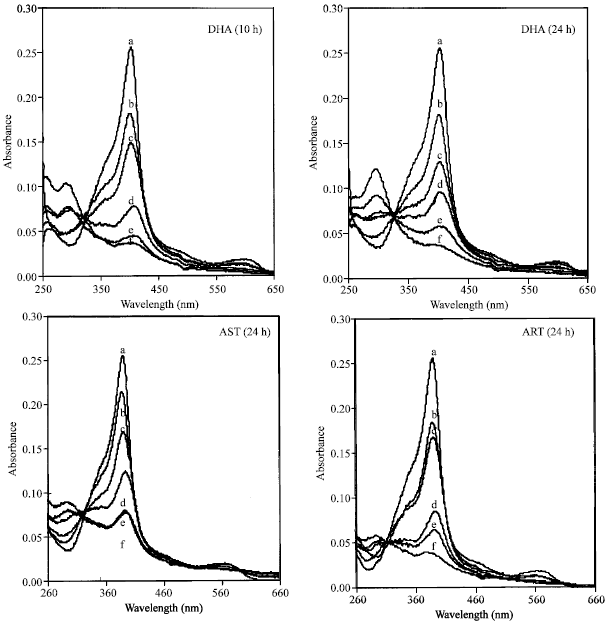
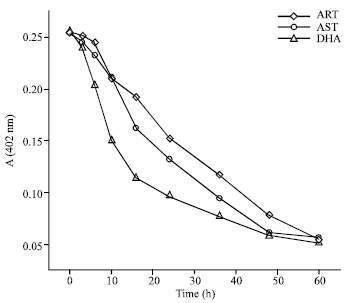
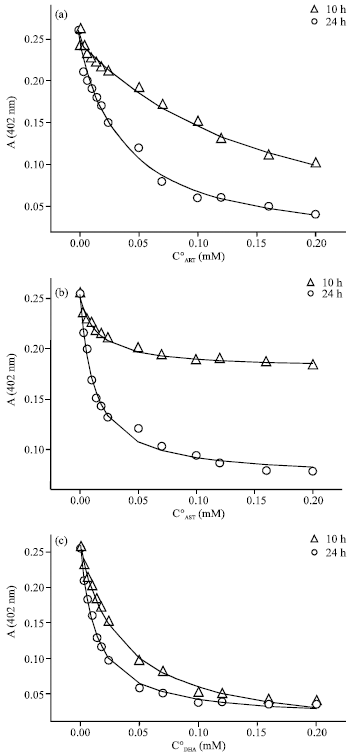
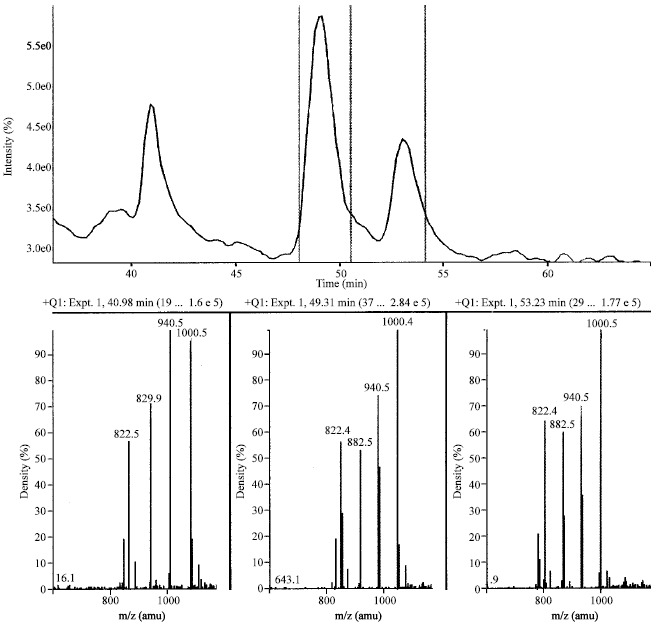
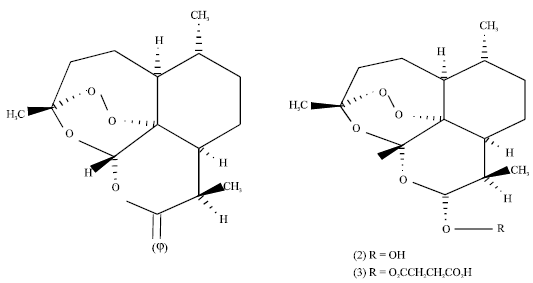
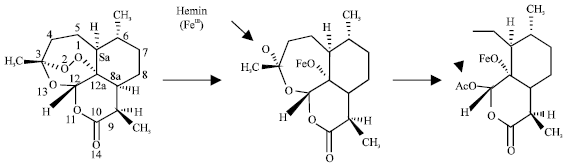
Case Study
Interaction of Artemisinin Based Antimalarial Drugs with Hemin in Water-DMSO Mixture
Department of Chemistry, Biomacromolecules Laboratory, Kinshasa University, BP 190, Kinshasa, RI Congo
B. Mavakala
Department of Chemistry, Biomacromolecules Laboratory, Kinshasa University, BP 190, Kinshasa, RI Congo
Yu Wu
Department of Chemistry, Tsinghua University, Beijing 100084, Peoples Republic of China