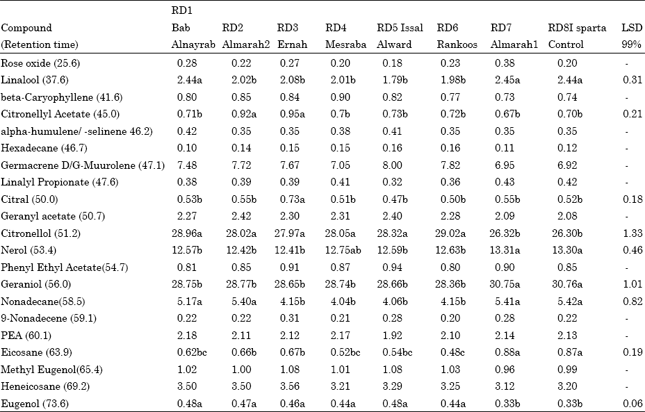
Research Article
Genetic Diversity and Qualitative Variation of Rosa damascena in Syria
Department of Horticultural Science, Faculty of Agriculture, Damascus University, P.O. Box 30621, Damascus, Syria
N. Albatal
Department of Horticultural Science, Faculty of Agriculture, Damascus University, P.O. Box 30621, Damascus, Syria
H. Baydar
Department of Horticultural Science, Faculty of Agriculture, Suleyman Demirel University, P.O. Box 42737, Isparta, Turkey
K. Almaarri
Department of Horticultural Science, Faculty of Agriculture, Damascus University, P.O. Box 30621, Damascus, Syria