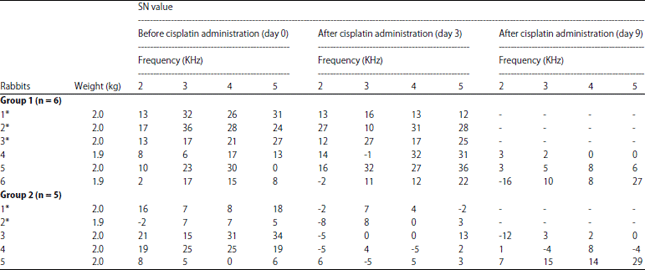
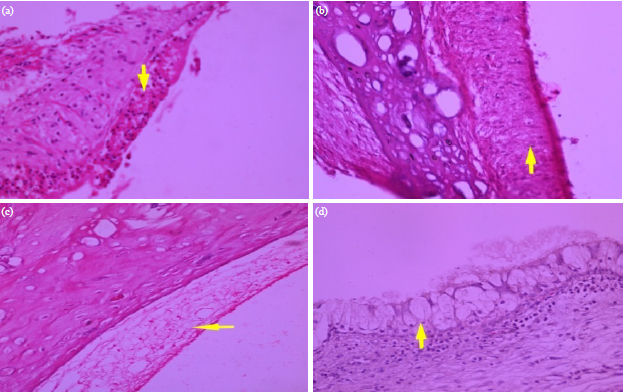
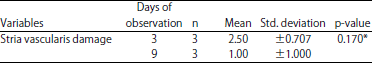Research Article
Light Microscope Study of Intratympanic Methylprednisolone Effect on Cisplatin Induced Ototoxicity
Department of Ear-Nose-Throat-Head-Neck Surgery, Faculty of Medicine, Hasanuddin University, Makassar, Indonesia
Ahmad Ardhani Pratama
Department of Ear-Nose-Throat-Head-Neck Surgery, Faculty of Medicine, Hasanuddin University, Makassar, Indonesia
Abdul Kadir
Department of Ear-Nose-Throat-Head-Neck Surgery, Faculty of Medicine, Hasanuddin University, Makassar, Indonesia
Eka Savitri
Department of Medical Faculty, Faculty of Medicine, Moslem University of Indonesia, Makassar, Indonesia
Andi Nilawati Usman
Department of Halal Center, Faculty of Public Health, Hasanuddin University, Makassar, Indonesia
LiveDNA: 62.15213