Review Article
Eco-friendly Surface Modifications of Wool Fiber for its Improved Functionality: An Overview
National Institute of Research on Jute and Allied Fiber Technology, 12, Regent Park, Kolkatta-700040, India
Textile industry uses substantial quantities of wool fibers in terms of textile and technical products. The conversion of fiber into fabric requires technological intervention for development of different kind and variety of products, while textile processing will used to enhance the value and functionality of textile product. Textile processing other than dyeing and finishing, preparatory or pre-treatment process have been used in the textile processing conventionally or specifically which depends on the end-uses of the products. For example bleaching needs for light shade woolen yarn, carbonizing requires for burr-free products, enzyme treatment requires for softness (Rippon, 1992, 2003). Pretreatment can also introduces in a textile processing either to save energy or to reduce the amount of chemicals to be used in the subsequent processing. Wool, an animal based natural fiber has distinct properties like warmness, reactivity, moisture regain, elasticity and flame resistant however, it has demerits like felting shrinkage, hydrophobicity and prone to moth attack due to presence of surface cuticle and cysteine based amino acids (Von Bergen, 1963; Ammayappan and Moses, 2006).
Initially researchers focused mainly on development of anti-shrinkage process for wool based textiles. They had developed so many processes like chlorination, sulfitolysis, oxidation and other pretreatments followed by polymer finishing (Ammayappan and Moses, 2009). However, there are so many processes developed, researchers have emphasis mainly on eco-friendly surface modifications with improved functionality of wool fiber. The main objective of this study is to review the importance of surface modifications for value addition of wool products with special reference to their morphological nature; important surface modification processes applicable to wool fiber and futuristic area of research on wool.
WOOL FIBER
Wool fiber is obtained from follicles of sheep, goat, camel, rabbit and camelids like vicuna, llama and alpaca as wool fleece and so it is an important animal hair. It is composed of eighteen different amino acids in which the importance amino acids are cysteine (13.1%), glutamate (11.1%) and serine (10.8%). Wool fiber, viewed under the scanning electron microscope is shown in Fig. 1 (Simpson and Crawshaw, 2005).
Grease wool fleece contains impurities like wool grease, perspiration products i.e. suint, adhered materials like dirt and vegetable matter (Garner, 1967; Glaser, 1996). Before spinning, these impurities are removed by wet processing methods like scouring and/or carbonizing (Lewis, 1992; Banerjee et al., 2009). Since, it is made from polypeptide chains (97%) and lipids (1%) with a heterogeneous morphological structure, it is considered as natural composite fiber (Heine and Hocker, 1995). Generally cross sectional view of the wool fiber is in elliptical cylinder, with diameter ranged from 15-100 μm, while the staple length can be determined by the breed, rate of growth of the wool and the frequency of shearing (Makinson, 1979).
Wool fiber has three distinct morphological parts in which the outer one is the cuticle layer, middle is the group of spindle-shaped cortex cells and they are separated from each other by a cell-membrane complex (Feughelman, 1997; Makinson, 1979; Plowman, 2003; Negri et al., 1993) and the inner is medulla as shown in Fig. 2 (Ammayappan, 2009a; Pourlis and Christtodoulopoulos, 2008; Ammayappan et al., 2012a).
 | |
| Fig. 1: | Scanning electron microscope of a fine wool |
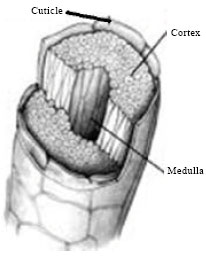 | |
| Fig. 2: | Components of a wool fiber |
Cuticle: Cuticle cells are the outer protective layer of the wool fiber and they form a sheath of overlapping scales and enveloping the cortical cells (Speakman, 1985, Negri et al., 1993). It comprises from 10-15% on the total weight of a fine wool fiber (Naik and Speakman, 1993). Each cuticle cell is composed of three distinct layers, called epi-cuticle, exo-cuticle and endo-cuticle (Rippon, 2003) as shown in Fig. 3.
The outermost layer of the wool fiber is epicuticle and it is composed of lipid so that it has good resistance against acid, oxidizing agent, reducing agent, enzyme and alkali (Makinson, 1979; Negri et al., 1993). Due to the presence of lipid component, it imparts hydrophobicity to wool fiber (Negri et al., 1993; Hocker, 2002). It bonded with cuticle cell through covalent isopeptide crosslinks. This lipid layer can be removed by treatment with alcoholic alkaline or chlorine solutions in order to enhance wettability (Negri et al., 1993; Brack et al., 1999; Ammayappan, 2009a).
Exocuticle A-layer is composed of cystine residues, peptide bonds as well as isodipeptide bonds through (ε-(γ-glutamyl)lysine) (Rippon, 1992; Heine and Hocker, 1995). The A and B layers of exo-cuticle can be solubilised by an oxidation or reduction treatment with prior enzyme treatment (Sawada and Ueda, 2001; Nolte et al., 1996).
Being rich in disulphide bonds, wool cuticle behaves as a diffusion barrier to chemicals (Schafer, 1994). Any chemical pre-treatment that can modify -S-S-bond, will improve the reactivity by changing the composition of cuticle cell as well as morphology of the wool fiber (Pascual and Julia, 2001). Due to elastic nature of wool polymer and presence of cuticle cells, wool fiber has five kinds of shrinkage: relaxation shrinkage, consolidation shrinkage, swelling shrinkage, hygral expansion and felting shrinkage in which felting shrinkage is undesirable one which occurs due to intrinsic properties of wool as well as it affects the performance of woolen products (Ammayappan et al., 2006; Bradbury, 1976; Shahbaz et al., 2002).
Cortex: Cortex occupies nearly 80-90% of the wool fiber and has the biggest influence on wool’s physical and chemical characteristics. It composed of three typical cortical cells called ortho cortex (60-90%), para cortex (40-10%) and meso cortex (only in low-crimp merino wool). The three cortex cells differ in their characteristics such as accessibility and ease of swelling in alkali, in birefringence and are wound around each other in phase with the fiber crimp. Ortho and para cortical cells differentiate each other in chemical and physical properties in which the ortho cortex is heavily dyed with basic dyes while para cortex is less reactive (Speakman, 1985). These differing properties are related to wool’s crimp and they impart resilience, loftiness or bulk (Simpson and Crawshaw, 2005; Bradbury, 1973; Ammayappan and Moses, 2005).
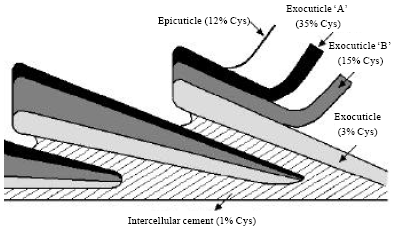 | |
| Fig. 3: | Schematic scale structure of the cuticle |
Medulla: Medulla is a network of air-occupied cell walls present in the middle of the medium or coarse wool fiber. Generally fine wools did not have medulla while even in coarse wool only some of the fibers are medullated. It occurs due to insufficient nutritional supply during growth of wool in sheep. Medulla of wool fiber may be interrupted or fragmented (hetero medullation), unbroken (hairy medullation) and heavily medullated (kemp) (Onions, 1962; Wildman, 1954). They are varied from 0-90% which depends on the sheep breed in which 20-30% medulla is responsible for good resilience in carpet and >50% responsible for brittleness and non-luster. Discontinuous (ladder type) medullas of definite patterns are found in Angora rabbit hair (Ammayappan and Moses, 2005, 2007c).
PREPARATORY PROCESSES OF WOOL
Wool is mainly used in the following broad product areas: Men’s and women’s woven outerwear, knitwear, underwear, socks, hand knitting yarns, blankets, upholstery, filled beddings, rugs and carpets. All these product areas offer challenge to the wool dyer and finisher to innovate and add value to the final product (Simpson and Crawshaw, 2002). Pretreatments and chemical modifications of any natural fibers materials and their biomasses are preferred to enhance the value as well as functionality, since they modify their physical, chemical as well mechanical properties (Nayak et al., 2012a, b). A variety of processes are available to improve the appearance, handle, performance and durability of the woolen materials in which preparatory processes are used to remove the impurities as well as enhance its process ability. Scouring, bleaching, carbonizing and even milling have been carried conventionally for all products (Ammayappan, 2009a, b).
Scouring of wool: Scouring is used to remove adhered and natural impurities present in the greasy wool fleece. Generally scouring can be carried in batch process in which sodium carbonate and non-ionic surfactant combined used. Depends on the percentage of grease (10-25%), the addition of sodium carbonate and duration of the treatment can be increased. Lanolin, a valuable organic by-product from the scouring can be recovered for cosmetic application (Lewin, 2007; Ammayappan et al., 2011b).
Carbonizing of wool: Carbonizing is the process to remove vegetable/cellulosic matter present in the wool fleece. Burred wool is padded with 10% sulphuric acid, dried and cured at 120°C for 30 min. Since, the process of carbonizing involves sulphuric acid and heat, care should be taken to prevent yellow coloration (Lewis, 1992).
Bleaching of wool: Wool is bleached with hydrogen peroxide at low temperature if it will intend to be white dyed or light dyed, like for baby clothes. At high temperature wool fiber may be damaged since there is possibility of oxidation of disulfide bonds (Raja et al., 2011). It is generally preferred for white shawls and stoles.
SURFACE MODIFICATION OF WOOL
Generally pretreatment of wool fiber can also modifies its physiochemical and mechanical properties as per the processors requirements. Among them, pretreatments using either oxidizing agents or reducing agents are applied to wool extensively in order to make them reactive which make the post treatment as effective and uniform. For example a reduction pretreatment using sodium bisulphite is given to wool prior to Papain treatment for effective anti-felting treatment for wool (Ammayappan et al., 2011a, c; Ammayappan and Moses, 2006).
Due to release of absorbable organic halogens-AOX to the effluents by the chlorine-Hercosett process, the development of eco-friendly pretreatment such as enzymatic process for shrink-resist treatment using different protease enzymes is focused in 1980’s. Recently chitosan, a biopolymer has been introduced as a shrink-resist chemical and plasma treatment which is also used as shrink-resist treatment as well as pretreatment for polymer applications (Heine, 2002; Lewis, 2002; Lewin, 2007; Ammayappan, 2007a, 2009c). However, there is research works used semi-durable polymer or high polymer polyphenols like tannic acid to enhance the reactivity of the natural fibers (Venkidusamy et al., 2002; Ammayappan and Moses, 2007b).
Oxidative/reductive treatment: Gaseous fluorine treatment oxidized the wool fiber surface, resulting in cystine bond cleavage and to form cysteic acid with a loss of surface sulphur and confers shrink resistance (Kidd et al., 1995). When wool fabric is treated with different oxidizing agents in the following steps imparted shrink resistance. The process sequence is Potassium persulphate→Potassium permanganate→Acid chlorination→Hydrogen peroxide bleaching. Treatment of wool fabrics with polar solvents such as n-propanol and concentrated formic acid removes lipid materials from cell membrane complex results in improvement in abrasion resistance. The improvements are stable to mild finishing, but are reduced by treatments in aqueous solutions above 85°C (Feldtman and Leeder, 1984).
An oxidative batch wise pretreatment for wool fabric using 6% mono-peroxyphthalate (over weight of wool) at 30°C for 30 min confers 0% shrinkage with 4% strength loss (Levene et al., 1996). Pretreatment of woolen material with soda ash leads to change in the fine structure which gives rise better dyeing properties in terms of higher rate of dyeing and dye bath exhaustion (Jovancic et al., 1993). Optimized a recipe for shrink resistance finish for wool fabric with Basolan-DC by studying their physio-chemical and mechanical properties. The Basolan DC gives optimum results at 2.5-4.5% (over weight of wool) concentration at pH 4-5/30 min/20°C with 10% sodium sulphate as additive (1993).
Kan et al. (1998a) found the alkali solubility, time of half dyeing (t½) and shrink resistance of wool fabric treated with Basolan-DC alone and combination with Low Temperature Plasma (LTP) and Basolan-MW polymer. The LTP/Basolan-DC combinations show high alkali solubility, low t½ and better shrink resistance than other combinations. Reduction or partial hydrolysis of wool followed by post succinylation with succinic anhydride raises water absorbability, hygroscopic nature and reduces crystalline region. Introduction of new carboxylic groups and increasing in amorphous region contribute to the raise in hygroscopic nature of wool (Kohara and Nakajima, 2004). Recently Scholler introduces EXP 4.0 process that employs natural salts as an oxidization agent to impart shrink-resistant finish.
Enzyme treatment: Enzymes are proteins of large molecular size, in which their active groups react with substrates to form products by decreasing the activation energy of the reaction. However, they are pH, time, temperature and substrate specific. Enzymes can be applied potentially to all stages of textile production (Shen et al., 1999; El-Sayed et al., 2002; Ammayappan and Gupta, 2011). Among different enzymes, protease and lipase enzymes are mainly used for wool. Proteases constitute the most important group of industrial enzymes which has major application is the detergent industry (Kantouch et al., 2005).
Addition of proteases and lipase to detergents considerably increases the cleaning effect by removing protein stains such as blood and egg and increases the consumption of surface-active substances thereby decreasing the pollution load (Nolte et al., 1996; Riva et al., 1991; Moreira et al., 2002; Grebeshova et al., 1999). The use of proteases to reduce prickle and improve softness in wool has been investigated with encouraging results (Heine, 2002). However, wool bio-finishing is not yet implemented at an industrial scale and attempts have been executing to implement enzymes periodically in wool finishing. The important chemical reactions occurred during enzyme treatment are given (Fig. 4) (Kantouch et al., 2005; Ammayappan et al., 2011c; Ammayappan and Moses, 2010).
Enzyme pretreatment on dyed wool fabrics improved softness, handle, drape, pilling resistance and change the colour (Riva et al., 2002; Raja and Thilagavathi, 2011; Sharma et al., 2011). Protease enzyme penetrates into amorphous region and causes swelling and it leads to changes in the disulphide region of cystine than amide components during chemical degradation. So percent content of ordered alpha helix region decreases with conversion of beta sheet form in enzyme pretreatment (Wojciechowska et al., 2004; Ammayappan and Moses, 2011). Riva et al. (1993) applied bacterium Streptomyces Fradiae (SFP) on wool fiber and inferred that it attacked the neutral keratin and hydrolyzed some peptide linkages of wool fiber. This enzyme treatment causes weight losses at higher concentrations without change in physio-chemical properties while the uniformity is not better (1993).
Shen et al. (1999) inferred that the activity of enzyme on wool depends on the bath buffer, its ionic strength and previous process history of substrate. Levene et al. (1996) showed that sodium bisulphite pretreatment enhanced the activity of alkaline bacterial protease enzyme and confers better shrink resistance to wool tops and fabric. Among enzymes Esperase was found to be most active while causing the least damage. Wool yarn spun from Novolan-L enzyme treated wool tops has lower number of neps, lower number of breaking through spinning, low co-efficient of hairiness, where the knits have softer feel with better drape, improved pilling performance and dimensional stability.
A pretreatment of wool with lipase/sodium-monoperoxyphthalate/sodium sulphite forms Bunte salt with low concentration of cystine monoxide and cystine dioxide and enhances the efficiency of Papain enzyme (El-Sayed et al., 2002). Cardamone et al. (2004) inferred that pretreatment of woven and knitted wool fabric of various weights with peroxy carboximide ruptured the peptide and cystine disulphide cross-linkages through hydrolysis and oxidation reaction. The subsequent proteolytic enzyme treatment bio-polishes and controls the shrinkage without appreciable loss in strength and elastic recovery. The addition of a protease enzyme in a oxidizing or reducing bleaching bath shortens the bleaching time by half for the same whiteness index (Levene, 1997). Treating of wool with a haloperoxidease (hydrogen peroxide+halide) and a proteolytic enzyme results in improved shrink resistance.
 | |
| Fig. 4: | Chemical reactions between enzyme and wool fiber |
In presence of hydrogen peroxide, wool cuticle degrading enzyme called Bacillus cereus strain NS-11 modified the cuticle components preferentially without damaging the inner components of wool fiber. Lantto et al. (2005) found that peroxidases are able to catalyze oxidation of wool fibers corresponding to 35-40% of the tyrosine residues located on the wool surface or 2% of the tyrosine residues in the wool fiber.
Enzyme pretreatment on wool fabric decreases the resistance of the fiber to dye diffusion and so it increases the adsorption rate constant and decreases in the apparent activation energy for the dyestuff when compared to untreated fabric (Riva et al., 2002). Enzyme pretreatment of wool fabric with trypsin increases exhaustion of natural dye like crocin, beta-carotene, curcumin, chlorophyll and carmine without change in fastness property (Liakopoulou et al., 1998; Tsatsaroni and Liakopoulou, 1995; Tsatsaroni et al., 1998). Levene and Shakkour (1995) found that a chlorination pretreatment followed by an alkaline protease treatment on wool fiber made it clearly descaled wool fiber with enhanced luster. The handle of the fiber can be retained in the post softening treatment.
Brier (2000) stated that treatment of wool fabric with Perizym-AFW improved shrink resistance, whiteness, pilling behavior, dye ability and wash ability. This is called as Lanazym process which is based on purely enzymatic antifelting finishing of wool. El Sayed et al. (2002) described an enzyme-based process to improve the felting resistance of wool top. In this process, lipase is used in the pretreatment step, glutathione reductase in the reduction step and Papain in the after treatment step. The lipase removes lipids from the outer surface of wool; glutathione reductase reduces the disulfide bonds in wool keratin together with nicotine amide adenine dinucleotide phosphate in the reduced form and Papain smoothes the wool scales. Wool fibers treated with this system showed good felting resistance as compared with untreated wool, but still was inferior to that treated with the chlorine/Hercosett process.
Ammayappan et al. (2011a) inferred that alkaline stable (Savinase) and neutral-stable enzyme (Papain) treatments on wool based fabrics have progressive effects on its properties such as handle, softness, comfort and mechanical properties, in which the extent of modification of wool fiber in cuticle as well as in cortical level is better in papain enzyme treatment than in savinase enzyme treatment. However, the hand value and softness of savinase-treated and finished fabrics are better than papain treated ones, since they exhausted moderate finishing chemicals. Ammayappan et al. (2009) inferred that enzyme pretreatment can be used to scour the rabbit hair, cotton, viscose rayon blended knitted fabric in order to protect the Angora rabbit hair.
UV radiation on wool: CSIRO has studied the effect of UV radiation on wool fabric in order to modify its surface characteristics. Aromatic amino acid and cystine residues in the wool fiber absorb strongly below 350 nm of UV radiation. During UV radiation, oxidation of surface fibers will occur which introduces more functional groups which modification can increase rate of dyeing and fastness properties. Since, it improves the surface morphology, CSIRO inferred that it could be used to produce heavy shades like black and navy blue (Ammayappan, 2009a).
Chitosan treatment on wool: Chitosan a natural biopolymer, chemically called as beta-(1, 4)-2-(amino)-2-deoxy-D-glycopyranose. In aqueous solution at pH<6.5, it is its protonated form (NH3+) and behaves like a cationic polyelectrolyte and interacting with negatively charged molecules. It can be applied by reactive bonding to protein fiber and by cross linking of chitosan at an acidic condition (pH 5). Chitosan has poor durability on wool fabrics, since it did not have strong bonding with wool polymers (Onar and Sariisik, 2004; Poovi et al., 2011; Ahmad et al., 2009).
Pretreatment of 100% wool fabric or wool/nylon blended fabric with alkaline peroxide improves the adsorption of chitosan. The adsorbed chitosan increases the dyeing rate of wool, K/S value, fastness property, wettability and shrink resistance (Pascual and Julia, 2001; Ammayappan et al., 2010a). Jovancic et al. (2001) found that a peroxide treatment followed by a protease treatment enhanced the adsorption of chitosan on wool. They described that alkaline peroxide bleaching generates new active sites on the wool surface (-SO3¯) and protease treatment modified the hydrophobic cuticle scales.
The cationised chitosan forms strong ionic bonds with sulphonic acid groups and form uniform layer which confers shrink resistance. Chitosan present on the surface of wool fiber can easily attracts and interacts with the dyestuff, so that it improves the dyeing rate and reduces the dyeing time. So, it is considered as a predominant dyeing site on wool fiber surface (Jocic et al., 2005; Jamil et al., 2010).
The enzymatic treatment enhances whiteness and confers shrink resistance to wool, but an increase in the enzyme concentration leads to a detrimental effect on the physio-mechanical properties. Being form a uniform film on the surface of the fiber, chitosan resists the subsequent enzymatic action on wool fiber (Vilchez et al., 2005).
Onar and Sariisik (2004) studied the effect of enzymatic treatment, bleaching treatment and polymer additive treatment on wool fabric both in separate and different successive forms. They observed that the protease treatment enhances chitosan adsorption on wool and also caused an increasing loss of tensile strength. Citric Acid (CA) is used as a cross linking agent in fixing of chitosan by pad-dry-cure treatment on woolen fabrics. Citric acid produced esterification with the -OH group of the wool and chitosan and transamidation with the -NH2 group of the permanganate oxidized wool and formed a crosslink. However, citric acid enhances the cross linking efficiency of oxidized wool fiber; it imparts antimicrobial and antiseptic effects of the woolen fabrics. Since, it imparts cross linking effect, this finishing reduces the fabric softness and increases the yellowness (Hsieh et al., 2004).
The hydrophobic character of chitosan finish is increased by incorporation of long-chain N-acyl groups and so it improved the anti-felting behavior better than chitosan finish itself (Roberts and Wood, 2001). Jeong et al. (2002) inferred that chitosan finishing on wool fabrics increase the bending rigidity and the magnitude of the changes being dependent on the molecular weight of the chitosan and the decatizing conditions.
Plasma treatment on wool: Plasma is produced from an electrical discharge of gaseous mixture, either under vacuum or atmospheric pressure and so it contains mixture of electrons, ions and free radicals and. Plasma can modify the surface morphology of textile fiber and increase the wettability (surface energy) so, it promotes reactivity during subsequent processing (Ammayappan et al., 2012b). The effect of a plasma treatment on wool is much the same in that the wettability of the fiber is enhanced considerably, an effect which has implications for improved dyeing, printing and the subsequent application of a variety of different chemical treatments (Kan et al., 1998b; Poll et al., 2001).
When woolen materials in top or fabric form pretreated with glow discharge in presence of non-polymerizing gases like air, oxygen and nitrogen, the outer surface of wool fiber (30-50 nm) modified due to plasma etching and surface oxidation. Plasma is attacking the wool fiber surface like gun and so it partially abrades the fatty acid layer from the cuticle and parts of the exocuticle-A. Simultaneously there will be a surface oxidation by oxygen radical which introduces new anionic groups i.e., sulphonic and carboxylic acid groups. Carboxylate groups are derived from a backbone of the protein chains and sulphonate groups by oxidation of disulphide bonds in the cuticle cells. The hydrophilicity of the wool reduces the hydrophobic effect between the fibers. A thin film of water can be built up on the fibers during washing which separates them each other and decreasing the differential frictional effect and therefore diminishing the felt ability of wool (Hesse et al., 1995).
The air plasma treatment of wool fabrics promotes the spreading and adhesion of the hydrophilic chitosan on wool fibers and produces a small physical cuticle surface change compare with oxygen plasma treatment (Erra et al., 1999). Plasma treatments represent probably the most elegant approach to fiber modification for shrink-resist effects because they are surface specific, totally effluent free and thus environmentally friendly but have still to be commercially proven (Kim and Kang, 2002). Chitosan is also improving the dye ability of the wool based-two layer knitted fabric after chitosan treatment by exhaustion method, however it slightly imparted stiffness to the material (Ammayappan et al., 2010b).
Consumer across the globe demanding comfort, easy care, relaxed dressing and value for the money they spend. However, there are so many surface modification processes have been developed, woolen materials required the suitable post functional finishing like softening, antimicrobial finishing, water-repellent finishing etc. It can offer a wide scope for woolen processors to develop diversified woolen products by incorporating all aesthetic properties based on consumer demands as well as it can improve the durability of the products. Development of surface modification process with introduction of some functional groups without deteriorating its instinct property of the wool fiber is the need of the hour.