ABSTRACT
Ethanolic extracts of 16 Turkish plant species used in folk medicine were investigated for their antimicrobial activities against nine bacterial species (Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus vulgaris, Bacillus cereus, Mycobacterium smegmatis, Listeria monocytogenes and Micrococcus luteus) and three yeasts (Candida albicans, Kluyveromyces fragilis and Rhodotorula rubra) using the disc diffusion method. Of the 16 plants tested, ten showed antimicrobial activity. Each plant species has unique against different microorganisms. The most active antimicrobial plant against bacteria and yeasts was Myrtus communis. It is conclude the plants studied may be sources of antimicrobial agents.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.104.107
URL: https://scialert.net/abstract/?doi=ajps.2004.104.107
INTRODUCTION
Medicinal plants have been used for a wide variety of purposes for many thousand of years in Turkey. In particular, extracts and oils of these plants have formed the basis of many applications, including raw and processed food preservation, pharmaceutical, alternative medicine and natural therapies.
Recently, the antimicrobial activity of various plant extracts has been studied on the growth of many microorganisms in Turkey[1-7]. However, these investigations were inadequate. This paper reports the antimicrobial screening results of selected Turkish medicinal plants with traditional uses.
MATERIALS AND METHODS
Plant Materials: 16 plant species commonly used in folk medicine in Turkey were selected (Table 1). Mature plants and their parts were collected from different cities in Turkey during the months of May-June of 2001. Voucher specimen of the plants was deposited in the herbarium of the Biology Department at Uludag University, Bursa-Turkey. The plant parts were air-dried.
Preparation of Extracts: Each dry powdered plant material (20 g) was extracted 100 ml 80% ethanol (Merck, Darmstadt) for 24 h by using Soxhlet equipment[8]. The extract was filtered using Whatman filter paper no.1 and the filtrates were then evaporated under reduced pressure and dried using a rotary evaporator at 55°C.
| Table 1: | Ehtnobotanic data of studied plants |
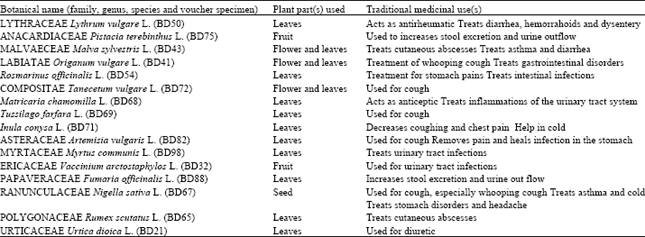 | |
Dried extract were stored in labeled sterile screw capped bottles at -20°C.
Microorganisms: The microorganisms used included : Escherichia coli ATCC 11230, Stapylococcus aureus ATCC 6538P, Klebsiella pneumoniae UC57, Pseudomonas aeruginosa ATCC 27853, Proteus vulgaris ATCC 8427, Bacillus cereus ATCC 7064, Mycobacterium smegmatis CCM 2067, Listeria monocytogenes ATCC 15313, Micrococcus luteus CCM 169, Candida albicans ATCC 10231, Rhodotorula rubra DSM 70403 and Kluyveromyces fragilis ATCC 8608.
Screening for antimicrobial activities: The dried plant extracts were dissolved in 10% aqueous dimethylsulfoxide (DMSO) to a final concentration of 200 mg mL-1 and sterilized by filtration through a 0.45 μm membrane filter. Empty sterilized antibiotic discs having a diameter of 6 mm (Schleicher and Schull No. 2668, Germany) were each impregnated with 50 μL of extract (10 mg disc-1) at a concentration of 200 mg mL-1. All the bacteria mentioned above were incubates at 35±0.1°C for 24 h by inoculation into Nutrient Broth (Difco) and the yeast cultures studied were incubated in Malt Extract Broth (Difco) at 25±0.1°C for 48 h. An inoculum containing 106 bacterial cells or 108 yeast cells/mL was spread on Mueller-Hinton Agar (Oxoid) plates (1 mL inoculum/plate). The discs injected with extracts were placed on the inoculated agar by pressing slightly. Petri dishes were placed at 4°C for 2 h, placks injected with the yeast cultures were incubated at 25±0.1°C and bacteria were incubated at 35±0.1°C for 24 h [9,10]. At the end of the period, inhibition zones formed on the medium were evaluated in mm. Studies were performed in triplicate. On each plate an appropriate reference antibiotic disc was applied depending on the test microorganisms for comparing.
RESULTS AND DISCUSSION
All plants shows antimicrobial activity against the test microorganisms with the exception of Artemisia vulgaris, Inula conysa, Nigella sativa, Rumex scutatus, Matricaria chamomilla and Urtica dioica which have no antimicrobial effects (Table 2). Moreover, it was determined that the extracts of Tanecetum vulgare, Tusssilago farfara, Myrtus communis, Vaccinium arctostaphylos and Fumaria officinalis have antiyeast effects, while the others do not.
The extracts obtained from Myrtus communis inhibit considerably the growth of all tested microorganisms except for Escherichia coli, Klebsiella pneumonia and Proteus vulgaris, having an inhibition zone of 14-34 mm. When the results obtained were compared to those of standard antibiotics, it was determined that the extracts of Myrtus communis have higher effect against these microorganisms.
Staphylococcus aureus is more susceptible to the extracts of Tanecetum vulgare, as compared to standard antibiotics except for OFX5 and TE30. Listeria monocytogenes is resistant and the other bacteria species are seen to be more susceptible to Rosmarinus officinalis extracts, as compared to the standard SAM20 and CTX30 antibiotics. Similarly, in comparison to CTX30 standard, it was seen that Klebsiella pneumoniae is more susceptible to Fumaria officinalis and Vaccinium arctostaphylos extracts.
The extracts obtained from Lythrum salicaria, Pistacia terebinthus, Malva sylvestris, Origanum vulgare and Tussilago farfara have moderately active against tested microorganisms when compared to standard antibiotics.
| Table 2: | Antimicrobial activity of plant extracts |
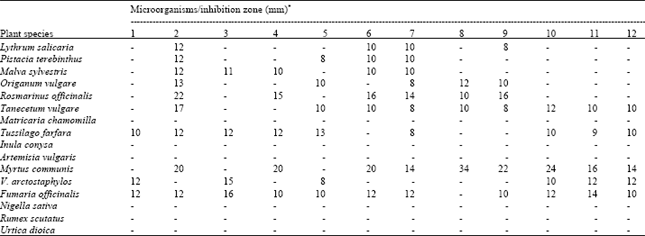 | |
| 1: Escherichia coli, 2: Staphylococcus aureus, 3: Klebsiella pneumoniae, 4: Pseudomonas aeruginosa, 5: Proteus vulgaris, 6: Bacillus cereus, 7: Mycobacterium smegmatis, 8: Listeria monocytogenes, 9: Micrococcus luteus, 10: Candida albicans, 11: Kluyveromyces fragilis, 12: Rhodotorula rubra Inactive (-); moderately active (8-13); higher active (>14) * includes diameter of disc (6 mm) | |
| Table 3: | Antimicrobial activities of some standard antibiotics |
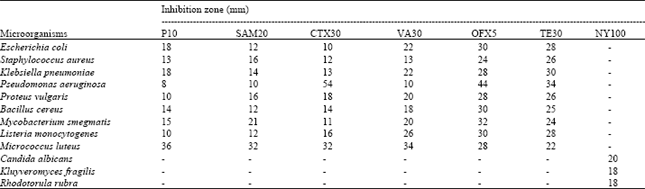 | |
| P10: Penicillin G (10 Units), SAM20: Ampicillin 10 μg, CTX30: Cefotaxime 30 μg, V30: Vancomycin 30 μg, OFX 5: Oflaxacin 5 μg, TE30: Tetracyclin 30 μg, N30: Nystatin 100 μg | |
Inhibition zone of the plants varying between 8 and 13 mm is formed.
The plants differ significantly in their activity against tested microorganisms. These differences may be attributed to fact that the cell wall in gram-positive bacteria of a single layer, whereas the gram-negative cell wall is multi-layered structure and the yeast cell wall is quite complex[11,12].
Ilçim et al.[13] reported that the extracts Myrtus communis showed antimicrobial activity, having inhibition zones 16-38 mm and Rumex scatatus extracts were found to be inactive. In another study, Rosmarinus officinalis extracts showed similar results against specific bacteria[14]. In similar study, ethanolic extracts of Fumaria officinalis inhibited Staphylococcus aureus but not Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa[15]. According to Mangena and Muyima[16], the antimicrobial activity of undiluted essential oil of Rosmarinus officinalis was 14 mm against Bacillus subtilis, 17.5 mm against Escherichia coli K12, 17 mm against Listeria monocytogenes ATCC 7644, 16 mm against Micrococcus luteus, 12 mm against Stapylococcus aureus and 16 mm against Yersinia enterocolitica ATCC 1610. Panizzi et al.[17] determined that the essential oil of Rosmarinus officinalis has little efficacy at 40 μg mL-1 Escherichia coli, 20 μg mL-1 against Stapylococcus aureus and 10 μg mL-1 against Bacillus subtilis. Our findings obtained from this study are similar to those stated above. It is thought that observed differences may result from the doses used in this study. In addition, microorganisms variable sensivity to chemical substances relates to different resistance levels between the strains[18].
A study reported that Urera baccifera from Urticaceae showed no antimicrobial activity against the test microorganisms; Candida albicans, Escherichia coli, Pseudomonas aeruginosa and Stapylococcus aureus[19]. The extracts of Urtica dioica showed no antimicrobial effects against the tested microorganisms in this study. Also, some studies confim our findings[14,15].
It is not surprising that there are differences in the antimicrobial effects of plant groups, due to phytochemical properties and differences among species. For the evaluation of plants which are naturally grown in Turkey and are potential useful resources, additional studies will be beneficial from medicinal and economic standpoints. In conclusion, whole extracts, especially the extracts of Myrtus communis can be used for protection against microorganisms.
REFERENCES
- Karaman, I., F. Sahin, M. Gulluce, H. Ogutcu, M. Sengul and A. Adiguzel, 2003. Antimicrobial activity of aqueous and methanol extracts of Juniperus oxycedrus L. J. Ethnopharmacol., 85: 231-235.
CrossRefPubMedDirect Link - Sahin, F., I. Karaman, M. Gulluce, H. Ogutcu and M. Sengul et al., 2003. Evaluation of antimicrobial activities of Satureja hortensis L. J. Ethnopharmacol., 87: 61-65.
CrossRefDirect Link - Khan, N.H., M. Rahman and M.S. Nur-e-Kamal, 1988. Antibacterial activity of Euphorbia thymifolia Linn. Indian J. Med. Res., 87: 395-397.
PubMedDirect Link - Mangena, T. and N.Y.O. Muyima, 1999. Comparative evaluation of the antimicrobial activities of essential oils of Artemisia afra, Pteronia incana and Rosmarinus officinalis on selected bacteria and yeast strains. Lett. Applied Microbial., 28: 291-296.
PubMedDirect Link - Haznedaroglu, M.Z., N.U. Karabay and U. Zeybek, 2001. Antibacterial activity of Salvia tomentosa essential oil. Fitoterapia, 72: 829-831.
CrossRefPubMedDirect Link








