Research Article
Efficacy of Botanical Extracts against Termites, Macrotermes spp., (Isoptera: Termitidae) under Laboratory Conditions
College of Agriculture and Veterinary Medicine, Jimma University, P.O. Box 307, Jimma, Ethiopia
D. Mohamed
Ethiopian Institute of Agricultural Research, Ambo Plant Protection Research Center, P.O. Box 2003, Addis Ababa, Ethiopia
S. Waktole
College of Agriculture and Veterinary Medicine, Jimma University, P.O. Box 307, Jimma, Ethiopia









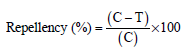
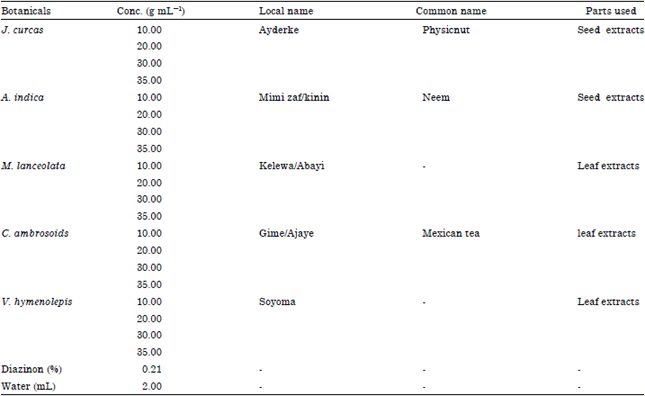
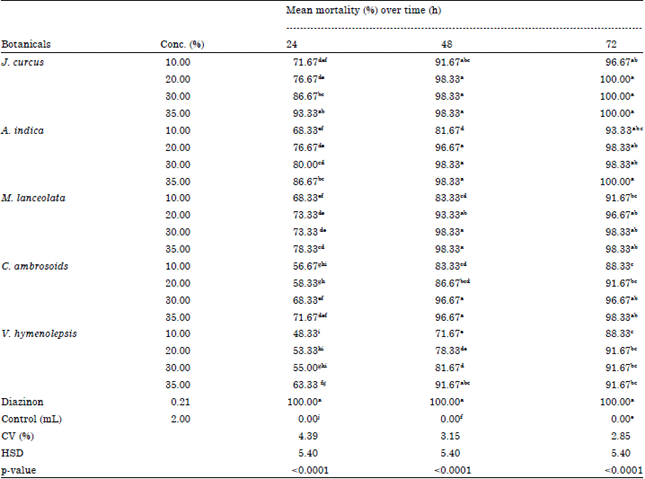
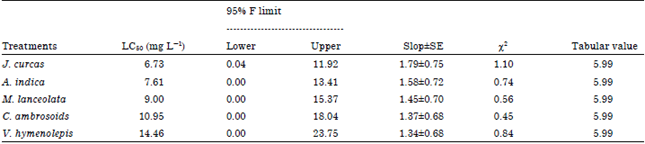

Dr. G. Singh Reply
Please need this for study purpose please kindly mail.
with regards
Dr. G. Singh
Assistant Entomologist