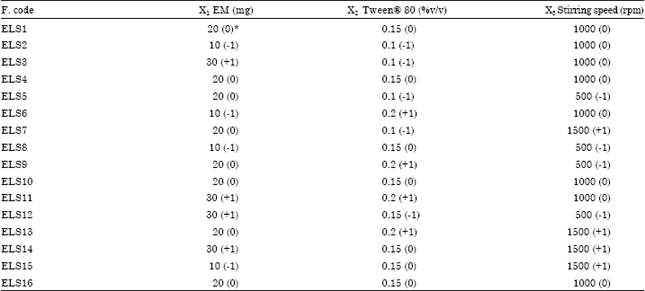
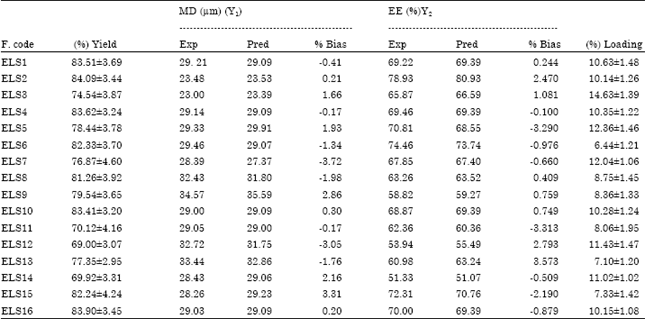
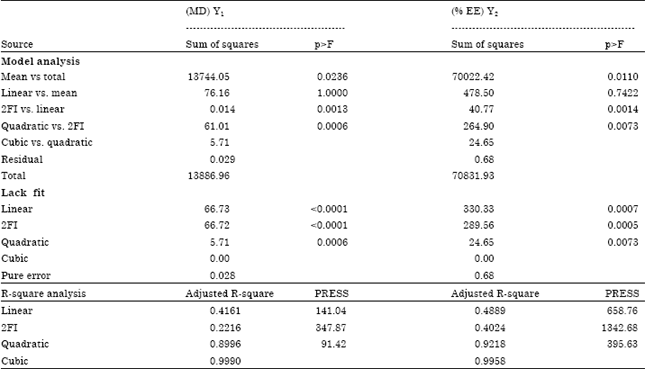
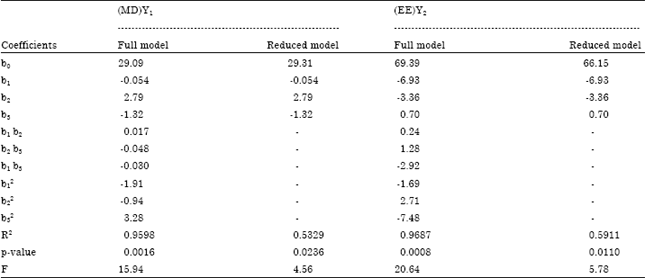
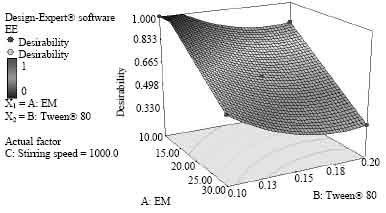
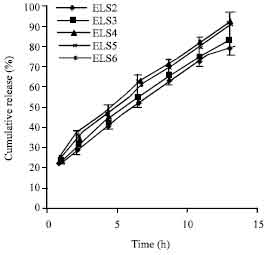
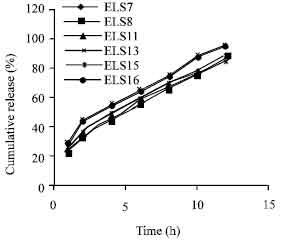
Research Article
Influence of Selected Formulation Variables on the Preparation of Peptide Loaded Lipospheres
University Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur, 492010, India
Deependra Singh
University Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur, 492010, India
Swarnlata Saraf
University Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur, 492010, India