Research Article
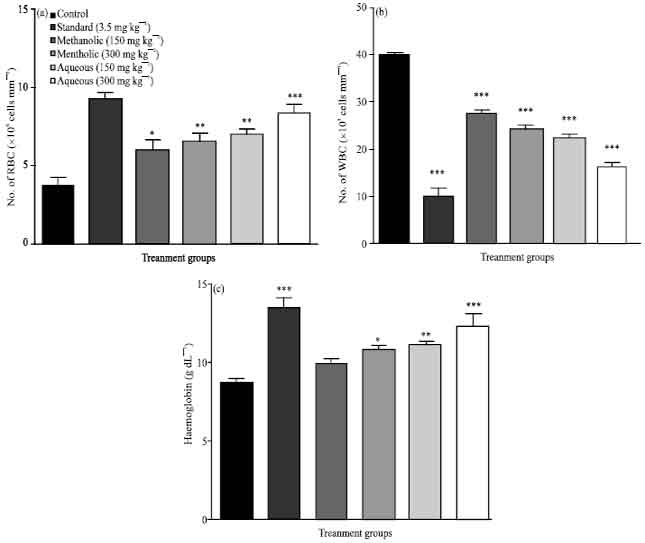
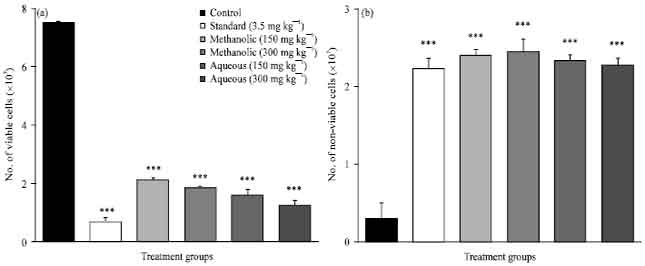
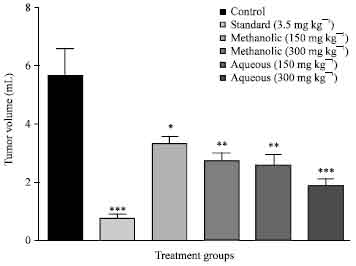
Evaluation of Anticancer Activity of Macrosolen parasiticus (L.) Danser on Ehrlich's Ascites Carcinoma Treated Mice
Department of Pharmacognosy, Manipal College of Pharmaceutical Sciences, Manipal University, Manipal-576 104, Karnataka, India
N. Dashora
Department of Pharmacognosy, Manipal College of Pharmaceutical Sciences, Manipal University, Manipal-576 104, Karnataka, India
K.S. Prabhu
Department of Pharmacognosy, Manipal College of Pharmaceutical Sciences, Manipal University, Manipal-576 104, Karnataka, India
R. Lobo
Department of Pharmacognosy, Manipal College of Pharmaceutical Sciences, Manipal University, Manipal-576 104, Karnataka, India