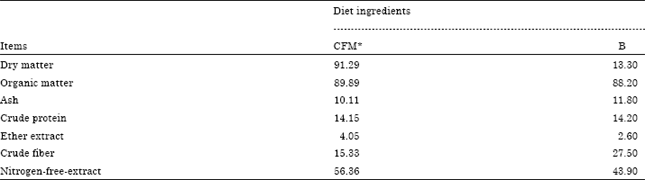
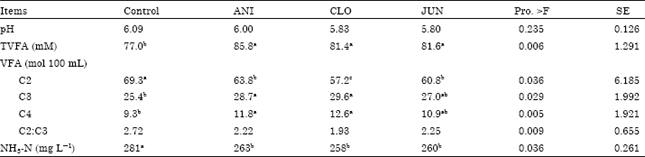
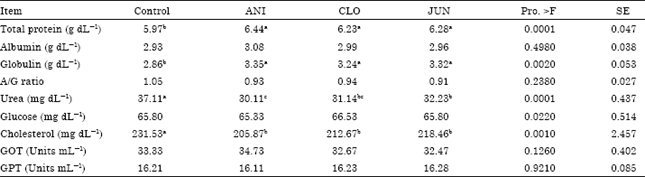
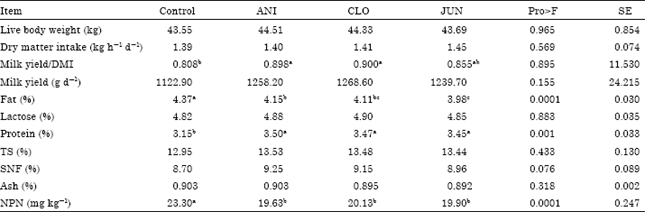
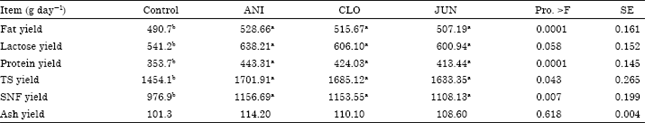
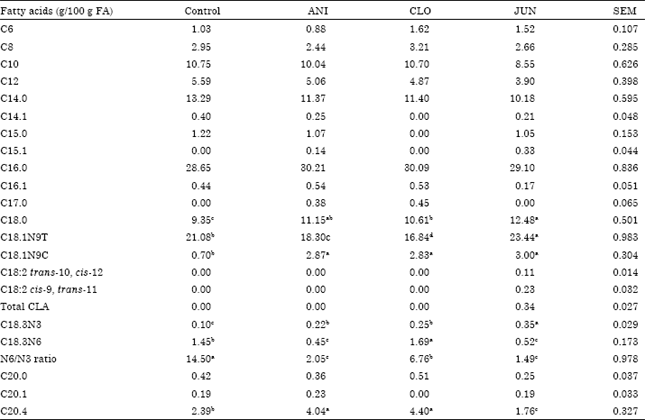
Research Article
Impact of Anise, Clove and Juniper Oils as Feed Additives on the Productive Performance of Lactating Goats
Department of Dairy Science, National Research Center, Dokki, Cairo, Egypt
S.M. Kholif
Department of Dairy Science, National Research Center, Dokki, Cairo, Egypt
O.H. Matloup
Department of Dairy Science, National Research Center, Dokki, Cairo, Egypt
M.M. Abdo
Department of Dairy Science, National Research Center, Dokki, Cairo, Egypt
M.H. El-Shafie
Animal Production Research Institute, Agriculture Research Center, Cairo, Egypt