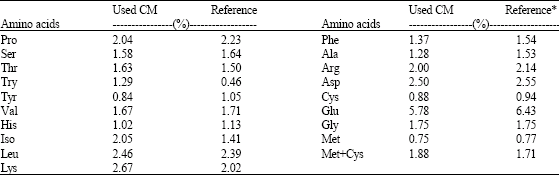
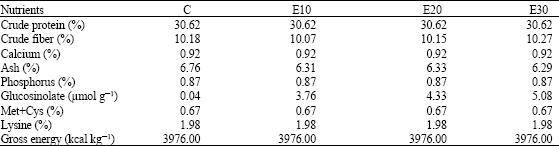
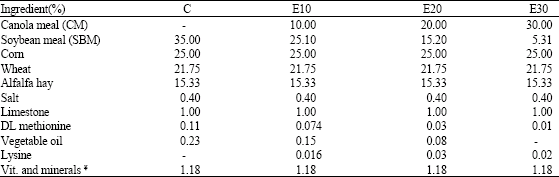
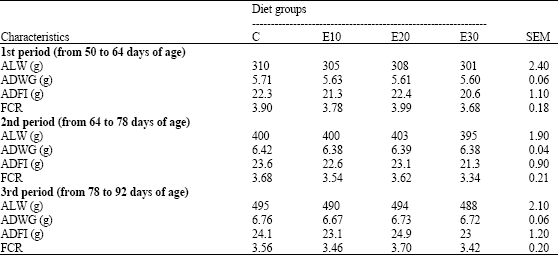
Research Article
Evaluation of Adding Canola Meal to Diet on Growth Performance of Male Wistar Rats
Department of Animal Biology, Faculty of Basic Sciences, Shahed University, Opposite Imam Khomeini Shrine, Tehran-Qom Highway, Tehran, Iran
H. Safafar
Food and Chemical Analysis Laboratory, Techno Azma Co., Tehran, Iran