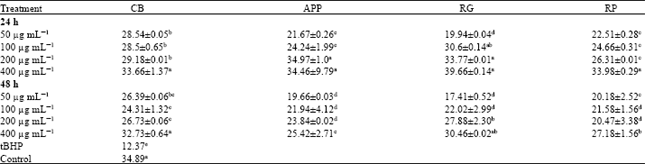
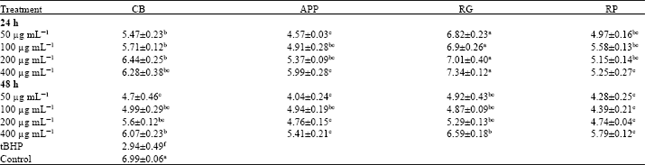
Research Article
Protective Effects of the Phenolic Extracts of Fruits against Oxidative Stress in Human Lung Cells
Nutritional Biochemistry and Carcinogenesis laboratory, Alabama A and M University, Normal, AL, USA
M. Verghese
Nutritional Biochemistry and Carcinogenesis laboratory, Alabama A and M University, Normal, AL, USA