Research Article
Cost-effective Approaches for in vitro mass Propagation of Rauwolfia serpentina Benth. Ex Kurz.
Not Available
Arun Kumar Kukreja
Not Available
Suman Preet Singh Khanuja
Not Available
In vitro growth of plants is largely determined by the composition of the culture medium. Whenever we read any research paper related to tissue culture studies, in most of the cases, we will find a generalized statement that microbial and plant tissue culture materials agar has been the medium was solidified with 0.6-0.8% agar. It will be worth to say that, agar has been taken for granted in the most frequently used gelling agent in tissue culture media as it is biologically inert and has high gel clarity, stability and resistance to digestion by plant enzymes (Henderson and Kinnersley, 1988). In spite of its exclusive use and advantages as a gelling agent, many reports on its adverse effects have been published over the years (Romberger and Tabor, 1971; Debergh, 1983), which includes batch to batch variability, presence of impurities which may have inhibitory effects on growth. Moreover agar happens to be the most expensive constituent of plant tissue culture media followed by sucrose. Additionally local accumulation of heat, hindrance of the availability of dissolved oxygen to the cultured plant parts in media, contamination of media through agar sticking to the neck of the culture vessels etc have also been reported to be other disadvantages of agar (Bhattacharya et al., 1994). Sometimes roots grown in agar gelled media often get damaged during washing prior to transplantation and remains of agar gelled medium in the roots call for unwanted bacterial and fungal contamination. At commercial level, agar is extracted from some red algae (Mac Lachlan, 1985). Huge demand of agar has been resulting in over exploitation and shrinkage of its natural resources.
In the search for an alternative, eco-friendly and economical constituent of tissue culture media, a number of support matrices (Kong and Chin, 1988; Titel et al., 1987; Henderson and Kinnersley, 1988; Bhattacharya et al., 1994; Babbar and Jain, 1998; Puchooa et al., 1999; Gangopadhyay et al., 2002, 2004; Jain and Babbar, 2005, 2006; Babbar et al., 2005) have been tested as potent alternative to agar. But most of these materials are less explored in application than agar and have their own limitations. During the last few years, there has been immense increase in the efforts to have suitable substitutes for agar. The high production costs in the micropropagation procedures can drastically be reduced if cheap and (or) reusable alternatives to highly expensive tissue culture grade purified agar could be employed without compromising the quality of regenerated plants. The present study hence, aims at the development of a reliable yet simple and cost effective micropropagation protocol using glass beads as reusable and biologically inert alternative support matrix in liquid culture medium for Rauwolfia serpentina and also the quality assurance of the look like clones produced.
MATERIALS AND METHODS
Young and tender shoots of Rauwolfia serpentina were collected from the one-year plants maintained in the experimental farms at CIMAP. After thorough washing and surface sterilization with HgCl2 (0.1%) for 4 min followed by 4-5 rinses with sterile distilled water, single nodal explants (1-2 cm) were cultured on basal MS medium (Murashige and Skoog, 1962) gelled with 0.8% agar and supplemented with 4.44 μM 6-benzyl amino purine (BAP), 0.54 μM α-Naphthalene Acetic Acid (NAA) and 3% sucrose. These mother cultures served as source of inoculum (i.e., single nodal explant) for initiating the cultures for further experiments.
The glass beads (1.5 mm diameter) were soaked overnight in chromic acid and washed with Teepol 1% and distilled water and dried in hot air oven prior to use. About 30 g glass beads were added as support matrix instead of agar to 30 mL of liquid medium per 250 mL culture flask to which six explants were inoculated. The pH of the medium was adjusted to 5.80±0.02 prior to sterilization at 15 lbs. and 121°C for 20 min. All the cultures were incubated in a culture room at 25±2°C under a 16 h photoperiod and 60% relative humidity. The liquid cultures, with or without glass bead matrix were maintained in agitated (70-75 rpm) as well as static conditions. The cultures on semisolid medium (containing 0.8% agar) were treated as control. A minimum of three replicates were maintained for each treatment and observations were recorded after six weeks of culture initiation. Shoot multiplication rate was also compared on glass bead support matrix of two different sizes i.e., 1.5 and 4.0 mm. In another experiment Conventional carbon source in plant tissue culture studies i.e., Sucrose was replaced by low-grade market sugars. Two different grades were tested. One was ordinary market sugar @ Rs. 20 kg and other was Daurala sugar (cubes) @ Rs. 53 kg. All the sugars were added to the medium at 3% concentration. For induction of in vitro rhizogenesis in regenerated single shoots, only 5 mL of liquid half-strength basal MS medium containing 2% sucrose and supplemented with 5.4 μM NAA was dispensed and support was provided by adding 8 g of glass beads per culture tube. A minimum of ten replicates were maintained for each treatment. Relative cost analysis was calculated on the basis of producing a complete single regenerated shoot depending upon the use of conventional (i.e., agar, sucrose) and/or non-conventional (glass beads, Daurala sugar, market sugar).
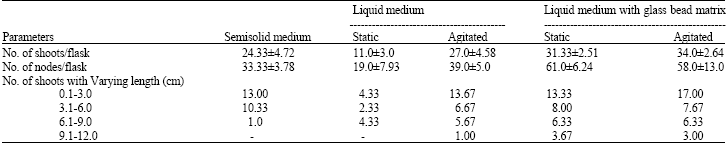
Recorded observations revealed that glass beads as support matrix in liquid medium both in static and agitated conditions supported active proliferation of R. serpentina shoots devoid of any sign of vitrification (Fig. 1 and Table 1).
However shoots cultured on liquid medium with glass bead matrix in agitated conditions exhibited better growth and multiplication rate (34.0±2.64 shoots/flask) followed by that of the static liquid medium supported with glass beads (31.33±2.51 shoots/flask) and liquid agitated medium without glass beads (27.0±4.58 shoots/flask) as against the control (24.33±4.72 shoots/flask). Interestingly, a very low shoot multiplication rate (11.0±3.0 shoots/ flask) was recorded in static liquid cultures. The number of nodes regarded as the multiplication factor in the present study also followed almost the same pattern as that of number of shoots per flask. The number of nodes per flask recorded was highest in static liquid medium supported with glass bead matrix (61.0±6.24), followed by that of the agitated liquid medium with glass bead support (58.0±13.0). Number of nodes on liquid agitated medium without glass beads was also higher (39.0±5.0) than the control (33.3±3.78). A very low response (19.0±7.93) in terms of number of nodes/flask was observed in static liquid culture medium. This was most likely due to the asphyxiation of the explant resulted by their submergence in the liquid medium. An equally good response of growth and multiplication of in vitro shoots was attained on liquid culture medium containing glass bead matrix of both sizes (1.5 and 4.0 mm diameters). Observations showed that growth performance was better in market sugar (25.6±1.52), as well as in Daurala sugar (27.0±2.64).
In vitro regenerated single shoots showed root initiation (8 roots per shoot) and growth in rooting medium supported with glass bead matrix, which was comparable to those on semisolid media (10.2 roots per shoot). Although the number of roots were 1.25 times higher in semisolid medium, but roots developed on glass bead matrix were longer and thinner, soft and flexible. Upon transfer under the glass house conditions, the in vitro raised plantlets showed 85-90% survival irrespective of their culture source.
Elimination of agar from the culture medium reduced the cost of the per liter culture medium about 65%. By dispensing 60 mL of semisolid medium per flask, only 16 culture flasks were prepared per liter medium. But in case of using liquid culture medium with or without glass beads, only 30 mL volume of the culture medium was sufficient per culture vessel. Thus, 33 culture flasks could be prepared using one liter of liquid culture medium. Hence, a substantial reduction in culture medium consumption was achieved, which ultimately resulted in reduction in plant production cost. If we compare the cost of these three sugars, then cost of sugar component was drifted from 1/10 to 1/25. By using market grade sugar in glass bead supported liquid medium, up to 94% reduction in the medium cost was achieved (Fig. 2, Table 2).
| Table 1: | Growth performance of shoots cultures of R. serpentina on different support matrix |
 | |
 | |
| Fig. 1: | In vitro culture of R. serpentina: (A) Shoots cultured on liquid medium with glass bead matrix, (B) Root development in glass bead matrix supported liquid medium and (C) Plant growing under glass house conditions |
By comparing the cost of single shoot raised on semisolid medium (Rs. 0.126), a single shoot regenerated in liquid medium with glass beads as support matrix cost only Rs. 0.016, showing about 87% reduction in production cost (Table 2). Additionally, the cost of single shoot produced on medium having glass bead along with market sugar medium and Daurala sugar was just Rs. 0.0034 and Rs. 0.004, respectively. Hence replacement of sucrose by market sugar further cut down the production cost of single shoot up to 96-97%.
| Table 2: | Cost analysis of single regenerated shoot of R. serpentina on different states of the culture medium |
 | |
| Cost of chemical ingredients (with or without agar) required for preparing one liter of MS basal culture medium (Murashige and Skoog, 1962) calculated as per HIMEDIA price list 2005-2006. Cost of medium does not include the cost of distilled water) | |
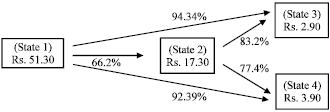 | |
| Fig. 2: | Reduction in the % cost of MS medium based on sugar component |
Although initial cost of glass beads is comparatively higher, however, these glass beads can be repeatedly used for indefinite times after proper washing and drying. If unfortunately, culture medium gets any fungal or bacterial infection, in that case also these beads could be reused after decontamination followed by washing and rinsing and hence are economical. These glass beads are chemically inert, resistant to heat and acid or alkali action. Whereas, certain limitations associated with other synthetic supports restrict their use (Titel et al., 1987; Henderson and Kinnersley, 1988; Bhattacharya et al., 1994; Gangopadhyay et al., 2002; Jain and Babbar, 2005). The addition of fresh sterilized medium to the culture vessel under aseptic conditions further eliminates regular subculture practice for regular maintenance and further minimized the frequency of contamination during subculture. Furthermore, this technique can also be useful in the experiments where removal and addition of the culture medium after a particular culture period is required. Medium can easily be sucked by sterilized pipette. Penetration of roots as observed in semi-solid culture medium causes injury to roots during removal and cleaning of roots, hence, poses deleterious effects on their field adaptability. However, the use of glass beads rendered the removal of plantlets from the medium very easy. MacLeod and Nowak (1990) have earlier reported the use of glass beads for understanding the role of polyamines, however since then, less attention has been paid on the use of this biologically inert support matrix. In the earlier report glass beads were used as a support matrix in test tube culture vessel system. Since the growing shoots developed in test tubes, get easily supported by the walls of the test tube because of their smaller area. However, it becomes very important to investigate whether they can be used with same efficiency in large size culture vessels (250 mL-1 Erlenmeyer flask). They also reported about 60% saving in medium consumption by substituting agar with glass beads. Using liquid culture medium amount of medium consumption was also drastically reduced approximately to half, hence, there is substantial increase in the shoot multiplication per liter culture medium consumed and use of market sugar ultimately resulting in the reduction in the cost of single regenerated shoot up to 96-97%.
The purpose of selecting Rauwolfia serpentina as test plant was because of its wide demand in pharmaceutical industry. It is commonly known as ‘Sarpagandha’ and is an important endangered medicinal plant distributed in tropical Asia and America and is the source of reserpine having a depressant action on central nervous system producing sedation and a lowering of a blood pressure. Several reports on tissue culture related studies in different species of Rauwolfia have been published earlier (Roja et al., 1985, 1987; Mathur et al., 1987; Sarkar et al., 1996; Patil and Jayanthi, 1997). In the view of the overwhelming interest in this important medicinal plant species, it becomes very necessary to device some low cost approaches for mass propagation of this medicinally important plant employing tissue culture techniques. A successful attempt has been made in the present study to adopt a simple and cost effective approach for micropropagation of R. serpentina and to present relative cost analysis based on support matrix and carbon source in culture medium as cost factor.
The use of glass bead matrix in liquid medium (static or agitated) can therefore be considered as an effective substitute to replace conventional practice of using agar (which is consumed during every culture and significantly adds towards the cost of culture medium). Through this paper authors would like to encourage the use of glass beads as support matrix and to popularize this practice, which is safe particularly in micro propagation studies, where cost of planting material is the major concern. Employing static mode of the cultures further reduction in production cost can be expected which reduces the energy inputs.
On the basis of the discussion, it can be stated that the currently standardized technique not only supports efficient multiplication of the R. serpentina but also reduces the cost of in vitro practices to a significant extent, making it economical and suitable to be adopted by micropropagation industries world over.
Authors are grateful to Director, CIMAP for providing all facilities during the studies. Award of junior research fellowship to MKG by the Council of Scientific and Industrial Research, New Delhi is gratefully acknowledged.