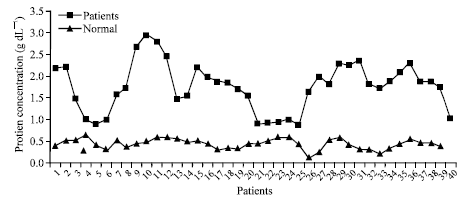
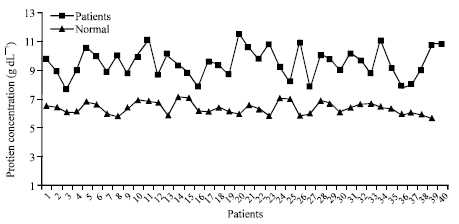
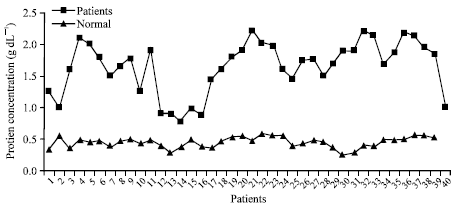
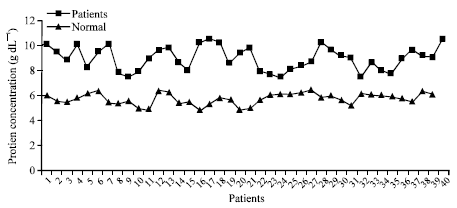
Research Article
Comparative Study on Saliva Proteins in Patients of Brain Tumors and Healthy Individuals
Department of Medical Allied, Zarka University College, Al-Balqa Applied University, Jordan
M.A. Ibrahim
Royal Scientific Society, Amman, Jordan
S.I. Al-Mohtasib
Department of Medical Allied, Zarka University College, Al-Balqa Applied University, Jordan
K.N. Al-Kobasi
Department of Medical Allied, Zarka University College, Al-Balqa Applied University, Jordan