Research Article
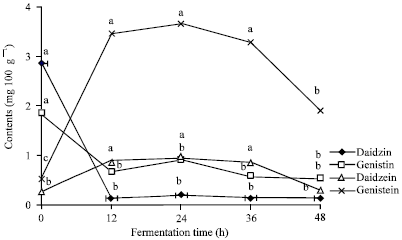
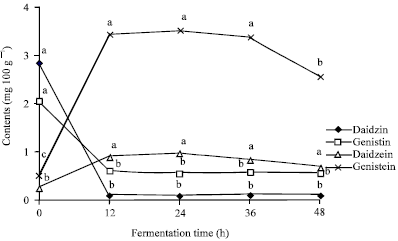
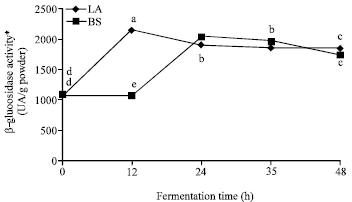
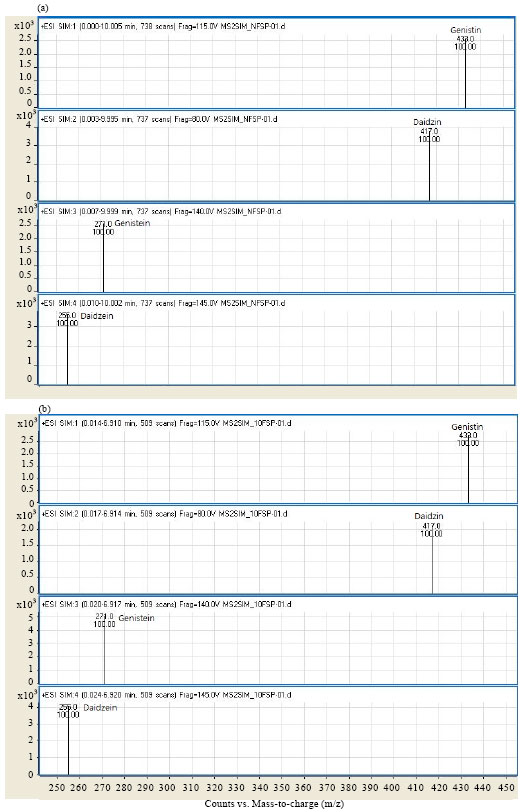
Fermentation Increases Isoflavone Aglycone Contents in Black Soybean Pulp
Department of Food Science and Biotechnology of Animal Resources, Konkuk University, Seoul, 143-701, Korea
Prabhat Kumar Mandal
Rajiv Gandhi College of Veterinary and Animal Sciences, Puducherry, 605 009, India
Ki-Won Lim
Department of Physical Education, Konkuk University, Seoul, 143-701, Korea
Chi-Ho Lee
Department of Food Science and Biotechnology of Animal Resources, Konkuk University, Seoul, 143-701, Korea