ABSTRACT
Recent advances in molecular genomic analysis showed the possibility of resolving the unexplained mysteries and problems of various biological phenomena. DNA methylation is one of three molecular epigenomic mechanisms involved in controlling gene expression during the process of cellular differentiation, development and aging. This genomic phenomenon has become of great interest in applied research of molecular medicine for its participation in development of various diseases. There is much evidence of possible connection between genomic DNA methylation profiles and psychiatric disorders. Recent studies have demonstrated possible role of DNA methylation in schizophrenia, depression, suicide and bipolar disorder. Aberrations in DNA methylation of several promoter regions of genes have been identified in the genomes of patients with psychiatric disorders. The aim of this effort is to review research work and looking into possible link between abnormal DNA methylation in genomic DNA and psychiatric disorders.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=tmr.2010.1.15
INTRODUCTION
Early studies showed the role of genetic factors and mutations in mood and psychiatric disorders, these were found associated with sexual and aggressive behavior, hysteria, depression, schizophrenia and others (Ibrahim, 1978; Klopfer, 1974). Recent studies proved that epigenetic mechanisms, which are influenced by environmental factors, are involved in various biological phenomena including behavioral and psychiatric disorders. Epigenetics refers to phenotypic changes not explained by coded information in DNA and describes patterns of gene expression caused by mechanisms other than changes in the nucleotide sequence; as a result of active research in this field it was possible to reveal three molecular epigenetic mechanisms which control gene expression: genomic DNA methylation, histone modification and RNAi (Guil and Esteller, 2009; Holliday, 2006; Kawasaki and Taira, 2005; Sledz and Williams, 2005; Strahl and Allis, 2000).
Genomic DNA methylation occurs because of addition of a methyl group at position 5 of the cytosine pyrimidine ring next guanine in CpG dinucleotides; in consequence, DNA methylation disrupts the binding of transcription factors and draws methyl-binding proteins, which are linked with gene silencing and chromatin packaging (Strathdee and Brown, 2002; Weissbach, 1993). It is apparent from foregoing published research results, the success of the human genome sequencing project has created a wide-spread interest in exploring the human genome DNA methylation profile (methylome) in order to elucidate how the genome executes the information it holds (Beck and Rakyan, 2008; Eckhardt et al., 2006; Hocquette, 2005; Lister and Ecker, 2009; McKusick and Ruddle, 1987). One of interesting aspects of methylomes is that although all human’s nucleated cells effectively contain the same genome, they contain very different methylomes (Guil and Esteller, 2009). Specific DNA methylation has been shown to reflect stable, long term characteristics and persistent commitment along a cell type and lineage (Baron et al., 2006). This epigenomic mechanism has been shown to lead to molecular changes which are known to have most important input in development of various types of cancers (Ibrahim, 2010; Jones and Baylin, 2002) and associated with several other developmental phenomena and syndromes e.g., aging (Brunet and Rando, 2007; Ibrahim et al., 2004; Richardson, 2003), fragile X syndrome (Oostra and Willemsen, 2002), Beckwith-Weideman syndrome (Maher and Reik, 2000) and Angelman syndrome (Nicholls et al., 1998). Moreover, research in molecular medicine has shown epigenomic mechanisms are major contributor in the pathogenesis of psychiatric disorders including schizophrenia and bipolar disorder (Tsankova et al., 2007). In this study states of DNA methylations profiles of genes associated with psychiatric disorders are reviewed.
Schizophrenia
It had been reported that the estimated risk of developing schizophrenia over one's lifetime range from 0.3-2.0% with an average of approximately 0.7% (Saha et al., 2005), other studies showed that population wide morbidity is approaching 1% (Grayson et al., 2005; Kohlrausch et al., 2010). Schizophrenia is the most genetically studied psychiatric disorder; however the results of various studies on the genetics basis of schizophrenia have not provided a clarification of underlying etiological factors that define the symptomatology of the disease (Petronis, 2001; Petronis et al., 2003). Another approach for investigating schizophrenia recognized the role of epigenetic factors; this approach has shown possible molecular explanation of this psychiatric disorder. The studies have been focused on possible potential role of DNA methylation states of genes associated with the pathogenesis of schizophrenia (Petronis, 2004; Tamminga and Holcomb, 2005; Tremolizzo et al., 2002).
Mood Disorders
In this study epigenomic states of two types of mood disorders will be reviewed, major depressive disorder and bipolar disorder, these two psychiatric disorders are influenced by both genetic and environmental factors. Bipolar disorder is a chronic illness characterized by shifts between states of mania and depression; recent investigations showed that this disorder affects approximately 2.8% of Americans population (Harvey et al., 2007; Kessler et al., 2005). Mill et al. (2008) indicated that epigenetic misregulation is consistent with various non-mendelian features of bipolar disorder. The other mood disorder, major depressive disorder, is described by an intense sadness, depressed mood and discouragement or loss of interest that persists for at least two weeks; this state impairs the individual’s social functioning and it can also be accompanied by other symptoms, such as irritability, changes in appetite, weight and sleep, decreased energy, feelings of worthlessness or guilt and recurrent thoughts of death (Harvey et al., 2007). One study showed that the incidence of depression was observed in the range 0.9 to 2.0%, this range was distributed among participants randomized to hypnotics (2%) as compared to 0.9% among those randomized in parallel to placebo (Kripke, 2007). Other investigators reported that the lifetime prevalence for mood disorders ranges between 6.6 and 11.9% (Baumeister and Harter, 2007).
Suicide
Suicide is a leading cause of death particularly in males and is considered an act deliberately initiated and performed by a person with full knowledge that a fatal outcome is probable; suicidal behavior is responsible for considerable morbidity and mortality in schizophrenia and bipolar disorder (De Luca et al., 2009; Mann, 2002; Turecki, 2001). It has been reported that according to calculations based on data reported to World Health Organization by its Member States, in 1998 suicide represented 1.8% of the global burden of disease and it is expected to increase to 2.4% by the year 2020 and considered is among the 10 leading causes of death for all ages and it is among the top three causes of death for people aged 15-34 years (Bertolote and Fleischmann, 2002). The results reported in published research showed that alterations in epigenetic markers in suicide victims, might suggest a link between mechanisms that regulate gene expression and major depressive disorder (Autry and Monteggia, 2009).
States of DNA Methylation of Genes Associated with Psychiatricc Disorders
The data presented in Table 1 summarized the results of the research work performed during last decade to investigate the possible involvement of DNA methylation changes in gene expression of several psychic disorders. In the following the involvement of DNA methylation states of genes associated with psychiatric disorders will be reviewed.
It was noted that among greater than 100 different genes examined, reelin protein gene (RELN ) and glutamic acid decarboxylase gene (GAD67) are the most abnormal in the context of schizophrenia and bipolar illness (Torrey et al., 2005). RELN gene is still under extensive investigation for its role in schizophrenia and other psychiatric disorders; this gene is coding for reelin protein, which is necessary for neuronal migration and synaptogenesis. Reelin is a glycoprotein that is expressed during development and was found important for appropriate neural positioning during brain development (Grayson et al., 2005, 2006). An investigation was carried out by Abdolmaleky et al. (2004, 2005) showed that protein production of reelin is reduced in patients suffering schizophrenia and bipolar disorder, suggesting hypermethylation of the promoter region in these disorders. However, the results of a study performed by Tochigi et al. (2008) did not confirm the hypermethylation of the RELN promoter region in the brains of schizophrenic patients, in their investigation they examined the DNA methylation status of the promoter region of RELN by using the pyrosequencing method in the prefrontal cortices of 14 patients with schizophrenia and 13 control subjects.
| Table 1: | States of DNA methylation associated with psychiatric disorders |
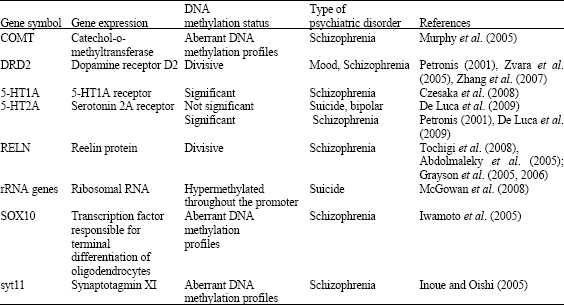 | |
However, Tochigi and his collaborators noted that their results did not rule out a possibility that DNA methylation of the RELN promoter region plays a role in the pathophysiology of schizophrenia. It is worth mentioned that prefrontal cortex and cerebellar expression of RELN mRNA was significantly decreased by 30 to 50% in patients with schizophrenia or bipolar disorder with psychosis, but not in those with unipolar depression without psychosis when compared with non psychiatric subjects (Guidotti et al., 2000).
It is worth noting the results of other research groups about DNA methylation status of RELN gene. Chen et al. (2002) emphasized on the evidence that the RELN promoter contains a large CpG island, this suggested of possible role of DNA methylation in regulating its expression. Whereas, Grayson et al. (2005) analyzed the extent and pattern of methylation within the CpG island of the reelin promoter in genomic DNA isolated from cortices of schizophrenia patients and non psychiatric subjects. Specific regions in the promoter were amplified after bisulphite treatment and sequenced. Their results showed that within the promoter region there were interesting regional variations. There was increased methylation at positions -134 and -139; this promoter region binds a protein present in neuronal precursor nuclear extracts that express very low levels of reelin mRNA; i.e., an oligonucleotide corresponding to this region and that contains methylated cytosines binds more tightly to extracts from non expressing cells than the non methylated counterpart. Collectively, their data showed that this promoter region has positive and negative properties and that the function of this complex cis element relates to its methylation status (Grayson et al., 2005).
Other investigators reported the effect of methionine administration on the methylation status of promoter region of RELN gene, the results showed hypermethylation of the promoter and this caused down regulation of reelin transcription in the mouse model of schizophrenia (Dong et al., 2005; Tremolizzo et al., 2002). In this context the earliest observation was made thirty years ago by group of clinical investigators who were able to observe the effects of methionine in schizophrenia (Antun et al., 1971). By contrast, treatment with the methylation inhibitor 5-aza-2'-deoxycytidine upregulated reelin expression in vitro (Chen et al., 2002).
Czesaka et al. (2008) investigated DNA methylation profiles of the serotonin (5-HT) system which is strongly implicated in major depression and suicide and the 5-HT1A receptor is considered a critical regulator of serotonergic activity. They hypothesized increased DNA methylation of the 5-HT1A polymorphism may increase risk for depression or suicide. In their experiment genomic DNA was isolated from cortical brain tissue and analyzed for methylation aberration using modified bisulfite procedure. DNA methylation was examined at two CpG sites: C(-1019) (POLY) and C(-1007) (HES) in the Deaf-1/ HES5 elements. Methylation of POLY was low (< 10%), detected in schizophrenia, alcoholism and controls. Methylation of HES ranged from 0 to15%, with three fold greater methylation in schizophrenia vs. non schizophrenia or depressed patient samples.
HT2A gene is coding for serotonin 2A receptor, has been implicated in the pathogenesis of suicidal behavior; De Luca and his associates were able to develop an improved quantitative assay for the measurement of allele-specific methylation of the 5-HT2A gene and found that the methylation of the C allele in the pre-frontal cortex of heterozygous suicide victims, was not significantly different in comparison with the non-suicide group, but methylation analysis of the C allele in white blood cell DNA from bipolar and schizophrenic attempters showed a significant difference in the schizophrenic attempters but not in the bipolar attempters (De Luca et al., 2009).
DRD2 gene which is coding for dopamine receptor D2 has been found to be over-expressed in peripheral blood lymphocytes of schizophrenics (Zvara et al., 2005). In addition, different methylation patterns in the partial promoter region of DRD2 have been detected in the PBL of two pairs of schizophrenic monozygotic twins (Petronis et al., 2003). In contrast to this an investigation of the status of DNA methylation in a typical CpGs island in the 5'-regulatory region of DRD2 was ascertained by bisulphite sequencing in schizophrenics of 48 discordant sib pairs with schizophrenia, did not support site-specific cytosine methylation of DRD2 as playing a role in the psychopathology of schizophrenia (Zhang et al., 2007).
Iwamoto et al. (2005) noticed that the cytosine-guanine dinucleotide (CpG) island of sex-determining region Y-box containing gene SOX10, coding for an oligodendrocyte-specific transcription factor, tended to be highly methylated in brains of patients with schizophrenia, this is correlated with reduced expression of SOX10. The authors concluded DNA methylation status of the SOX10 CpG island could be an epigenetic sign of oligodendrocyte dysfunction in schizophrenia.
Murphy et al. (2005) investigated the methylation status of COMT gene which is located on chromosome 22q11, the gene is coding for catechol-o-methyltransferase. This gene has been considered a strong candidate for schizophrenia susceptibility. They examined 31 brain regions and 51 individual blood samples to ascertain the cytosine DNA methylation profile of the COMT promoter, their results ruled out COMT promoter methylation as a common cause of schizophrenia. But they reported unique observation of a completely methylated cytosine at one site in one patient, this may have the potential to affect COMT mRNA transcription and gene activity, but they indicated that this observation remains to be evaluated.
Inoue and Oishi (2005) studied the effects of methylation status of the promoter region of syt11 gene on the expression of human synaptotagmin XI. This gene (syt11) is implicated in the onset of schizophrenia, sequence analysis showed that cytosine residues not in the CpG sequence but still within the promoter region of the gene, are partially methylated.
McGowan et al. (2008) studied the rRNA genes that encode ribosomal RNA in the genome of suicide brain, these are the backbone of the protein synthesis machinery and levels of rRNA gene promoter methylation determine rRNA transcription. They found that coding region for rRNA was significantly hypermethylated throughout the promoter and 5- regulatory region in the brain of suicide subjects, consistent with reduced rRNA expression in the hippocampus. This difference in rRNA methylation was not evident in the cerebellum and occurred in the absence of genome-wide changes in methylation, as assessed by nearest neighbor. They suggested that their data implicated the epigenetic modulation of rRNA in the pathophysiology of suicide.
The involvement of DNA methyltransferases in psychiatric disorders has been investigated, this is because addition of the methyl group at cytosine ring of 5--CpG-3- sequence is catalyzed by one of three DNA methyl transferases (DNMT1, DNMT3a and DNMT3b) with S-adenosyl methionine as the methyl donor (Bird,1992). The DNMT3 family establishes the initial CpG methylation pattern de novo, whereas DNMT1 maintains this pattern during chromosome replication (Chen and Li, 2006; Cheng and Blumenthal, 2008; Hermann et al., 2004). Poulter et al. (2008) demonstrated that DNA methyltransferase expression (DNMT3b) is increased in suicide completers compared with control subjects in frontopolar cortex. Additionally the authors observed, this increase was more pronounced in female versus male postmortem tissue and emphasized that such sexual dimorphism is of note, since, Major Depression Disorder (MDD) is twice as prevalent in women. Their study further revealed that DNMT3b upregulation may contribute to hypermethylation of the gamma-aminobutyric acid type A (GABAA) receptor promoter, thereby potentially explaining the down regulation of GABA-A expression in suicide completers (Poulter et al., 2008). It is worth noting that cortical dysfunction in schizophrenia is associated with changes in GABAergic circuitry, including altered expression of the 67 kDa isoform of glutamic acid decarboxylase (GAD67), an enzyme for GABA synthesis in cortical interneurons (Benes and Berretta, 2001) . Huang and Akbarian (2007) reported significant decrease in CpG methylation at the proximal GAD1 promoter in contrast to what was expected that subjects with schizophrenia should show increased GAD1 DNA methylation. The authors suggested further studies are necessary in order to determine if these changes are related to altered GAD1 gene transcription, notably that the schizophrenia subjects in their study had lower GAD1 mRNA levels in comparison to the matched control (Huang and Akbarian, 2007). In this respect it is worth noting that aberrant methylation of the CpGs in regulatory region in genome of cancer patients affected gene expression by changing binding affinity by means of transcription factors (Costello et al., 2000).
An extensive study carried out by Siegmund and his group, they were able to identify DNA methylation status at 50 loci, encompassing primarily 59 CpG islands of genes related to CNS growth and development, in temporal neocortex of 125 subjects ranging in age from 17 weeks of gestation to 104 years old. Two psychiatric diseases were included Alzheimer and schizophrenia. Disease-associated changes were limited to 2/50 loci in the Alzheimer’s cohort, which appeared to reflect an acceleration of the age-related change in normal brain. The methylation of PAX8, a gene encoding a paired box containing transcription factor important for CNS and thyroid development, was higher in schizophrenics than in controls but this was not considered statistically significant , thus they concluded that schizophrenia is not accompanied by consistent DNA methylation changes at the 50 gene loci included in their study (Siegmund et al., 2007).
Epigenetic Therapy of Behavioral and Psychiatric Disorders
As mentioned earlier, the correlation between psychiatric disorders and the states of genomic methylation is under extensive investigation. The reported results by Mill et al. (2008) and other investigators are consistent with possible epigenetic role of DNA methylation changes in schizophrenia, bipolar disorder and others psychiatric disorders. Knowing that epigegnetic mechanisms are reversible, hence it is possible to suggest that epigenetic defects might be repaired. Thus there is possibility to discover and to use suitable drugs that can amend epigenomic defects; this area of research is called pharmacogenomic or epigenomic therapy (Zhang et al., 2008). It is expected that epigenomic therapy can help preventing progression of the disease caused by epigenetic defects. There are two groups of drugs which have received the most attention, DNA methyltransferase inhibitors and histone deacetylase inhibitors (Gavin and Sharma, 2010; Kirk et al., 2008). In view of growing interest in the epigenetic therapies, it is worth mentioned the success of epigenetic drugs in cancer treatment, already there are applications of DNA methylation inhibitors in treatments of cancers. Two types of DNA methylation inhibitors, azacitidine and decitabine, have generated much interest in cancer therapies (Ibrahim, 2010).
It is possible to suggest there is a possibility that DNA methylation inhibitors might have applications in psychiatric therapy. In this respect it was suggested agents which can reactivate gene expression, such as inhibitors of DNMT1 might therefore provide improved pharmacological treatment for schizophrenia (Grayson et al., 2006). On the other hand there are indications of possible use of other types of epigenetic drugs for treatment of psychiatric disorders. Phiel et al. (2001) reported that valproic acid which is one of epigenetic drugs is used widely to treat epilepsy and bipolar and showed that valproic acid action is through inhibition of histone deacetylase (HDAC1). The administration of valproate in conjunction with antipsychotic medication had been shown to accelerate the onset of the antipsychotic effects in patients with schizophrenia (Casey et al., 2003). It is interesting to note that valproic acid has shown promising antineoplastic effects when used concurrently and may increase the antitumor efficacy of current cytotoxic agents (Chavez-Blanco et al., 2006).
There are reports which indicate that HDAC inhibitors reduce DNA methylation, although the mechanisms by which HDAC inhibitors reduce DNA methylation are unknown, it is thought that hyperacetylation can regulate the accessibility of DNMT1 to promoter regions or that it might induce DNA demethylase activity (Cervoni and Szyf, 2001; Cervoni et al., 2002).
Some nutritional components such as S-adenosyl-methionine (SAM) and sulfophrane can mitigate changes in DNA methylation and chromatin structure akin to those observed by classical drugs used to treat psychopathology, such as valproic acid and the monoamine oxidase inhibitors. Several investigators speculated that nutritional components, especially those to which humans are exposed developmentally or via sustained exposure, particularly to those which act to modify chromatin, will have effects on mental health and risk for psychopathology. The possible involvement of DNA methylation in schizophrenia implies that pharmacological and nutritional agents, for example methionine treatment which increases SAM levels in the brain, to aggravate schizophrenia (Brune and Himwich, 1962; Israelstam et al., 1967). Other research focused on folate deficiency, this seems to be associated with depression; however the evidence for an association between aberrant folate status and schizophrenia seems less convincing (Muntjewerff and Blom, 2005). Another study showed the possible interactions between dietary components that modify the DNA methylation machinery and their effects on mental health in humans, this may be found in the effects of SAM in mood disorders (McGowan and Kato, 2008). Other investigators have found SAM to have antidepressive effects (Papakostas et al., 2003). Interestingly, in one study, nine of 11 patients with bipolar depression treated with SAM switched to mania, suggesting a specific effect of SAM on bipolar depression (Carney et al., 1989). Other investigators reported that methionine infusion reverses the effect of maternal behavior on DNA methylation, nerve growth factor-inducible protein-A binding to the exon 1(7) promoter, glucocorticoid receptor (GR) expression and hypothalamic-pituitary-adrenal and behavioral responses to stress, suggesting a causal relationship among epigenomic state, GR expression and stress responses in the adult offspring. The investigators concluded that these results demonstrated that, despite the inherent stability of the epigenomic marks established early in life through behavioral programming, they are potentially reversible in the adult brain (Weaver et al., 2005).
Impact of Environmental Factors on Psychiatric Disorders
Epigenetics provides the link between the environment and the development of diseases (Sharma, 2005). Environmental factors are capable of causing epigenetic changes in DNA that can potentially alter imprint gene expression and that can result in genetic diseases including cancer and behavioral disorders (Jirtle et al., 2000). The influence of environmental factors on epigenetic mechanisms in general and DNA methylation in particular, have been studied and well documented in recent published literatures and there are indications which prove that these changes in turn can be inherited by daughter cells during cell division and can also be inherited through the germ line (Baccarelli and Bollati,2009; Heindel et al., 2006; Perera et al., 2009).
Environmental factors affecting DNA methylation include lifestyles, stress, famine, diet, proteins, drugs, hormones and others. Investigators have provided evidences on the affect of various environmental factors on the development of psychiatric disorders. Twin, family and adoption studies have demonstrated that schizophrenia is a multifactorial disorder in which genetic and environmental elements contribute to overall risk (Corrigall and Murray, 1994; Ingraham and Kety, 2000; Sullivan et al., 2003). The influences of famine and nutrition on prevalence of psychiatric disorders have been under investigation. One study demonstrated that prenatal exposure to famine significantly increases risk of schizophrenia in later life (St Clair et al., 2005). On the other hand several studies have supported the affect of nutrition, notably pre-natal nutrition was found to exert a significant role in the etiology and severity of schizophrenia (Abel, 2004; Davis and Bracha, 1996; Dubertret et al., 2004; Glatt and Jonsson, 2006; McDonald and Murray, 2000).
There are indications that exposure to an unfavorable prenatal environment considerably increases the risk of psychiatric and behavioral disorders later in life, e.g., schizophrenia (Penner and Brown, 2007) and autism (Kinney et al., 2008). Experiments with laboratory animals indicated that prenatal stress can disrupt brain development (Mulder et al., 2002), through several mechanisms e.g., alteration of gene expression in neurons (Meaney and Szyf, 2005).
A number of studies have demonstrated an effect of the pre-natal environment upon susceptibility to eating disorders, with low birth weight repeatedly identified as a risk factor (Cnattingius et al., 1999; Favaro et al., 2006; Lindberg and Hjern, 2003; Procopio and Marriott, 2007). Similarly, the pre-natal environment has been linked to the risks of developing Attention-Deficit Hyperactivity Disorder (ADHD), diagnosed among school children. This disorder is characterized by deficient attention and problem solving, along with hyperactivity and difficulty with holding incorrect responses; several biological and environmental factors have also been proposed as risk factors for ADHD, including food additives, diet, lead contamination, cigarette and alcohol exposure, maternal smoking during pregnancy and low birth weight (Banerjee et al., 2007).
Future Research
Human’s methylome has an important feature that is its response to environmental factors. There is increasing evidence which suggests that environmental exposures early in development have a role in susceptibility to disease in later life, in addition, some of these environmental effects seem to be passed on through subsequent generations (Baccarelli and Bollati, 2009; Heindel et al., 2006; Jirtle and Skinner, 2007; Perera et al., 2009). In this respect psychiatric disorders are affected by environmental factors and this might due to epigenomic basis of these disorders and possibly DNA methylation is at the epicenter of epigenomic mechanisms. It is expected epigenomic research can give explanation for the relationship between an individual's epigenetic background, the environment and mental health. It can do so because the epigenetic state is affected by various external environmental factors, these will make the methylome varies among individuals and during a lifetime, whereas the DNA sequence remains essentially the same. It is worth mentioned an epigenetic contribution to disease was first realized three decades ago, when it was possible to distinguish human cancer from normal tissues based on DNA methylation levels (Feinberg and Vogelstein, 1983) . To date, it appears that malignancy represents most of the diseases for which epigenetic defects have been shown to contribute to disease pathogenesis (Claus and Lubbert, 2003; Ibrahim, 2010). Thus, it is interesting to consider that epigenetic may play same role in human mental health. Epidemiological research suggested that both an individual's genes and the environment underlie the pathophysiology of psychiatric disorders. There are published results, although some are divisive, have shown possible role of aberrant DNA methylation profiles in promoter regions of genes associated with schizophrenia and other psychiatric disorders. To clarify the role of DNA methylation of the RELN promoter in schizophrenia, further molecular studies are required. Disregulation of the serotonin (5-HT) system is strongly implicated in major depression and suicide and the 5-HT1A receptor is a critical regulator of serotonergic activity, it is expected to develop pharmacogenomic drugs to reform this defect (Czesaka et al., 2008). Research in this field is likely to lead to the development and discovery of epigenetic drugs especially from plant origin (Kirk et al., 2008) for treatment of various psychiatric disorders. Further more future studies might investigate the possible role of genomic repetitive sequences in psychiatric disorders (Batzer and Deininger, 2002; Collier, 2002) and utilize molecular genomic markers for early diagnosis and follow up therapy of psychiatric disorders; currently possible use of specific DNA markers for diagnosis or prognosis is under active investigation in cancer research (Ibrahim, 2010; Ibrahim et al., 2009, 2010a, b; Saleh et al., 2010).
CONCLUSIONS
Molecular studies gave evidence that epigenetic regulation is involved in psychiatric disorders such as schizophrenia; bipolar disorder, depression and suicide. DNA methylation profiles of promoter regions of a number of loci were found being altered in the genomes of individuals with psychiatric disorders. Epigenomic studies revealed that environmental conditions affect state of methylome and this has direct impact on gene expression of acquired mental health problems and might cause genomic imprinting allowing environmental effects to be passed through subsequent generations. Epigenomic therapy is promising and might assist in treatment and curing of these disorders.
ACKNOWLEDGMENT
The author would like to thank International Institute of Education (IIE) for the fellowship.
REFERENCES
- Abdolmaleky, H.M., C.L. Smith, S.V. Faraone, R. Shafa, W. Stone, S.J. Glatt and M.T. Tsuang, 2004. Methylomics in psychiatry: Modulation of gene-environment interactions may be through DNA methylation. Am. J. Med. Genet. B Neuropsychiatr. Genet., 127B: 51-59.
PubMed - Abdolmaleky, H.M., K.H. Cheng, K.H., A. Russo, C.L. Smith and S.V. Faraone et al., 2005. Hypermethylation of the reelin (RELN) promoter in the brain of schizophrenic patients: A preliminary report. Am. J. Med. Genet. B Neuropsychiatr. Genet., 134B: 60-66.
PubMed - Abel, K.M., 2004. Foetal origins of schizophrenia: Testable hypotheses of genetic and environmental influences. Br. J. Psychiatry, 184: 383-385.
PubMed - Antun, F.T., G.B. Burnett, A.J. Cooper, R.J. Daly, J.R. Smythies and A.K. Zealley, 1971. The effects of L-methionine (without MAOI) in schizophrenia. J. Psychiatr. Res., 8: 63-71.
PubMed - Autry, A.E. and L.M. Monteggia, 2009. Epigenetics in suicide and depression. Biol. Psychiatry, 66: 812-813.
CrossRef - Baccarelli, A. and V. Bollati, 2009. Epigenetics and environmental chemicals. Curr. Opin. Pediatrics, 21: 243-251.
CrossRef - Banerjee, T.D., F. Middleton and S.V. Faraone, 2007. Environmental risk factors for attention-deficit hyperactivity disorder. Acta Paediatr., 96: 1269-1274.
CrossRef - Baron, U., I. Turbachova, A. Hellwag, F. Eckhardt and K. Berlin et al., 2006. DNA methylation analysis as a tool for cell typing. Epigenetics, 1: 55-60.
PubMed - Batzer, M.A. and P.L. Deininger, 2002. ALu repeats and human genomic diversity. Nat. Rev. Genet., 3: 370-380.
CrossRef - Baumeister, H. and M. Harter, 2007. Prevalence of mental disorders based on general population surveys. Soc. Psychiatry Psychiatr. Epidemiol., 42: 537-546.
PubMed - Beck, S. and V.K. Rakyan, 2008. The methylome: Approaches for global DNA methylation profiling. Trends. Genet., 24: 231-237.
CrossRef - Benes, F.M. and S. Berretta, 2001. GABAergic interneurons: Implications for understanding schizophrenia and bipolar disorder. Neuropsychopharmacology, 25: 1-27.
PubMed - Bertolote, J.M. and A. Fleischmann, 2002. Suicide and psychiatric diagnosis: A worldwide perspective. World Psychiatry, 1: 181-185.
PubMed - Brune, G.G. and H.E. Himwich, 1962. Effects of methionine loading on the behavior of schizophrenic patients. J. Nerv. Ment. Dis., 134: 447-450.
PubMed - Carney, M.W., T.K. Chary, T. Bottiglieri and E.H. Reynolds, 1989. The switch mechanism and the bipolar/unipolar dichotomy. Br. J. Psychiatry, 154: 48-51.
PubMed - Cervoni, N., N. Detich, S.B. Seo, D. Chakravarti and M. Szyf, 2002. The oncoprotein Set/TAF-1β, an inhibitor of histone acetyltransferase, inhibits activedemethylation of DNA, integrating DNA methylation and transcriptional silencing. J. Biol. Chem., 277: 25026-25031.
PubMed - Cervoni, N. and M. Szyf, 2001. Demethylase activity is directed by histone acetylation. J. Biol. Chem., 276: 40778-40787.
CrossRef - Casey, D.E., D.G. Daniel, A.A. Wassef, K.A. Tracy and P. Wozniak et al., 2003. Effect of divalproex combined with olanzapine or risperidone in patients with an acute exacerbation of schizophrenia. Neuropsychopharmacology, 28: 182-192.
PubMed - Chen, T. and E. Li, 2006. Establishment and maintenance of DNA methylation patterns in mammals. Curr. Top. Microbiol. Immunol., 301: 179-201.
PubMed - Chen, Y., R.P. Sharma, R.H. Costa, E. Costa and D.R. Grayson, 2002. On the epigenetic regulation of the human reelin promoter. Nucleic Acids Res., 30: 2930-2939.
PubMed - Cheng, X. and R.M. Blumenthal, 2008. Mammalian DNA methyltransferases: A structural perspective. Structure, 16: 341-350.
PubMed - Claus, R. and M. Lubbert, 2003. Epigenetic targets in hematopoetic malignancies. Oncogene, 22: 6489-6496.
CrossRef - Cnattingius, S., C.M. Hultman, M. Dahl and P. Sparen, 1999. Very preterm birth, birth trauma and the risk of anorexia nervosa among girls. Arch. Gen. Psychiatry, 56: 634-638.
PubMed - Collier, D.A., 2002. FISH, flexible joints and panic: Are anxiety disorders really expressions of instability in the human genome?. Br. J. Psychiatry, 181: 457-459.
PubMed - Corrigall, R.J. and R.M. Murray, 1994. Twin concordance for congenital and adult-onset psychosis: A preliminary study of the validity of a novel classification of schizophrenia. Acta Psychiatr. Scand., 89: 142-145.
PubMed - Costello, J.F., M.C. Fruhwald, D.J. Smiraglia, L.J. Rush and G.P. Robertson, 2000. Aberrant CpG-island methylation has non-random and tumour-type-specific patterns. Nat. Genet., 24: 132-138.
PubMed - Czesaka, M., J. Lua, C.A. Stockmeierb, M.C. Austin, H.Y. Meltzer and P.R. Albert, 2008. DNA methylation at 5-HT1A receptor promoter C(−1019)G polymorphism CpG sites in schizophrenia and depression. J. Affective Disorders, 107: S53-S122.
CrossRef - Davis, J.O. and H.S. Bracha, 1996. Prenatal growth markers in schizophrenia: A monozygotic co-twin control study. Am. J. Psychiatry, 153: 1166-1172.
PubMed - De Luca, V., E. Viggiano, R. Dhoot, J.L. Kennedy and A.H.C. Wong, 2009. Methylation and QTDT analysis of the 5-HT2A receptor 102C allele: Analysis of suicidality in major psychosis. J. Psychiatric Res., 43: 532-537.
PubMed - Dong, E., R.C. Agis-Balboa, M.V. Simonini, D.R. Grayson, E. Costa and A. Guidotti, 2005. Reelin and glutamic acid decarboxylase67 promoter remodeling in an epigenetic methionine-induced mouse model of schizophrenia. Proc. Natl. Acad. Sci. USA., 102: 12578-12583.
CrossRefPubMedDirect Link - Dubertret, C., L. Gouya, N. Hanoun, J.C. Deybach, J. Ades, M. Hamon and P. Gorwood, 2004. The 3 region of the DRD2 gene is involved in genetic susceptibility to schizophrenia. Schizophr. Res., 67: 75-85.
PubMed - Eckhardt, F., J. Lewin, R. Cortese, V.K. Rakyan and J. Attwood et al., 2006. DNA methylation profiling of human chromosomes 6, 20 and 22. Nat. Genet., 38: 1378-1385.
PubMed - Favaro, C., A.E. Tenconi and P. Santonastaso, 2006. Perinatal factors and the risk of developing anorexia nervosa and bulimia nervosa. Arch. Gen. Psychiatry, 63: 82-88.
PubMed - Feinberg, A.P. and B. Vogelstein, 1983. Hypomethylation distinguishes genes of some human cancers from their normal counterparts. Nature, 301: 89-92.
PubMed - Gavin, D.P. and R.P. Sharma, 2010. Histone modifications, DNA methylation and Schizophrenia. Neurosci. Biobehav. Rev., 34: 882-888.
CrossRef - Glatt, S.J. and E.G. Jonsson, 2006. The Cys allele of the DRD2 Ser311Cys polymorphism has a dominant effect on risk for schizophrenia: evidence from fixed- and random-effects metaanalyses. Am. J. Med. Genet. B Neuropsychiatr. Genet., 141B: 149-154.
PubMed - Grayson, D.R., Y. Chen, E. Costa, E. Dong, A. Guidotti, M. Kundakovic and R.P. Sharma, 2006. The human reelin gene: Transcription factors (+), repressors (-) and the methylation switch (+/-) in schizophrenia. Pharmacol. Ther., 111: 272-286.
CrossRefPubMedDirect Link - Grayson, D.R., X. Jia, Y. Chen, R.P. Sharma, C.P. Mitchell, A. Guidotti and E. Costa, 2005. Reelin promoter hypermethylation in schizophrenia. Proc. Natl. Acad. Sci. USA., 102: 9341-9346.
CrossRefPubMedDirect Link - Guidotti, A., J. Auta, J.M. Davis, V. Di Giorgi-Gerevini and Y. Dwivedi et al., 2000. Decrease in reelin and glutamic acid decarboxylase67 (GAD67) expression in schizophrenia and bipolar disorder: A postmortem brain study. Arch. Gen. Psychiatry, 57: 1061-1069.
PubMed - Guil, S. and M. Esteller, 2009. DNA methylomes, histone codes and miRNAs: Tying it all together. Int. J. Biochem. Cell Biol., 41: 87-95.
PubMed - Harvey, M., P. Belleau and N. Barden, 2007. Gene interactions in depression: Pathways out of darkness. Trends. Genet., 23: 547-556.
CrossRef - Heindel, J.J., K.A. McAllister, L. Worth and F.L. Tyson, 2006. Environmental epigenomics, imprinting and disease susceptibility. Epigenetics, 1: 1-6.
Direct Link - Hermann, A., H. Gowher and A. Jeltsch, 2004. Biochemistry and biology of mammalian DNA methyltransferases. Cell. Mol. Life Sci., 61: 2571-2587.
PubMed - Huang, H.S. and S. Akbarian, 2007. GAD1 mRNA expression and DNA methylation in prefrontal cortex of subjects with schizophrenia. PLoS ONE, 2: e809-e809.
CrossRef - Ibrahim, M.A., 2010. Perspective of DNA methylation in cancer research. Int. J. Cancer Res., 6: 188-201.
CrossRefDirect Link - Ibrahim, M.A., N. Saleh, K.M. Mousawy, N. Al-Hmoud, E. Archoukieh, H.W. Al-Obaide and M.M. Al-Obaidi, 2009. Molecular analysis of RAPD-PCR genomic patterns in age related acute myeloid leukemia. Trends Med. Res., 4: 35-41.
CrossRefDirect Link - Ibrahim, M.A., N. Saleh, E. Archoukieh, H.W. Al-Obaide, M.M. Al-Obaidi and H.M. Said, 2010. Detection of novel genomic polymorphism in acute lymphoblastic leukemia by random amplified polymorphic DNA analysis. Int. J. Cancer Res., 6: 19-26.
CrossRef - Ingraham, L.J. and S.S. Kety, 2000. Adoption studies of schizophrenia. Am. J. Med. Genet., 97: 18-22.
PubMed - Inoue, S. and M. Oishi, 2005. Effects of methylation of non-CpG sequence in the promoter region on the expression of human synaptotagmin XI (syt11). Gene, 348: 123-134.
PubMed - Israelstam, D.M., A. Johnson and H.S. Winchell, 1967. Methionine and schizophrenia. J. Nucl. Med., 8: 325-326.
PubMed - Iwamoto, K., M. Bundo, K. Yamada, H. Takao, Y. Iwayama-Shigeno, T. Yoshikawa and T. Kato, 2005. DNA methylation status of SOX10 correlates with its downregulation and oligodendrocyte dysfunction in schizophrenia. J. Neurosci., 25: 5376-5381.
PubMed - Jirtle, R.L., M. Sander and J.C. Barreft, 2000. Genomic imprinting and environmental disease susceptibility. Environ. Health Perpect., 108: 271-277.
Direct Link - Jirtle, R.L. and M.K. Skinner, 2007. Environmental epigenomics and disease susceptibility. Nat. Rev. Genet., 8: 253-262.
CrossRefPubMedDirect Link - Jones, P.A. and S.B. Baylin, 2002. The fundamental role of epigenetic events in cancer. Nat. Rev. Genet., 3: 415-428.
CrossRefPubMedDirect Link - Kawasaki, H. and K. Taira, 2005. Transcriptional gene silencing by short interfering RNAs. Curr. Opin. Mol. Ther., 7: 125-131.
PubMed - Kessler, R.C., W.T. Chiu, O. Demler, K.R. Merikangas and E.E. Walters, 2005. Prevalence, severity, and comorbidity of twelve-month DSM-IV disorders in the National Comorbidity Survey Replication (NCS-R). Arch. General Psychiatry, 62: 617-627.
Direct Link - Kinney, D.K., K.M. Munir, D.J. Crowley and A.M. Miller, 2008. Prenatal stress and risk for autism. Neurosci. Biobehav. Rev., 32: 1519-1532.
CrossRef - Kirk, H., W.T. Cefalu, D. Ribnicky, Z. Liu and K.J. Eilertsen, 2008. Botanicals as epigenetic modulators for mechanisms contributing to development of metabolic syndrome. Metabolism Clin. Exp., 57: S16-S23.
CrossRef - Kohlrausch, F.B., A. Salatino-Oliveira, C.S. Gama, M.I. Lobato, P. Belmonte de Abreu and M. H. Hutz, 2010. Influence of serotonin transporter gene polymorphisms on clozapine response in Brazilian schizophrenics. J. Psychiatric Res.
CrossRef - Kripke, D.F., 2007. Greater incidence of depression with hypnotic use than with Placebo. BMC Psychiatry, 7: 42-42.
CrossRef - Lindberg, L. and A. Hjern, 2003. Risk factors for anorexia nervosa: A national cohort study. Int. J. Eat Disord., 34: 397-408.
PubMed - Lister, L. and J.R. Ecker, 2009. Finding the fifth base: Genome-wide sequencing of cytosine methylation. Genome Res., 19: 959-966.
PubMed - Maher, E.R. and W. Reik, 2000. Beckwith-Weideman syndrome: Imprinting in cluster revisited. J. Clin. Invest., 105: 247-252.
PubMedDirect Link - McDonald, C. and R.M. Murray, 2000. Early and late environmental risk factors for schizophrenia. Brain Res. Brain Res. Rev., 31: 130-137.
PubMed - McGowan, P.O., A. Sasaki, T.C.T. Huang, A. Unterberger and M. Suderman, et al., 2008. Promoter-wide hypermethylation of the ribosomal RNA gene promoter in the suicide brain. PLoS ONE, 3: e2085-e2085.
CrossRef - McKusick, V.A. and F.H. Ruddle, 1987. Toward a complete map of the human genome. Genomics, 1: 103-106.
CrossRef - Mann, J.J., 2002. A current perspective of suicide and attempted suicide. Ann. Intern. Med., 136: 302-311.
PubMed - Meaney, M.J. and M. Szyf, 2005. Environmental programming of stress responses through DNA methylation: Life at the interface between a dynamic environment and a fixed genome. Dialogues Clin. Neurosci., 7: 103-123.
PubMed - Mulder, E.J.H., P.G.R. de Medina, A.C. Huizink, B.R.H. van den Bergh, J.K. Buitelaar and G.H.A. Visser, 2002. Prenatal maternal stress: Effects on pregnancy and the (unborn) child. Early Hum. Dev., 70: 3-14.
CrossRefPubMedDirect Link - Murphy, B.C., R.L. OReilly and S.M. Singh, 2005. Site-specific cytosine methylation in S-COMT promoter in 31 brain regions with implications for studies involving schizophrenia. Am. J. Med. Genet. B Neuropsychiatr. Genet., 133: 37-42.
PubMed - Nicholls, R.D., S. Saitoh and B. Horsthemka, 1998. Imprinting in prader-willi and angeleman syndromes. Trend Genet., 14: 194-200.
CrossRef - Oostra, B.A. and R. Willemsen, 2002. The X chromosome and fragile X mental retardation. Cytogenet. Gen. Res., 99: 257-264.
CrossRef - Papakostas, G.I., J.E. Alpert and M. Fava, 2003. S-adenosyl-methionine in depression: A comprehensive review of the literature. Curr. Psychiatry Rep., 5: 460-466.
PubMed - Penner, J.D. and A.S. Brown, 2007. Prenatal infectious and nutritional factors and risk of adult schizophrenia. Expert Rev. Neurother, 7: 797-805.
PubMed - Perera, F., W.Y. Tang, J. Herbstman, D. Tang and L. Levin et al., 2009. Relation of DNA methylation of 59-CpG island of ACSL3 to transplacental exposure to airborne polycyclic aromatic hydrocarbons and childhood asthma. PLoS ONE, 4: e4488-e4488.
CrossRef - Petronis, A., 2001. Human morbid genetics revisited: Relevance of epigenetics. Trends. Genet., 17: 142-146.
CrossRef - Petronis, A., 2004. The origin of schizophrenia: Genetic thesis, epigenetic antithesis and resolving synthesis. Biol. Psychiatry, 55: 965-970.
PubMed - Petronis, A., I.I. Gottesman, P. Kan, J.L. Kennedy, V.S. Basile, A.D. Paterson and V. Popendikyte, 2003. Monozygotic twins exhibit numerous epigenetic differences: Clues to twin discordance?. Schizophr. Bull., 29: 169-178.
PubMed - Phiel, C.J., F. Zhang, E.Y. Huang, M.G. Guenther, M.A. Lazar and P.S. Klein, 2001. Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer and teratogen. J. Biol. Chem., 276: 36734-36741.
CrossRef - Poulter, M.O., L. Du, I.C. Weaver, M. Palkovits, G. Faludi and Z. Merali et al., 2008. GABAA receptor promoter hypermethylation in suicide brain: Implications for the involvement of epigenetic processes. Biol. Psychiatry, 64: 645-652.
PubMed - Procopio, M. and P. Marriott, 2007. Intrauterine hormonal environment and risk of developing anorexia nervosa. Arch. Gen. Psychiatry, 64: 1402-1407.
PubMed - Saha, S., D. Chant, J. Welham and J. McGrath, 2005. A systematic review of the prevalence of schizophrenia. PLoS Med., 2: 413-433.
CrossRef - Saleh, N., M.A. Ibrahim, E. Archoukieh, A. Makkiya, M. Al-Obaidi and H. Alobydi, 2010. Identification of genomic markers by RAPD-PCR primer in leukemia patients. Biotechnology, 9: 170-175.
CrossRefDirect Link - Sharma, R.P., 2005. Schizophrenia, epigenetics and ligand-activated nuclear receptors: A framework for chromatin therapeutics. Schizophr. Res., 72: 79-90.
PubMed - Siegmund, K.D., C.M. Connor, M. Campan, T. Long and D.J. Weisenberger et al., 2007. DNA methylation in the human cerebral cortex is dynamically regulated throughout the life span and involves differentiated neurons. PLoS One, 2: e895-e895.
CrossRef - Sledz, C.A. and B.R. Williams, 2005. RNA interference in biology and disease. Blood, 106: 787-794.
CrossRef - Strahl, B.D. and C.D. Allis, 2000. The language of covalent histone modifications. Nature, 403: 41-45.
PubMed - St Clair, D., M. Xu, P. Wang, Y. Yu and Y. Fang et al., 2005. Rates of adult schizophrenia following prenatal exposure to the Chinese famine of 1959-1961. J. Am. Med. Assco., 294: 557-562.
PubMed - Strathdee, G. and R. Brown, 2002. Aberrant DNA methylation in cancer: Potential clinical interventions. Expert. Rev. Mol. Med., 4: 1-17.
Direct Link - Sullivan, P.F., K.S. Kendler and M.C. Neale, 2003. Schizophrenia as a complex trait: Evidence from a meta-analysis of twin studies. Arch. Gen. Psychiatry, 60: 1187-1192.
PubMed - Tamminga, C.A. and H.H. Holcomb, 2005. Phenotype of schizophrenia: A review and formulation. Mol. Psychiatry, 10: 27-39.
PubMed - Tochigi, M., K. Iwamoto, M. Bundo, A. Komori and T. Sasaki et al., 2008. Methylation status of the reelin promoter region in the brain of schizophrenic patients. Biol. Psychiatry, 63: 530-533.
CrossRef - Torrey, E.F., B.M. Barci, M.J. Webster, J.J. Bartko, J.H. Meador-Woodruff and M.B. Knable, 2005. Neurochemical markers for schizophrenia, bipolar disorder and major depression in postmortem brains. Biol. Psychiatry, 57: 252-260.
CrossRef - Tremolizzo, L., G. Carboni , W.B. Ruzicka, C.P. Mitchell and I. Sugaya et al., 2002. An epigenetic mouse model for molecular and behavioral neuropathologies related to schizophrenia vulnerability. Proc. Natl. Acad. Sci. USA., 99: 17095-17100.
PubMed - Tsankova, N., W. Renthal, A. Kumar and E.J. Nestle, 2007. Epigenetic regulation in psychiatric disorders. Nat. Rev. Neurosci., 8: 355-367.
CrossRef - Turecki, G., 2001. Suicidal behavior: Is there a genetic predisposition?. Bipolar, 3: 335-349.
PubMed - Weaver, I.C., F.A. Champagne , S.E. Brown , S. Dymov and S. Sharma et al., 2005. Reversal of maternal programming of stress responses in adult offspring through methyl supplementation: Altering epigenetic marking later in life. J. Neurosci., 25: 11045-11054.
PubMed - Zhang, W., R.S. Huang and M.E. Dolan, 2008. Integrating epigenomics in to pharmacogenomic studies. Pharmacogenomics Personalized Med., 1: 7-14.
Direct Link - Zhang, A.P., J. Yu, J.X. Liu, H.Y. Zhang and Y.Y. Du et al., 2007. The DNA methylation profile within the 5-regulatory region of DRD2 in discordant sib pairs with schizophrenia. Schizophrenia Res., 90: 97-103.
CrossRef - Zvara, A., G. Szekeres, Z. Janka, J.Z. Kelemen C. Cimmer, M. Santha and L.G. Puskas, 2005. Over-xpression of dopamine D2 receptor and inwardly rectifying potassium channel genes in drug-naive schizophrenic peripheral blood lymphocytes as potential diagnostic markers. Dis. Markers, 21: 61-69.
PubMed - Chavez-Blanco, A., C. Perez-Plasencia, E. Perez-Cardenas, C. Carrasco-Legleu and E. Rangel-Lopez et al., 2006. Antineoplastic effects of the DNA methylation inhibitor hydralazine and the histone deacetylase inhibitor valproic acid in cancer cell lines. Cancer Cell Int., Vol. 6.
CrossRefDirect Link








