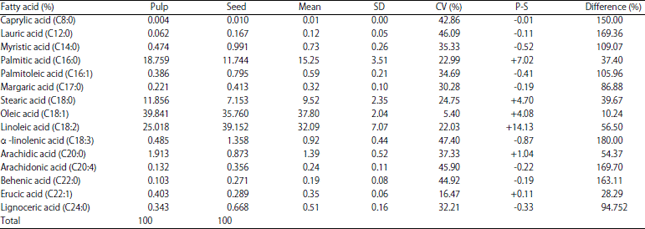
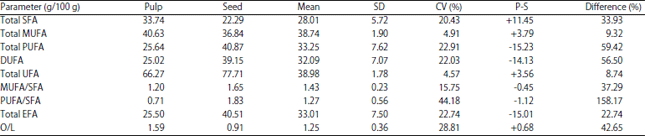
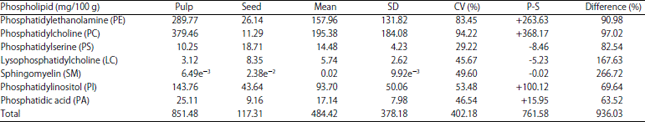
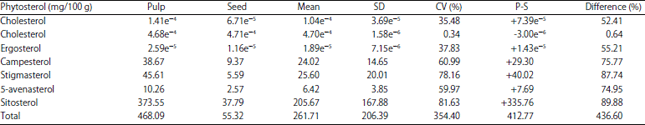
Research Article
Health Effect of Lipid Components Extracted from Avocado Pear (Persea americana) Pulp and Seed
Department of Chemical Sciences, Federal University Wukari, P.M.B. 1020, Taraba State, Nigeria
LiveDNA: 234.1611
Michael Augustine Odey
Department of Chemical Sciences, Federal University Wukari, P.M.B. 1020, Taraba State, Nigeria
Lawrencia Labaran
Department of Chemistry, Federal University of Lafia, P.M.B. 146, Lafia, Nasarawa State, Nigeria
Chibuzo Carole Nweze
Department of Biochemistry and Molecular Biology, Nasarawa State University, P.M.B. 1022, Keffi, Nigeria
Rasaq Bolakale Salau
Department of Chemistry, Federal University of Technology, Minna, P.M.B. 65, Niger State, Nigeria
LiveDNA: 234.30709
Stella Chintua Ortutu
Department of Pure and Industrial Chemistry, Nnamdi Azikiwe University, P.M.B. 5025, Awka, Nigeria