Research Article
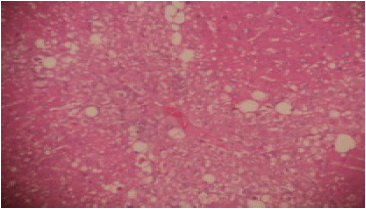
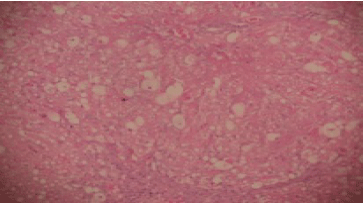
Hepatoprotective Potential of Commiphora kerstingii Engl. Stem Bark Against Carbon Tetrachloride-induced Acute Liver Injury
Department of Pharmacology and Toxicology, National Institute for Pharmaceutical Research and Development, Idu, Abuja, Nigeria
LiveDNA: 234.3605
I. Okoro
Department of Pharmacology and Toxicology, National Institute for Pharmaceutical Research and Development, Idu, Abuja, Nigeria
C.A. Ufondu
Department of Pharmacology and Toxicology, National Institute for Pharmaceutical Research and Development, Idu, Abuja, Nigeria