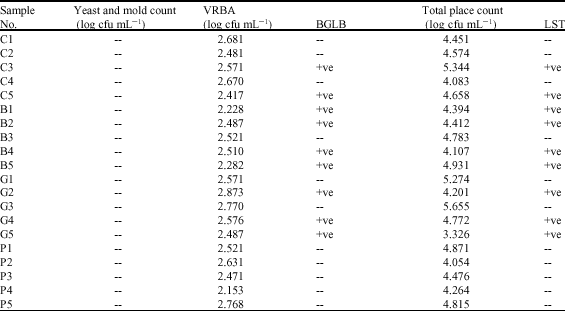
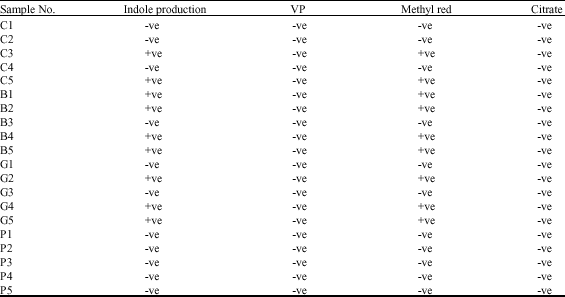
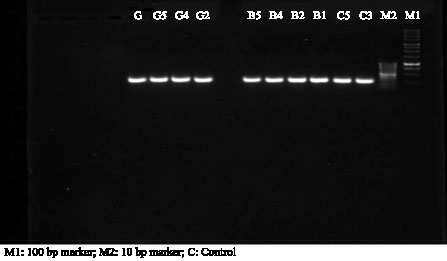
Research Article
Molecular and Bacteriological Examination of Milk from Different Milch Animals with Special Reference to Coliforms
Laboratory of Food Biotechnology, Department of Home Science, Sardar Patel University, Vallabh Vidyanagar-388120, India
R. Subhash
Laboratory of Food Biotechnology, Department of Home Science, Sardar Patel University, Vallabh Vidyanagar-388120, India