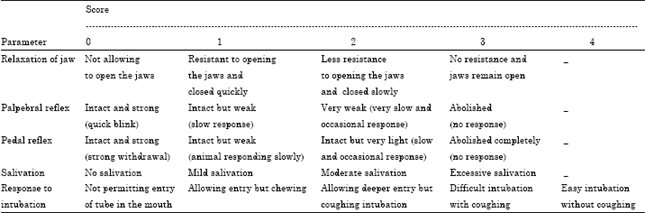
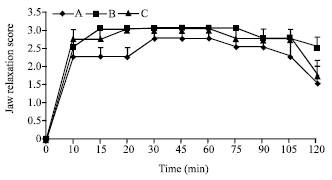
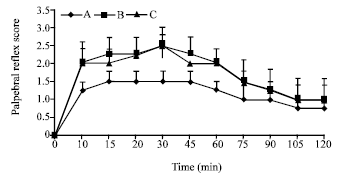
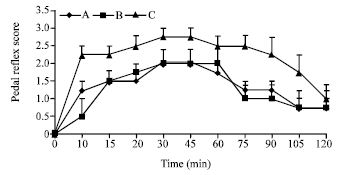
Research Article
Effects of Midazolam or Midazolam-Fentanyl on Sedation and Analgesia Produced by Intramuscular Dexmedetomidine in Dogs
Division of Veterinary Surgery, Indian Veterinary Research Institute, Izatnagar-243122, India
Amarpal
Division of Veterinary Surgery, Indian Veterinary Research Institute, Izatnagar-243122, India
P. Kinjavdekar
Division of Veterinary Surgery, Indian Veterinary Research Institute, Izatnagar-243122, India
H.P. Aithal
Division of Veterinary Surgery, Indian Veterinary Research Institute, Izatnagar-243122, India
A.M. Pawde
Division of Veterinary Surgery, Indian Veterinary Research Institute, Izatnagar-243122, India
D. Kumar
Division of Veterinary Pharmacology and Toxicology, Indian Veterinary Research Institute, Izatnagar-243122, India